Summary
Synopsis
Formoterol, a long-acting β2-selective adrenoceptor agonist, produces dose-proportional bronchodilation in patients with obstructive airways disease with a reversible component. A significant effect occurs within minutes of inhalation of a therapeutic formoterol dose and persists for approximately 12 hours. Oral formoterol has a slower onset of action than the inhaled formulations, but also produces prolonged bronchodilatory effects.
Inhaled formoterol has shown a therapeutic efficacy equivalent to or better than comparable dosages of the conventional β2-agonists salbutamol, fenoterol and terbutaline in short and long term trials, in both adults and children with asthma. Its prolonged duration of action permits a twice-daily dosage regimen and results in improved control of nocturnal symptoms by reducing the ‘morning dip’. Formoterol also compares well with oral slow release theophylline. In addition, significantly more patients with chronic obstructive airways disease (COAD) had an improvement in symptoms when treated with formoterol compared with salbutamol or fenoterol. Noncomparative studies indicate formoterol also provides effective prophylaxis of exercise-induced asthma. Development of tachyphylaxis has not been observed.
Formoterol is generally well tolerated. Adverse effects observed represent predictable extensions of its pharmacology. Tremor and palpitations are most frequently reported. The incidence of adverse events is dose-proportional and therefore related to the route of administration, being more frequent following oral than inhalation therapy.
The long-acting β2-agonists, including formoterol, represent a significant advance over current maintenance or prophylactic bronchodilator therapy with intermediate-acting β2-agonists such as salbutamol, fenoterol and terbutaline, predominantly because of the twice daily administration regimen. However, comparisons with other long-acting β2-agonists, such as salmeterol, evaluation of its role in improving symptom control in patients failing to respond to prophylactic therapy, and clarification of the optimal role of β2-agonists in asthma maintenance therapy are required to fully determine the value of formoterol in the management of obstructive airways disease.
Pharmacodynamic Properties
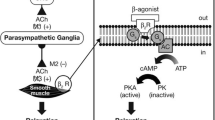
Formoterol is a long-acting selective fo-adrenoceptor agonist which has demonstrated significant bronchodilatory effects. In patients with asthma inhaled formoterol produced rapid, dose-proportional bronchodilation with increases in forced expiratory volume in 1 second (FEV1), forced vital capacity (FVC), peak expiratory flow rate (PEFR) and specific airways conductance (sGaw). Effective doses of inhaled formoterol significantly decreased specific airways resistance within minutes. Maximum bronchodilation was achieved within 2 hours, with effects persisting for approximately 12 hours, significantly longer than the bronchodilatory effects of equivalent doses of salbutamol, fenoterol or terbutaline. Oral formoterol had a slower onset of action than inhaled formoterol but also produced prolonged effects. Formoterol also demonstrated significant antiallergic activity in animals and in healthy volunteers, and in inhaled form had a protective effect in provocation tests in asthmatic patients.
Formoterol produced a dose-proportional increase in heart rate and a decrease in blood pressure in healthy volunteers and patients with asthma, but these changes were small and within physiological norms at therapeutic dosages.
Pharmacokinetic Properties
Following therapeutic doses formoterol concentrations in body tissues and fluids are often below the detection limits of currently available assays. An elimination half-life of 1.7 to 2.3 hours after formoterol inhalation has been calculated in humans. After oral formoterol 40μg or inhaled formoterol 24μg, 9.6% and 24%, respectively, of the dose was recovered from the urine over 24 hours and a glucuronic acid conjugate was identified. It is possible formoterol has a prolonged elimination half-life which has not yet been detected in humans.
Therapeutic Use
The therapeutic efficacy of inhaled or oral formoterol was demonstrated in several short term and a few long term studies of adults with asthma or chronic obstructive airways diseases (COAD) and children with asthma or non-asthmatic respiratory disorders. Noncomparative studies in patients with chronic bronchial asthma showed formoterol improved subjective clinical symptoms and lung function parameters. No evidence of tachyphylaxis was found in trials lasting up to 252 weeks, during which 70 to 100% of asthmatic adults showed some improvement. Reduction in the severity of clinical symptoms, and a significant increase in FEV1, resulted in decreased usage of ‘rescue’ medication.
Symptoms diminished in 64 to 100% of children receiving oral formoterol and in 79 to 87% when formoterol was administered by inhalation. FEV1 and airways resistance also improved during a 2-week study, and the mean frequency of nocturnal and diurnal asthma attacks decreased.
Overall efficacy of formoterol was at least equivalent to and in some trials significantly greater than, an equipotent dose of salbutamol, and diurnal variation in pre-bronchodilator PEFR was significantly less pronounced in patients taking formoterol (12jug twice daily) compared with salbutamol (200μg 4 times daily) [17 vs 42 L/min]. Patients receiving formoterol experienced fewer asthma attacks and required less rescue medication than those taking salbutamol. There was an overall patient preference for formoterol. In patients with nocturnal asthma, a single night-time dose of inhaled formoterol 12μg was more effective than salbutamol 200μg.
