Abstract
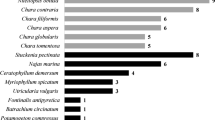
We compared benthic community structure among stands of Typha angustifolia (narrow-leaf cattail) and herbicide-treated (Glypro) and untreated Phragmites australis (common reed) over two summers in a Lake Erie coastal wetland (i.e., drowned river mouth). Both macrophytes are invasives, but only Phragmites is currently controlled by herbicides because of its reputed “undesirable” effects on wetland community structure and function. Macroinvertebrate diversity was similar among stand types and relatively high (Shannon-Weaver indices ∼2.6–4.2), probably because of high system primary productivity and a mix of lentic and riverine species. Proportions of macroinvertebrate functional feeding groups were also similar, but Jaccard’s similarity indices were relatively low (29%–57%), suggesting macroinvertebrate compositional differences among stand types. Coleopterans particularly affected species presence/absence patterns, but their presence was associated with low water level rather than hydrophyte type per se. Moreover, total macroinvertebrate densities were greater in both Phragmites treatments than in Typha; a pattern generated mostly by gastropods (≥ 95% Gyraulus deflectus and Physella gyrina) and chironomids. Microalgal food supply likely plays a part in explaining these density differences, given diatom-dominated epiphyton was denser on submerged shoots of Phragmites than on Typha. Common diatom assemblages were similar among stand types, but species richness was significantly greater on untreated -Phragmites than on herbicide-treated, early senescent Phragmites and untreated-Typha. However, advanced senescence from herbicide application (∼3 months) did not apparently affect macroscale habitat suitability and structure above and below the waterline, given counts of ovipositing odonates (mostly Ischnura and Enallagma) and captures of juvenile fishes (> 90% Lepomis spp.) were similar among stand types. Overall, our results suggest that benthic community structure is comparable between similarly-aged stands (∼4 yrs old) of invading reed and cattail and is not directly or indirectly affected by Glypro application.
Similar content being viewed by others
Literature Cited
Able, K. W. and S. M. Hagan. 2000. Effects of common reed (Phragmites australis) invasion on marsh surface macrofauna: response of fishes and decapod crustaceans. Estuaries 23: 633–46.
Able, K. W. and S. M. Hagan. 2003. Impact of common reed, Phragmites australis, on essential fish habitat: influence on reproduction, embryological development, and larval abundance of mummichog (Fundulus heteroclitus). Estuaries 26: 40–50.
Amsberry, L., M. A. Baker, P. J. Ewanchuk, and M. D. Bertness. 2000. Clonal integration and the expansion of Phragmites australis. Ecological Applications 10: 1110–18.
Angradi, T. R., S. M. Hagan, and K. W. Able. 2001. Vegetation type and the intertidal macroinvertebrate fauna of a brackish marsh: Phragmites vs. Spartina. Wetlands 21: 75–92.
Armstrong, J., F. Afreen-Zobayed, and W. Armstrong. 1996. Phragmites die-back: sulphide- and acetic acid-induced bud and root death, lignifications, and blockages with aeration and vascular systems. New Phytologist 134: 601–14.
Benoit, L. K. and R. A. Askins. 1999. Impact of the spread of Phragmites on the distribution of birds in Connecticut tidal marshes. Wetlands 19: 194–208.
Besitka, M. A. R. 1996. An ecological and historical study of Phragmites australis along the Atlantic Coast. M.S. Thesis. Drexel University, Philadelphia, PA, USA.
Brigham, A. R., W. U. Brigham, and A. Gnilka. 1982. Aquatic Insects and Oligochaetes of North and South Carolina. Midwest Aquatic Enterprises, Mahomet, IL, USA.
Chambers, R. M., L. A. Meyerson, and K. Saltonstall. 1999. Expansion of Phragmites australis into tidal wetlands of North America. Aquatic Botany 64: 261–73.
Chen, C. Y., K. M. Hathaway, and C. L. Folt. 2004. Multiple stress effects of Vision herbicide, pH, and food on Zooplankton and larval amphibian species from forest wetlands. Environmental Toxicology and Chemistry 23: 823–31.
Cronk, J. K. and W. J. Mitsch. 1994. Periphyton productivity on artificial and natural surfaces in constructed freshwater wetlands under different hydrologic regimes. Aquatic Botany 48: 325–41.
Detenbeck, N. E., R. Hermanutz, K. Allen, and M. C. Swift. 1996. Fate and effects of the herbicide atrazine in flow-through wetland mesocosms. Environmental Toxicology and Chemistry 15: 937–46.
Fell, P. E., R. S. Warren, J. K. Light, R. L. Rawson, Jr., and S. M. Fairley. 2003. Comparison of fish and macroinvertebrate use of Typha angustifolia, Phragmites australis, and treated Phragmites marshes along the lower Connecticut River. Estuaries 26: 534–51.
