Abstract
Background: The oral direct thrombin inhibitor ximelagatran is a new class of anticoagulant currently in clinical development for the prevention and treatment of thromboembolic disease. After oral administration, ximelagatran is rapidly absorbed and bioconverted to its active form melagatran.
Objective: To investigate the influence of mild-to-moderate hepatic impairment on the pharmacokinetic and pharmacodynamic properties of ximelagatran.
Study design: Nonblinded, nonrandomised study.
Participants: Twelve volunteers with mild-to-moderate hepatic impairment (classified as Child-Pugh A or B) and 12 age-, weight-, and sex-matched control volunteers with normal hepatic function.
Methods: Volunteers received a single oral dose of ximelagatran 24mg. Plasma and urine samples were collected for pharmacokinetic and pharmacodynamic analyses.
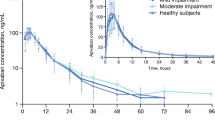
Results: The absorption and bioconversion of ximelagatran to melagatran were rapid in both groups. The maximum plasma concentration of melagatran (Cmax) was achieved 2–3 hours after administration; the mean elimination half-life (t½z) was 3.6 hours for hepatically impaired volunteers and 3.1 hours for the control volunteers. The area under the plasma concentration-time curve (AUC) and Cmax of melagatran in volunteers with hepatic impairment were 11 and 25% lower than in control volunteers, respectively. However, after correcting for the higher renal function (i.e. higher calculated creatinine clearance) in the hepatically impaired volunteers, the ratio of melagatran AUC for hepatically impaired/control volunteers was 0.98 (90% CI 0.80, 1.22), suggesting that mild-to-moderate hepatic impairment had no influence on the pharmacokinetics of ximelagatran. Melagatran was the predominant compound in urine, accounting for 13–14% of the ximelagatran dose. Renal clearance of melagatran was 13% higher in hepatically impaired than in control volunteers. There were no significant differences between the two groups in the concentration-response relationship between plasma melagatran concentration and activated partial thromboplastin time (APTT). Baseline prothrombin time (PT) was slightly longer in the hepatically impaired patients than in the control volunteers, probably reflecting a slight decrease in the activity of coagulation factors. However, when concentrations of melagatran were at their peak, the increase in PT from baseline values was the same in both groups. Capillary bleeding time was measured in the hepatically impaired patients only, and was not increased by ximelagatran. Ximelagatran was well tolerated in both groups.
Conclusion: There were no differences in the pharmacokinetic or pharmacodynamic properties of melagatran following oral administration of ximelagatran between the hepatically impaired and control volunteers. These findings suggest that dose adjustment for patients with mild-to-moderate impairment of hepatic function is not necessary.







Similar content being viewed by others
Notes
Use of tradenames is for product identification only and does not imply endorsement.
References
Gustafsson D, Nyström J-E, Carlsson S, et al. The direct thrombin inhibitor melagatran and its oral prodrug H 376/95: intestinal absorption properties, biochemical and pharmacodynamic effects. Thromb Res 2001; 101: 171–81
Eriksson UG, Bredberg U, Hoffmann K-J, et al. Absorption, distribution, metabolism, and excretion of ximelagatran, an oral direct thrombin inhibitor, in rats, dogs, and humans. Drug Metab Dispos 2003; 31: 294–305
Formation of melagatran from H 415/04 in human intestine, kidney and lung microsomes. Mölndal: DMPK and Bioanalytical Chemistry, AstraZeneca, 2000. (Data on file)
Eriksson UG, Bredberg U, Gislén K, et al. Pharmacokinetics and pharmacodynamics of ximelagatran, a novel oral direct thrombin inhibitor, in young, healthy male subjects. Eur J Clin Pharmacol 2003; 59: 35–43
Sarich TC, Eriksson UG, Mattsson C, et al. Inhibition of thrombin generation by the oral direct thrombin inhibitor ximelagatran in shed blood from healthy male subjects. Thromb Haemost 2002; 87: 300–5
Johansson LC, Andersson M, Fager G, et al. No influence of ethnic origin on the pharmacokinetics and pharmacodynamics of melagatran, following oral administration of ximelagatran, a novel, oral direct thrombin inhibitor, to healthy male volunteers. Clin Pharmacokinet 2003; 42: 475–84
Johansson LC, Frison L, Logren U, et al. Influence of age on the pharmacokinetics and pharmacodynamics of ximelagatran, an oral direct thrombin inhibitor. Clin Pharmacokinet 2003; 42: 381–92
Elg M, Gustafsson D, Deinum J. The importance of enzyme inhibition kinetics for the effect of thrombin inhibitors in a rat model of arterial thrombosis. Thromb Haemost 1997; 78: 1268–92
Elg M, Gustafsson D, Carlsson S. Antithrombotic effects and bleeding time of thrombin inhibitors and warfarin in the rat. Thromb Res 1999; 94: 187–97
Eriksson BI, Bergqvist D, Kälebo P, et al. Ximelagatran and melagatran compared with dalteparin for prevention of venous thromboembolism after total hip or knee replacement: the METHRO II randomised trial. Lancet 2002; 360: 1441–7