Formoterol and fenoterol produced similar improvements in pulmonary function, but formoterol significantly reduced the number of nocturnal asthma attacks from baseline by the third week of treatment whereas fenoterol did not. Formoterol was at least as effective as terbutaline and provided better control of symptoms of nocturnal asthma. Formoterol showed a similar duration of action to slow release theophylline in patients with nocturnal asthma, and demonstrated superior tolerability, although sleep disturbance caused by asthma was more frequent in patients taking formoterol. Combined fenoterol plus ipratropium bromide induced bronchodilation to a similar extent as formoterol monotherapy and both agents were well tolerated. However from 4 to 12 hours after administration, formoterol produced a significantly better effect.
The few published studies assessing prophylaxis of exercise-induced asthma (EIA) have indicated that inhaled formoterol 24μg gives complete protection even when administered up to 8 hours before 5 to 8 minutes of strenuous exercise. Both inhaled formoterol 24/ug and fenoterol 400μg prevented EIA when administered 15 minutes before 6 minutes of exercise. Similarly, formoterol 12μg and salbutamol 200μg prevented EIA when given 2 hours before 5 to 8 minutes of exercise. However, when administered 4 hours prior to exercise, formoterol provided a protective efficacy significantly greater than that of salbutamol.
A number of small noncomparative Japanese trials have shown oral formoterol to reduce symptom severity in 75 to 100% of children with non-asthmatic respiratory disorders. Symptoms disappeared within 1 week of starting therapy in all patients administered formoterol syrup 3 to 4μg/kg/day. A large study of adults with COAD demonstrated therapeutic efficacy in 90% of patients, and 83% preferred formoterol to prior therapy. Significantly more patients with COAD experienced improvement whilst treated with formoterol compared to fenoterol or salbutamol in 2 large parallel studies. FEV1 increased significantly in formoterol recipients during the 2- and 4-week trials but not in patients receiving fenoterol or salbutamol. Further well designed, long term studies in large numbers of patients are required to confirm the above results.
Tolerability
Formoterol is tolerated well in adults and children, the adverse effects experienced being those seen with other β2-agonists. The most frequently reported effects were tremor and palpitation which occurred in 10.7% and 11.5% of asthmatic adults receiving oral formoterol 20 to 40μg once to 4 times daily. Inhaled formoterol 12/ug twice daily was associated with tremor in 6.4% and palpitation in 0.4% of 236 patients. Tachycardia occurred in 1.6% of patients receiving oral formoterol, however in 1 trial with oral formoterol the incidence was 15% (3/20 patients). This was resolved when the oral dosage was reduced from 120 to 60 Mg/day. Headache, cough, dizziness, dry mouth and nausea were occasionally reported.
Overall, the range and incidence of adverse effects observed with formoterol did not appear to be significantly different from that of other β2-agonists. However, it must be emphasised that only limited numbers of patients have been involved in comparative studies to date. As with other β2-agonists, high doses of formoterol may cause marked hypokalaemia.
Dosage and Administration
Inhaled formoterol 12 or 24μg (1 or 2 puffs) twice daily should be used as maintenance therapy. It has recently been recommended that long-acting β2-agonists be reserved for use in asthma patients with symptoms inadequately controlled by inhaled corticosteroids. The manufacturer also recommends that inhaled formoterol may be used as rescue medication, and for prophylaxis against exercise- or allergen-induced bronchoconstriction. No more than 2 puffs in 6 hours should be taken. The maximum recommended daily dose is 6 puffs (72μg). Tablet and dry syrup formulations are available in some countries. Recommended dosage of formoterol tablets is 80/ig 2 or 3 times daily in adults and 1.5 μg/kg 2 or 3 times daily in children. In children, formoterol 4 μg/kg/day in 2 or 3 doses, orally as a dry syrup, has also provided effective control of symptoms. Alternative or additional therapy is required if reduced efficacy of formoterol is noted, as this generally indicates worsening of asthma.
Similar content being viewed by others
References
Arvidsson P, Larsson S, Löfdahl C-G, Melander B, Wåhlander L, et al. Formoterol, a new long-acting bronchodilator for inhalation. European Respiratory Journal 2: 325–330, 1989