Fell, P. E., S. P. Weissbach, D. A. Jones, M. A. Fallon, J. A. Zeppieri, E. K. Faison, K. A. Lennon, K. J. Newberry, and L. K. Reddington. 1998. Does invasion of oligohaline tidal marshes by reed grass, Phragmites australis (Cav.) Trin. ex. Steud., affect the availability of prey resources for the mummichog, Fundulus heteroclitus L.? Journal of Experimental Marine Biology and Ecology 222: 59–77.
Galatowitsch, S. M., N. O. Anderson, and P. D. Ascher. 1999. Invasiveness in wetland plants in temperate North America. Wetlands 19: 733–55.
Gallardo, M. T., J. R. Ascher, M. J. Collier, B. B. Martin, and D. F. Martin. 1999. Effect of cattail (Typha domingensis) extracts, leachates, and selected phenolic compounds on rates of oxygen production by Salvinia (Salvinia minima). Journal of Aquatic Plant Management 37: 80–82.
Gallardo, M. T., B. B. Martin, and D. F. Martin. 1998. Inhibition of water fern Salvinia minima by cattail (Typha domingensis) extracts and by 2-chlorophenol and salicylaldehyde. Journal of Chemical Ecology 24: 1483–90.
Gessner, M. O. 2000. Breakdown and nutrient dynamics of submerged Phragmites shoots in the littoral zone of a temperate hardwater lake. Aquatic Botany 66: 9–20.
Giesy, J. P., S. Dobson, and K. R. Solomon. 2000. Ecotoxicological risk assessment for Roundup herbicide. Reviews of Environmental Contamination and Toxicology 167: 35–120.
Goldsborough, L. G. and G. G. C. Robinson. 1983. The effect of two triazine herbicides on the productivity of freshwater marsh periphyton. Aquatic Toxicology 4: 95–112.
Goldsborough, L. G. and G. G. C. Robinson. 1986. Changes in periphytic algal community structure as a consequence of short herbicide exposures. Hydrobiologia 139: 177–92.
Graca, M. A. S., C. Cressa, M. O. Gessner, M. J. Feio, K. A. Callies, and C. Barrios. 2001. Food quality, feeding preferences, survival and growth of shredders from temperate and tropical streams. Freshwater Biology 46: 947–57.
Gratton, C. and R. F. Denno. 2005. Restoration of arthropod assemblages in a Spartina salt marsh following removal of the invasive plant Phragmites australis. Restoration Ecology 13: 358–72.
Hann, B. J. 1991. Invertebrate grazer-periphyton interactions in a eutrophic marsh pond. Freshwater Biology 26: 87–96.
Hanson, S. R., D. T. Osgood, and D. J. Yozzo. 2002. Nekton use of a Phragmites australis marsh on the Hudson River, New York, USA. Wetlands 22: 326–37.
Harvill, M. L. 2000. Aquatic insects of Old Woman Creek Estuary. Lake Erie Protection Fund Report. Toledo, OH, USA.
Hellings, S. E. and J. L. Gallagher. 1992. The effects of salinity and flooding on Phragmites australis. Journal of Applied Ecology 29: 41–49.
Herdendorf, C. E. 1990. Great Lakes estuaries. Estuaries 13: 493–503.
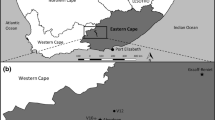
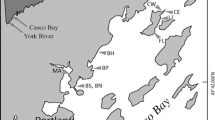
Herdendorf, C. E., D. M. Klarer, and R. C. Herdendorf. 2004. The ecology of Old Woman Creek, Ohio: an estuarine and watershed profile. Ohio Department of Natural Resources, Division of Natural Areas and Preserves, Columbus, OH, USA.
Kashian, D. R. and T. M. Burton. 2000. A comparison of macroinvertebrates of two Great Lakes coastal wetlands: testing potential metrics for an index of ecological integrity. Journal of Great Lakes Research 26: 460–81.
Klarer, D. M. and D. F. Millie. 1992. Aquatic macrophytes and algae at Old Woman Creek Estuary and other Great Lakes coastal wetlands. Journal of Great Lakes Research 18: 622–33.
Kneib, R. T. 1997. The role of tidal marshes in the ecology of estuarine nekton. Oceanography and Marine Biology Annual Review 35: 163–220.
Krammer, K. and H. Lange-Bertalot. 1986. Bacillariophyceae. 1. Teil: Naviculaceae. p. 1–876. In H. Ettl, J. Gerloff, H. Heynig, and D. Mollenhauer (eds.) Süsswasser flora von Mitteleuropa, Band 2/1. Gustav Fischer Verlag: Stuttgart, New York.
Krammer, K. and H. Lange-Bertalot. 1988. Bacillariophyceae. 2. Teil: Bacillariaceae, Epithemiaceae, Surirellaceae. p. 1–610. In H. Ettl, J. Gerloff, H. Heynig, and D. Mollenhauer (eds.) Süsswasserflora von Mitteleuropa, Band 2/3. VEB Gustav forthe Verlag: Stuttgart, Jena.