Eriksson BI, Agnelli G, Cohen AT, et al. Direct thrombin inhibitor melagatran followed by oral ximelagatran in comparison with enoxaparin for prevention of venous thromboembolism after total hip or knee replacement: The METHRO III Study. Thromb Haemost 2003; 89: 288–96
Eriksson BI, Arfwidsson A-C, Frison L, et al. A dose-ranging study of the oral direct thrombin inhibitor, ximelagatran, and its subcutaneous form, melagatran, compared with dalteparin in the prophylaxis of thromboembolism after hip or knee replacement: METHRO I. MElagatran for THRombin inhibition in Orthopaedic surgery. Thromb Haemost 2002; 87: 231–7
Francis CW, Davidson BL, Berkowitz SD, et al. Ximelagatran versus warfarin for the prevention of venous thromboembolism after total knee arthroplasty: a randomized, double-blind trial. Ann Intern Med 2002; 137: 648–55
Heit JA, Colwell CW, Francis CW, et al. Comparison of the oral direct thrombin inhibitor ximelagatran with enoxaparin as prophylaxis against venous thromboembolism after total knee replacement: a phase 2 dose-finding study. Arch Intern Med 2001; 161: 2215–21
Eriksson H, Eriksson UG, Frison L, et al. Pharmacokinetics and pharmacodynamics of melagatran, a novel synthetic LMW thrombin inhibitor, in patients with acute DVT. Thromb Haemost 1999; 81: 358–63
Eriksson H, Wåhlander K, Gustafsson D, et al. A randomized, controlled, dose-guiding study of the oral direct thrombin inhibitor ximelagatran compared with standard therapy for the treatment of acute deep vein thrombosis: THRIVE I. J Thromb Haemost 2003; 1: 41–7
Wåhlander K, Lapidus L, Olsson CG, et al. Pharmacokinetics, pharmacodynamics and clinical effects of the oral direct thrombin inhibitor ximelagatran in acute treatment of patients with pulmonary embolism and deep vein thrombosis. Thromb Res 2002; 107: 93–9
Petersen P, Grind M, Adler A, SPORTIF II Investigators. Ximelagatran versus warfarin for stroke prevention in patients with nonvalvular atrial fibrillation. SPORTIF II: a dose-guiding, tolerability and safety study. J Am Coll Cardiol 2003; 41: 1445–51
Westphal JF, Brogard JM. Drug administration in chronic liver disease. Drug Saf 1997; 17: 47–73
McLean AJ, Morgan DJ. Clinical pharmacokinetics in patients with liver disease. Clin Pharmacokinet 1991; 21: 42–69
Williams RL, Mamelok RD. Hepatic disease and drug pharmacokinetics. Clin Pharmacokinet 1980; 5: 528–47
Verbeeck RK, Horsmans Y. Effect of hepatic insufficiency on pharmacokinetics and drug dosing. Pharm World Sci 1998; 20: 183–92
Morgan DJ, McLean AJ. Clinical pharmacokinetic and pharmacodynamic considerations in patients with liver disease: an update. Clin Pharmacokinet 1995; 29: 370–91
Sonne J. Drug metabolism in liver disease: implications for therapeutic drug monitoring. Ther Drug Monit 1996; 18: 397–401
Pugh RN, Murray-Lyon IM, Dawson JL, et al. Transection of the oesophagus for bleeding oesophageal varices. Br J Surg 1973; 60: 646–9
Caesar J, Shaldon S, Chianussi L, et al. The use of indocyanine green in the measurement of hepatic blood flow and as a test of hepatic function. Clin Sci 1961; 21: 43–57
Miotti T, Bircher J, Preisig R. The 30-minute aminopyrine breath test: optimization of sampling times and intravenous administration of 14C-aminopyrine. Digestion 1988; 39: 241–50
Larsson M, Ahnoff M, Abrahamsson A, et al. Determination of ximelagatran, an oral direct thrombin inhibitor, its active metabolite melagatran, and the intermediate metabolites, in biological samples by liquid chromatography-mass spectrometry. J Chromatogr B Analyt Technol Biomed Life Sci 2003; 783: 335–47
Cockcroft DW, Gault MH. Prediction of creatinine clearance from serum creatinine. Nephron 1976; 16: 31–41
Eriksson UG, Mandema JW, Karlsson MO, et al. Pharmacokinetics of melagatran and the effect on ex vivo coagulation time in orthopaedic surgery patients receiving subcutaneous melagatran and oral ximelagatran: a population model analysis. Clin Pharmacokinet. 2003; 42: 687–701
Deitcher SR. Interpretation of the international normalised ratio in patients with liver disease. Lancet 2002; 359: 47–8
Mattsson C, Menschiek-Lundin A, Wåhlander K, et al. Effect of melagatran on prothrombin time assays depends on the sensitivity of the thromboplastin and the final dilution of the plasma sample. Thromb Haemost 2001; 86: 611–5
Acknowledgements
This study was sponsored by AstraZeneca. The authors acknowledge the valuable contributions of Drs Gabriela Popescu, Karl-Heitz Molz, Angelika Weil, and study nurses Andrea Couturier and Alexandra Köbrich at APEX GmbH, Germany, to the planning and performance of this study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wåhlander, K., Eriksson-Lepkowska, M., Frison, L. et al. No Influence of Mild-To-Moderate Hepatic Impairment on the Pharmacokinetics and Pharmacodynamics of Ximelagatran, an Oral Direct Thrombin Inhibitor. Clin Pharmacokinet 42, 755–764 (2003). https://doi.org/10.2165/00003088-200342080-00004
Published:
Issue Date:
DOI: https://doi.org/10.2165/00003088-200342080-00004