Becker AB, Simons FER. Formoterol, a new long-acting selective β2-adrenergic receptor agonist: double-blind comparison with salbutamol and placebo in children with asthma. Journal of Clinical-Immunology 84: 891–895, 1989
Bibi H, Becker AB, Lilley MK, Simons FER, Simons KJ, et al. The effect of inhaled beta-agonists on airway responses to antigen in a canine model of asthma. Abstract 5. Journal of Allergy and Clinical Immunology 85: 295, 1990
Brandt H-J, Dietlein G. Comparison of formoterol versus terbutaline (metered-dose aerosol) in patients with chronic obstructive lung disease. In Gerhard Sybrecht (Ed.) Formoterol — a new long-acting bronchodilator, pp. 51–53, Hans Huber Publishers, Toronto, New York, Bern, Stuttgart, 1988
British Thoracic Society. Guidelines for management of asthma in adults: I. chronic persistent asthma. British Medical Journal 301: 651–653, 1990a
British Thoracic Society. Guidelines for management of asthma in adults: II. acute severe asthma. British Medical Journal 301: 797–800, 1990b
Chuchalin AG, Tsoy AN, Yashina LA, Abazova FI. Comparison of inhaled formoterol and terbutaline in reversible obstructive airways disease. Abstract. European Respiratory Journal 3 (Suppl. 10): 419s, 1990
Clauzel AM. Characteristics of bronchodilating activity of formoterol. Lung (Suppl.): 71–75, 1990
Clauzel AM, Rifal N, Bony C, Michel FB. Formoterol aerosol — a dose finding trial with regard to duration of action. Abstract 57. Journal of Allergy and Clinical Immunology 83: 186, 1989
Clauzel AM, Rifal N, Godard Ph, Bony C, Vergnaud A, et al. Efficacity and tolerance of formoterol long term treatment (12 months) in severe asthmatic patients. Abstract. American Review of Respiratory Disease 141: A206, 1990
Daffner HW. Formoterol in children with reversible obstructive airway disease. Abstract. European Respiratory Journal 1 (Suppl. 2): 38IS, 1988
Debelic M, Wessollek I. Comparison of inhaled formoterol and fenoterol in prevention of exercise-induced asthma. Abstract. European Respiratory Journal 1 (Suppl. 2): 381S, 1988
Decker N, Quennedey MC, Rouot B, Schwartz J, Velley J. Effects of N-aralkyl substitution of β-agonists on α- and β-adrenoceptor subtypes: pharmacological studies and binding assays. Journal of Pharmacy and Pharmacology 34: 107–112, 1982
Derom E, Pauwels R, Van Der Straeten M. Time course of the bronchodilating effect of inhaled formoterol. Abstract. European Respiratory Journal 2 (Suppl. 5): 392S, 1989
Editorial. β2-Agonists in asthma: relief, prevention, morbidity. Lancet 336: 1411–1412, 1990
Fabel H. Nocturnal asthma: comparison of inhaled formoterol with oral slow-release terbutaline. In Barnes PJ & Matthys H (Eds) Formoterol — a new generation fo-agonist, pp. 64–65, Hogrefe and Huber Publishers, Toronto, Lewiston, N.Y., Bern, Göttingen, Stuttgart, 1990
Ferguson H, Thomas KE, Davies RJ. Comparison of single dose formoterol and terbutaline on the immediate asthmatic response to allergen provocation testing. Abstract. Clinical and Experimental Allergy 19: 118, 1989
Firkusny L, Deeg M, Gradin-Frimmer G, Ensslin-Haasis B, Antonin K-H, et al. Untersuchung zur Pharmakodynamik und Resorption des β2-Sympathikomimetikums Formoterol nach Inhalation an gesunden Probanden. Abstract. Klinische Wochenschrift 68 (Suppl. 19): 42, 1990
Freyss-Beguin M, Griffaton G, Lechat P, Picken D, Quennedey MC, et al. Comparison of the chronotropic effect and the cyclic AMP accumulation induced by fo-agonists in rat heart cell culture. British Journal of Pharmacology 78: 717–723, 1983
Funabashi S, Nagayama Y, Honda A. Clinical study of formoterol (BD 40A), a new bronchodilator, in the pediatric patients with non-asthmatic respiratory disorders. Shonika Rinsho 36(9): 2153–2157, 1983
Geisler L. Results of placebo-controlled dose-finding study with formoterol. Abstract. Allergologia et Immunopathologia 15: 308, 1987
Godfrey S. Bronchodilators in exercise-induced asthma. In Clark & Cochrane (Eds) Bronchodilator therapy, pp. 112–130, ADIS Press, Auckland, 1984
Graff-Lonnevig V, Browaldh L. Twelve hours’ bronchodilating effect of inhaled formoterol in children with asthma: a double-blind cross-over study versus salbutamol. Clinical and Experimental Allergy 20: 429–432, 1990
Grönneberg R, Zetterström O. Inhibition of anti-IgE induced skin response in normals by formoterol, a new β2-adrenoceptor agonist, and terbutaline. 1. Dose response relation and duration of effect on the early wheal and flare response. Allergy 45: 334–339, 1990a
Grönneberg R, Zetterström O. Inhibition of anti-IgE induced skin response in normals by formoterol, a new β2-adrenoceptor agonist, and terbutaline. 2. Effect on the late phase reaction. Allergy 45: 340–346, 1990b
Hargreave FE, Dolovich J, Newhouse MT (Eds). The assessment and treatment of asthma: a conference report. Journal of Allergy and Clinical Immunology 85: 1098–1111, 1990
Hedenström H, Wegener T. Onset of bronchodilatation after formoterol inhalation. Abstract. European Respiratory Journal 3 (Suppl. 10): 320S–321S, 1990
Hekking PR, Maesen F, Greefhorst A, Prins J, Tan Y, et al. Long-term efficacy of formoterol compared to salbutamol. Lung (Suppl.): 76–82, 1990
Hiratani M, Muto K, Oshida Y, Ito S, Shirai M, et al. Clinical trial of formoterol (BD40A) in the pediatric patients with bronchial asthma. Kiso to Rinsho 15: 4958–4964, 1981
Honma N, Hirao J, Nagai Y, Tonozuka S, Sato J, et al. Efficacy of BD 40A in the treatment of diseases other than bronchial asthma with the chief complaint of wheezing and coughing. Shonika Rinsho 36: 1611–1616, 1983
Ichimura T, Nagai Y, Fujinuma S, Aoki I, Hirayama T, et al. Clinical experiences with BD 40A (formoterol) dry syrup in the treatment of so-called asthmatic bronchitis syndrome — particularly in regard to dose dependency. Allergy in Practice 28: 59–64, 1983
Ida H. Cardiorespiratory activities of 3-formylamino-4-hydroxy-α-(N-1-methyl-2-p-methoxyphenethylaminomethyl)-benzylalcohol-hemifumarate (BD 40A) and some other β-adrenoceptor stimulants in conscious guinea pigs. Arzneimittel-Forschung 26: 1337–1340, 1976a
Ida H. Comparison of the action of BD 40 A and some other β-adrenoceptor stimulants on the isolated trachea and atria of the guinea pig. Arzneimittel-Forschung 26: 839–842, 1976b
Ida H. General pharmacology of (αRS)-3-formamido-4-hydroxy-α-[[[(αRS)-p-methoxy-α-methylphenethyl]amino]methyl] benzyl alcohol fumarate dihydrate (BD 40A), a new bronchodilator agent. Folia Pharmacologica Japonica 76: 633–654, 1980
Ida H. Pharmacology of formoterol, (αRS)-3-foramido-4-hydroxy-α-[[[αRS)-p-methoxy-α-methylphenethyl]amino]methyl] benzyl alcohol fumarate dihydrate (BD 40A). Oyo Yakuri 21: 201–210, 1981
Itoh S, Kubota M, Mikawa H. Clinical studies on formoterol (BD 40A) in pediatric patients with bronchitis. Shonika Rinsho 35: 2177–2181, 1982
Jeppsson A-B, Löfdahl C-G, Waldeck B, Widmark E. On the predictive value of experiments in vitro in the evaluation of the effect duration of bronchodilator drugs for local administration. Pulmonary Pharmacology 2: 81–85, 1989
Kajita T, Mishima T, Mita H, Shida T. Release of slow-reacting substance of anaphylaxis (SRS-A) from human lung and inhibitory effect by β-stimulants. Japanese Journal of Allergology 33: 43–51, 1984
Kamimura H, Sasaki H, Higuchi S, Shiobara Y. Quantitative determination of the β-adrenoceptor stimulant formoterol in urine by gas chromatography mass spectrometry. Journal of Chromatography 229: 337–345, 1982
Kardos P, Friedrich P. Comparison between four weeks’ treatment with formoterol versus fenoterol in patients with reversible obstructive respiratory tract diseases. In Sybrecht G (Ed.) Formoterol — a new long-acting bronchodilator, pp. 57–59, Hans Huber Publishers, Toronto, 1988
Kawai M, Maekawa N, Kawakami Y, Kimura K, Nagaoka S, et al. Evaluation of formoterol (BD 40A) on clinical effects and safety in long-term repeated administration. Yakuri to Chiryo 11: 4381–4404, 1983
Kawai T, Mitsui T. Clinical evaluation of formoterol, a β-stimulant, in chronic bronchitis. Rinsho Seijin-Byo 13: 1369–1376, 1983
Kawakami Y. Evaluation of therapeutic effects of formoterol, a β-adrenoceptor stimulating bronchodilator, on bronchial asthma — a multicentre double-blind single-dose study. Clinical Evaluation 12: 697–727, 1984
Kawakami Y, Nakajima S, Takahashi T, Noguchi E, Shida T, et al. Studies on the bronchodilator effects of single dose administration of formoterol (BD 40A) tablets in bronchial asthma. Kiso to Rinsho (8): 2637–2648, 1983b
Kawakami Y, Noguchi E, Takahashi T, Shida T, Ito K, et al. Clinical trial of formoterol (BD 40A) on bronchial asthma. Yakuri to Chiryo 11: 3923–3934, 1983a
Kawakami Y, Takenaka S. The clinical evaluation of formoterol (BD40A) in bronchial asthma with long-term administration and in chronic obstructive pulmonary disease with short term administration. Yakuri to Chiryo 11: 3935–3942, 1983
Kesten S, Rebuck AS. Management of chronic obstructive pulmonary disease. Drugs 38: 160–174, 1989
Kesten S, Chapman KR, Broder I, Cartier A, Knight A, et al. A three month comparison of twice daily inhaled formoterol versus four times daily inhaled albuterol in the management of stable asthma. American Review of Respiratory Disease, in press, 1991
Kimura K, Tamura Y, Fujibayashi S, Miura J, Sawada Y, et al. Clinical studies on formoterol (BD 40A) in pediatric patients with bronchial asthma. Shonika Rinsho 34: 2647–2655, 1981
Kito H, Inagaki H, Togari H, Sugiyama K, Kamiya K, et al. Clinical results with BD 40A (formoterol) dry syrup in child bronchial asthma. Shonika Rinsho 36: 2159–2168, 1983
Kono O, Morita M. Evaluation of duration of action and clinical effect on formoterol (BD 40A) in single doses for bronchial asthma. Yakuri to Chiryo 11: 4405–4416, 1983
Kronenberger H, Kullmer T, Schultze-Werninghaus G, Lintl H, Siekmeier R, et al. Bronchodilatory effect of formoterol in healthy smokers and non-smokers. Abstract. American Review of Respiratory Disease 141: A469, 1990
Kronenberger H, Lintl H, Kullmer T, Siekmeier R, Schiller-Scotland C, et al. Evaluation of the bronchodilatory effect of formoterol with a new aerosol method in healthy non smokers. Abstract. European Respiratory Journal 2 (Suppl. 5): 392S, 1989
Kullmer T, Kronenberger H, Lintl H, Schiller-Scotland ChF, Siekmeier R, et al. Post bronchodilator effects of the new β2-mimetic formoterol versus fenoterol on conventional lung-function and effective airway diameters in healthy smokers. American Review of Respiratory Disease 141: A469, 1990
Larsson S, Löfdahl C-G, Arvidsson P. 12 hours bronchodilating effect duration of inhaled formoterol in asthma. Abstract. American Review of Respiratory Disease 141: A27, 1990
Larsson S, Löfdahl CG, Arvidsson P, Svedmyr N. Formoterol, a new long-acting β2-agonist for inhalation in a 12 month comparison with salbutamol. Studies of tachyphylaxis. Abstract. American Review of Respiratory Disease 139: A432, 1989
Leuenberger P, Gebre-Michel I. Preventative effect of formoterol aerosol in methacholine-induced bronchoconstriction. In Davies RJ (Ed.) Formoterol in asthma — clinical profile of a new long-acting inhaled β2-agonist, pp. 17–22, Hans Huber Publishers, Toronto, 1988
Löfdahl C-G, Arvidsson P, Melander B, Svedmyr N, Wåhlorder L, et al. Longer duration of the effects of formoterol, a new β2-adrenoceptor agonist compared with salbutamol in a two-week trial in asthmatic patients. In Sybrecht G (Ed.) Formoterol — a new long-acting bronchodilator, pp. 32–37, Hans Huber Publishers, Toronto, 1987
Löfdahl C-G, Svedmyr N. Formoterol fumarate, a new β2-adrenoceptor agonist. Acute studies of selectivity and duration of effect after inhaled and oral administration. Allergy 44: 264–271, 1989
Lusuardi M, Capelli A, Carli S, Zaccaria S, Donner CF. Effect of formoterol vs salbutamol on Superoxide anion (O2−) production by human alveolar macrophages (HAM). Abstract. European Respiratory Journal 3 (Suppl. 10): 432S–433S, 1990
Maesen FPV, Costongs R, Smeets JJ, Brombacher PJ, Zweers PGMA. The effect of maximal dosages of formoterol and salbutamol from a metered dose inhaler on serum potassium and pulse rate. Abstract. European Respiratory Journal 3 (Suppl. 10): 235s–236s, 1990a
Maesen FPV, Costongs RJMG, Smeets JJ, Zweers PGMA. Dose finding study of different dosages of formoterol dry powder inhalation. Abstract. Chest 98 (Suppl.): 27s, 1990b
Maesen FPV, Smeets JJ, Gubbelmans HLL, Zweers PGMA. Bronchodilator effect of inhaled formoterol vs salbutamol over 12 hours. Chest 97: 590–594, 1990c
Maesen FPV, Smeets JJ, Gubbelmans HLL, Zweers PGMA. Formoterol in the treatment of nocturnal asthma. Chest 98: 866–870, 1990d
Magnussen H. Die protektive Wirkung inhalativer Beta-2-Sym-pathikomimetika. Bronchospasmolyse mit Beta-2-Sympathi-komimetika. Erfahrungen mit d.langwirkenden Dosier-Aerosol Formoterol. 21. Kolloquium d.Bad Reichenhaller Forschungsanst.d. Atmungsorgane, 17 Juni 1988. Hrsg.v. D. Nolte und A. Burghele. Muenchen-Deisenhofen, Dustri, pp. 108–112, 1989
Malo J-L, Cartier A, Trudeau C, Ghezzo H, Gontovnick L. Formoterol, a new inhaled beta-2 adrenergic agonist, has a longer blocking effect than albuterol on hyperventilation-induced bronchoconstriction. American Review of Respiratory Disease 142: 1147–1152, 1990
McAlpine LG, Thomson NC. Prophylaxis of exercise-induced asthma with inhaled formoterol, a long-acting β2-adrenergic agonist. Respiratory Medicine 84: 293–295, 1990
Midgren B, Melander B, Persson G. Formoterol vs salbutamol b.i.d. in stable asthmatics. Abstract. Chest 98 (Suppl.): 265, 1990
Mita H, Shida T. Anti-allergic activity of formoterol, a new beta-adrenoceptor stimulant, and salbutamol in human leukocytes and human lung tissue. Allergy 38: 547–552, 1983
Müller Ph, Milovanovic D, Howald H, Franke B, MacNab M. Tolerability and pharmacodynamic effects of inhaled formoterol in healthy subjects. Abstract. European Journal of Clinical Pharmacology 36 (Suppl.): A281, 1989
Nagano H, Nishimoto Y, Egashira Y, Oyamada M, Yamakido M, et al. Double-blind comparative study of formoterol for chronic obstructive diseases, mainly for chronic bronchitis and pulmonary emphysema in multiple institutions. Igaku no Ayumi 129: 578–594, 1984
Nakajima S, Oishi M, Suetsugu S, Kono O, Mitsui K, et al. Studies on the duration of bronchodilative effects of formoterol. Kiso to Rinsho 17: 3629–3637, 1983a
Nakajima S, Oishi M, Takagi H, Shida T, Nishimura H, et al. Dose-response evaluation of bronchodilation effect of formoterol (BD 40A) in single doses in bronchial asthma. Igaku to Yakugaku 10: 571–582, 1983b
Nakajima S, Okada T, Okayasu M, Kawakami Y, Nakajima H, et al. Evaluation of bronchodilator effect of formoterol (BD 40A) administered in single doses — preliminary clinical studies at nine collaborating institutions. Igaku to Yakugaku 9: 1913–1920, 1983c
Nials AT, Butchers PR, Coleman RA, Johnson M, Vardey CJ. Salmeterol and formoterol: are they both long-acting β2-adrenoceptor agonists? Abstract. British Journal of Pharmacology 99 (Suppl.): 120P, 1990
Nishikori K, Maeno H. Characterization of β-adrenergic receptor and adenylate cyclase in canine cerebellum. Archives of Biochemistry and Biophysics 195: 505–517, 1979
Nishimura Y, Hosoe A, Yamamoto T, Ohtani T, Hirata S, et al. Clinical study on formoterol (BD 40A) in pediatric patients with bronchitis. Igaku to Yakugaku 7: 1677–1685, 1982
Nix A, Nichol GM, Robson A, Barnes PJ, Chung KF. Effect of formoterol, a long-lasting β2-adrenoceptor agonist, against methacholine-induced bronchoconstriction. British Journal of Clinical Pharmacology 29: 321–324, 1990
Nolte D, Rolke M. Comparative trial of the acute bronchospasmolytic action of formoterol versus fenoterol over 12 hours. In Sybrecht G (Ed.) Formoterol — a new long-acting bronchodilator, pp. 24–31, Hans Huber Publishers, Toronto, 1987
Nowak D, Reuss G, Bonnet R, Jörres R, Magnussen H. Inhaled formoterol has a longer bronchodilator effect and a later maximum protective effect than fenoterol. In Sybrecht G (Ed.) Formoterol — a new long-acting bronchodilator, p. 60, Hans Huber Publishers, Toronto, 1987
Ohta Y, Goto I. Clinical experience with formoterol (BD 40A) in chronic bronchitis. Igaku to Yakugaku 7: 1205–1209, 1982
Osterman K, Grettve L, Lundbäck B, Piituilainen E, Stålenheim G, et al. Inhaled formoterol twice daily in the treatment of asthma. A three months multicentre study of efficacy and tolerability. Abstract P76. Clinical and Experimental Allergy 20 (Suppl. 1): 34, 1990
Palmqvist M, Balder B, Löwhagen O, Melander B, Svedmyr N, et al. Late asthmatic reaction prevented by inhaled salbutamol and formoterol. Abstract. Journal of Allergy and Clinical Immunology 83: 244, 1989
Palmqvist M, Löwhagen O, Svedmyr N. One year double-blind treatment with inhaled formoterol in nocturnal asthma. Abstract. American Review of Respiratory Disease 141: A210, 1990
Petro W, Kaspar P, Haiti W. Einfluss von Formoterol auf die Schlagfrequenz menschlicher Cilien. Bronchospasmolyse mit Beta-2-Sympathikomimetika. Erfahrungen mit d.langwirkenden Dosier-Aerosol Formoterol. 21. Kolloquium d.Bad Reichenhaller Forschungsanst.d.Atmungsorgane, 17 Juni 1988. Hrsg.v. D. Nolte und A. Burghele, Muenchen-Deisenhofen, Dustri, pp. 131–139, 1989
Price AH, Clissold SP. Salbutamol in the 1980s: a reappraisal of its clinical efficacy. Drugs 38: 77–122, 1989
Rabe K, Dent G, Giembycz M, Ravenall S, Barnes PJ, et al. Effects of long acting beta adrenoceptor agonist formoterol vs albuterol on LTB4 induced Ca2+ mobilisation, H2O2 and TxB2 release in guinea pig eosinophils. Abstract. American Review of Respiratory Disease 141: A29, 1990
Ramsdale EH, Otis J, Kline PA, Gantovnick LA, Hargreave FE, et al. Prolonged protective effect against methacholine-induced bronchoconstriction by the inhaled β2-agonist formoterol. American Review of Respiratory Disease 143: 998–1001, 1991
Reeds PJ. Guidelines for the management of asthma in adults. British Medical Journal 301: 771–772, 1990
Rees J. β2 Agonists and asthma. Still the mainstay of symptomatic treatment. British Medical Journal 302: 1166–1167, 1991
Rosenhall L, Sandström T, Wallin A. Formoterol, a long acting inhaled β2-agonist, twice daily for 1 year in asthmatic patients. Abstract. American Review of Respiratory Disease 141: A210, 1990
Rudolf M, Malins DR. Trial of a new long-acting inhaled beta-2 agonist, formoterol fumarate. Abstract. American Review of Respiratory Disease 141: A210, 1990
Sasaki H, Kamimura H, Shiobarà Y. Absorption and distribution of formoterol fumarate in rats. Oyo Butsuri 25: 981–991, 1983
Sasaki H, Kamimura H, Shiobara Y, Esumi Y, Takaichi M, et al. Disposition and metabolism of formoterol fumarate, a new bronchodilator, in rats and dogs. Xenobiotica 12: 803–812, 1982
Schnabel D, Sybrecht G, Hemmerling T. Protective efficacy of formoterol compared with theophylline in patients with nocturnal bronchial asthma. In Davies RJ (Ed.) Formoterol in asthma — clinical profile of a new long-acting inhaled β2-agonist, pp. 23–28, Hogrefe & Huber Publishers, Toronto, 1988
Schultze-Werninghaus G. Multicenter 1-year trial on formoterol, a new long-acting β2-agonist, in chronic obstructive airway disease. Lung (Suppl.): 83–89, 1990
Sears MR, Taylor DR, Print CG, Lake DC, Li Q, et al. Regular inhaled beta-agonist treatment in bronchial asthma. Lancet 336: 1391–1396, 1990
Seki T, Miyazaki M. Effect of bronchodilator BD 40A on healthy subjects. Igaku to Yakugaku 9: 1815–1824, 1983
Seyama K, Takagi J, Tomot I, Hirohata M, Hirao M, et al. Results of clinical use of BD 40A dry syrup for pediatric bronchial-asthma. Shonika Shinryo 45; 958–962, 1982
Shima K, Takenaka S. The clinical evaluation of formoterol (BD 40A) in bronchial asthma with long term administration and in chronic obstructive pulmonary disease with short term administration. Yakuri to Chiryo 11: 3935–3942, 1983
Shioda H, Akasaka T, Wagatsuma Y, Nemoto N, Yamaguchi T, et al. Clinical results of repeated administration of formoterol (BD 40A) dry syrup in the pediatric patients with bronchial asthma. Shonika Shinryo 45: 1883–1890, 1982a
Shioda H, Nakayama Y, Akasaka T, Suzuki I, Arai Y, et al. Efficacy of formoterol (BD 40A) with single dose for bronchial asthma in children: clinical results in 8 institutions. Shonika Rinsho 35: 403–411, 1982
Stålenheim G, Grettve L, Lundbåck B, Osterman K, Pittulainen E, et al. Treatment with the long acting β2 agonist formoterol for three months. Efficacy and tolerability. Journal of Clinical Immunology 85 (Suppl.): 219, 1990
Stauder J, Burghele A. A double-blind study assessing the efficacy and tolerability of formoterol in comparison with fenoterol. In Gerhard Sybrecht (Ed.) Formoterol — a new long-acting bronchodilator, pp. 54–56, Hans Huber Publishers, Toronto, Lewiston, NY, Bern, Stuttgart, 1988
Subramanian N. Inhibition of immunological and non-immunological histamine release from human basophils and lung mast cells by formoterol. Arzneimittel-Forschung 36: 502–505, 1986
Suetsugu S. The effects of long-term administration of formoterol (BD 40A) in patients with bronchial asthma. Kiso to Rinsho 17: 2945–2950, 1983
Sykes AP, Ayres JG. A study of the duration of the bronchodilator effect of 12μg and 24μg of inhaled formoterol and 200 g inhaled salbutamol in asthma. Respiratory Medicine 84: 135–138, 1990
Takeda M, Takagi T. Effects of a new bronchodilator, (αRS)-3-formamido-4-hydroxy-α-[[[(αRS)-p-methoxy-α-methylphenethyl] amino] methyl] benzyl alcohol fumarate dihydrate (BD 40A) on gastric acid secretion and gastrointestinal motility. Abstract. Folia Pharmacologica Japonica 76: 185–191, 1980
Tasaka T. Formoterol (Atock®): a new orally active and selective β2-receptor stimulant. Drugs of Today 22: 505–519, 1986
Tokuyama K, Lötvall JO, Löfdahl C-G, Barnes PJ, Chung KF. Inhaled formoterol inhibits histamine-induced airflow obstruction and airway microvascular leakage. European Journal of Pharmacology 193: 35–39, 1991
Tomioka K, Yamada T, Ida H. Anti-allergic activities of the β-adrenoceptor stimulant formoterol (BD 40A). Archives Internationales de Pharmacodynamie et de Thérapie 250: 279–292, 1981
Tomioka K, Yamada T, Tachikawa S. Effects of formoterol (BD 40A), a β-adrenoceptor stimulant, on isolated guinea-pig lung parenchymal strips and antigen-induced SRS-A release in rats. Archives Internationales de Pharmacodynamie et de Thérapie 267: 91–102, 1984
Tomita Y, Joh K. Clinical effect of formoterol dry syrup on allergic cough. Kiso to Rinsho 17: 3003–3008, 1983
Tsoy AN, Cheltzov VV, Zaseyeva OV, Chumburidze VB, Chervinskaya GA. Formoterol and terbutaline aerosols in nocturnal asthma. Abstract. European Respiratory Journal 3 (Suppl. 10): 418s–419s, 1990a
Tsoy AN, Cheltzov VV, Zaseyeva OV, Shilinsh V, Yashino LA. Preventive effect of formoterol aerosol in exercise-induced bronchoconstriction. European Respiratory Journal 3 (Suppl. 10): 235s, 1990b
Uchida Y. Clinical experiences with formoterol (BD40A), a new bronchodilator against bronchitis. Igaku to Yakugaku 7: 829–835, 1982
von Berg A, Berdel D. Comparison of formoterol, a new β2-agonist, with salbutamol in the treatment of bronchospasm in asthmatic children. In Sybrecht G (Ed.) Formoterol — a new long-acting bronchodilator, pp. 38–47, Hans Huber Publishers, Toronto, 1987
von Berg A, Berdel D. Formoterol and salbutamol metered aerosols: comparison of a new and an established beta-2-agonist for their bronchodilating efficacy in the treatment of childhood bronchial asthma. Pediatric Pulmonology 7: 89–93, 1989
von Berg A, Berdel D. Efficacy of formoterol metered aerosol in children. Lung (Suppl.): 90–98, 1990
Walker SR, Evans ME, Renards AJ, Paterson JW. The clinical pharmacology of oral and inhaled salbutamol. Clinical Pharmacology and Therapeutics 13: 861–867, 1972
Wallin A, Melander B, Rosenhall L, Sandström T, Wåhlander L. Formoterol, a new long acting beta2 agonist for inhalation twice daily, compared with salbutamol in the treatment of asthma. Thorax 45: 259–261, 1990
Watanabe K. Clinical evidence for several new β2-stimulating bronchodilators. Doai Igaku Zasslei 12: 93–106, 1982
Webster T, Plit M. Comparison of inhaled formoterol 12/Ltg (a new generation, long acting β2-agonist) and inhaled salbutamol (200μg) in exercise induced asthmatic (EIA) adults. Abstract. European Respiratory Journal 3 (Suppl. 10): 235S, 1990
Wegener T, Hedenström H, Boman G. Effect of formoterol on gas distribution and diffusing capacity in asthma. Abstract. Journal of Allergy and Clinical Immunology 85 (Suppl.): 147, 1990
Wèttengel R. Comparison of the bronchodilator effect of formoterol and fenoterol after inhalation. In Sybrecht G (Ed.) Formoterol — a new long-acting bronchodilator, p. 61, Hans Huber Publishers, Toronto, 1987
Wettengel R. Vergleichende Utersuchungen der bronchospas-molytischen Wirkung und Vertraglichkeit von Formoterol and Fenoterol als Dosier-Aerosol. Bronchospasmolyse mit Beta-2-Sympathikomimetika. Erfahrungen mit d.langwirkenden Dosier-Aerosol Formoterol. 21. Kolloquium d.Bad Reichenhaller Forschungsanstd.Atmungsorgane, 17 Juni 1988. Hrsg.v. D. Nolte und A. Burghele, Muenchen-Deisenhofen, Dustri, pp. 69–82, 1989
Wong CS, Pavord ID, Williams J, Britton JR, Tattersfield AE. Bronchodilator, cardiovascular, and hypokalaemic effects of fenoterol, salbutamol, and terbutaline in asthma. Lancet 336: 1396–1399, 1990
Wönne R, Hofmann D. Bronchodilating duration of action of inhaled formoterol compared to fenoterol in children with bronchial asthma. Abstract. European Respiratory Journal 1 (Suppl. 2): 194S, 1988
Wylicil P, Krause P, Burghele A. Formoterol versus berodual in patients with obstructive airways diseases. Abstract. European Respiratory Journal 3 (Suppl. 10): 236, 1990
Yamakido M, Inamizu T, Ikuta T, Onari K, Tanabe M, et al. Evaluation of a new bronchodilator, formoterol, using biochemical parameters. International Journal of Clinical Pharmacology, Therapy and Toxicology 23: 461–468, 1985
Yokoi K, Murase K, Shiobara Y. The development of a radioim-munoassay for formoterol. Life Sciences 33: 1665–1672, 1983
Yoshida T, Saito T, Suzuki H, Miki H, Okamiya H, et al. Acute, subacute and chronic toxicity studies of a bronchodilator, formotemol fumarate (BD 40A). Pharmacometrics 26: 811–829, 1983
Zanotti E, Crotti P, Moscato G, Dellabianca A, Rampulla C. Effect of inhaled formoterol on ‘fog’-induced bronchospasm: a double-blind, three-period cross-over study. Abstract. European Respiratory Journal 3 (Suppl. 10): 133S–134S, 1990
Author information
Authors and Affiliations
Additional information
Various sections of the manuscript reviewed by: P.J. Barnes, National Heart and Lung Institute, London, England; A.B. Becker, Section of Allergy and Clinical Immunology, Health Services Centre, Winnipeg, Manitoba, Canada; R.C. Bone, Department of Medicine, Rush Medical College, Chicago, Illinois, USA; G.K. Crompton, Respiratory Medicine Service, Respiratory Unit, Northern General Hospital, Edinburgh, Scotland; N.J. Gross, Hines, Illinois, USA; S. Kesten, Asthma Centre, Toronto Hospital, Toronto, Ontario, Canada; F.P.V. Maesen, De Wever Ziekenhuis, Heerlen, The Netherlands; C.P. Page, Biomedical Sciences Division, Kings College London, University of London, London, England; R. Pauwels, Department of Respiratory Disease, University Hospital, Ghent, Belgium; A.S. Rebuck, Asthma Centre, Toronto Hospital, Toronto, Ontario, Canada; A.E. Tattersfield, Faculty of Medicine, Respiratory Medicine Unit, City Hospital, Nottingham, England; J.H. Toogood, Allergy Clinic, Victoria Hospital, London, Ontario, Canada; M. Yamakido, Department of Internal Medicine, Hiroshima University School of Medicine, Hiroshima, Japan.
Rights and permissions
About this article
Cite this article
Faulds, D., Hollingshead, L.M. & Goa, K.L. Formoterol. Drugs 42, 115–137 (1991). https://doi.org/10.2165/00003495-199142010-00007
Published:
Issue Date:
DOI: https://doi.org/10.2165/00003495-199142010-00007