Krammer, K. and H. Lange-Bertalot. 1991b. Bacillariophyceae. 3. Teil: Centrales, Fragilariaceae, Eunotiaceae. p. 1–598. In H. Ettl, J. Gerloff, H. Heynig, and D. Mollenhauer (eds.) Süsswasserflora von Mitteleuropa, Band 2/3. Gustav Fischer Verlag: Stuttgart, Jena.
Krammer, K. and H. Lange-Bertalot. 1991b. Bacillariophyceae. 4. Teil: Achnanthaceae, Kritische Ergänzungen zu Navicula (Lineolatae) und Gomphonema, Gesamtliteraturverzeichnis Teil 1–4. p. 1–437. In H. Ettl, G. Gärtner, J. Gerloff, H. Heynig, and D. Mollenhauer (eds.) Süsswasserflora von Mitteleuropa, Band 2/4. Gustav Fischer Verlag: Stuttgart, Jena.
Krieger, K. A. and D. M. Klarer. 1992. Macroinvertebrate communities of the Old Woman Creek State Nature Preserve and National Estuarine Research Reserve. Old Woman Creek National Estuarine Research Reserve Technical Report No. 9.
Krieger, K. A. and D. M. Klarer. 1995. Spatial and seasonal distributions of nonplanktonic aquatic invertebrates in the Old Woman Creek National Estuarine Research Reserve. NOAA Technical Memorandum, Washington, DC, USA.
Kulesza, A. E. and J. R. Holomuzki. 2006. Amphipod performance responses to decaying leaf litter of Phragmites aus trails and Typha angustifolia from a Lake Erie coastal marsh. Wetlands 26: 1079–88.
Květ, J. and D. F. Westlake. 1998. Primary production in wetlands. p. 78–268. In D. F. Westlake, J. Květ, and A. Szczepański (eds.) The Production Ecology of Wetlands. Cambridge University Press, Cambridge, UK.
Lake, J. C. and M. R. Leishman. 2004. Invasion success of exotic plants in natural ecosystems: the role of disturbance, plant attributes and freedom from herbivores. Biological Conservation 117: 215–26.
Lynch, E. A. and K. Saltonstall. 2002. Paleocological and genetic analyses provide evidence for recent colonization of native Phragmites australis populations in a Lake Superior wetland. Wetlands 22: 637–46.
Marks, M., B. Lapin, and J. Randall. 1994. Phragmites australis (P. communis): threats, management, and monitoring. Natural Areas Journal 14: 285–94.
Mason, C. F. and R. J. Bryant. 1975. Production, nutrient content and decomposition of Phragmites communis Trin. and Typha latifolia L. Journal of Ecology 63: 71–95.
McClary, M., Jr. 2004. Spartina alterniflora and Phragmites australis as habitat for the ribbed mussel, Geukensia demissa, in Saw Mill Creek of New Jersey’s Hackensack Meadowlands. Urban Habitats 2: 83–90.
Merritt, R. W. and K. W. Cummins. 1996. An Introduction to the Aquatic Insects of North America, third edition. Kendall/Hunt Publishing, Dubuque, IA, USA.
Meyerson, L. A., K. Saltonstall, L. Windham, E. Kiviat, and S. Findlay. 2000. A comparison of Phragmites australis in freshwater and brackish marsh environments in North America. Wetlands Ecology and Management 8: 89–103.
Mueller-Dombois, D. and H. Ellenberg. 1974. Aims and Methods of Vegetation Ecology. John Wiley & Sons, Inc., New York, NY, USA.
Oertli, B. and J.-B. Lachavanne. 1995. The effects of shoot age on colonization of an emergent macrophyte (Typha latifolia) by macroinvertebrates. Freshwater Biology 34: 421–31.
Orson, R. A., R. S. Warren, and W. A. Niering. 1987. Development of a tidal marsh in a New England river valley. Estuaries 10: 20–27.
Pearson, J. A. and M. J. Leoschke. 1992. Floristic composition and conservation status of fens in Iowa. Journal of Iowa Academy of Science 99: 41–52.
Peckarsky, B. L., P. R. Fraissinet, M. A. Penton, and D. J. Conklin, Jr. 1990. Freshwater Macroinvertebrates of Northeastern North America. Cornell University Press, Ithaca, NY, USA.
Pennak, R. W. 1989. Freshwater Invertebrates of the United States: Protozoa to Mollusca, third edition. John Wiley & Sons, Inc., New York, NY, USA.
Polunin, N. V. C. 1982. Processes contributing to the decay of reed (Phragmites australis) litter in fresh water. Archiv für Hydrobiologie 94: 182–209.
Raichel, D. L., K. W. Able, and J. M. Hartman. 2003. The influence of Phragmites (Common Reed) on the distribution, abundance, and potential prey of a resident marsh fish in the Hackensack Meadowlands, New Jersey. Estuaries 26: 511–21.
Reed, P. B. 1988. National list of plant species that occur in wetlands: Northeast (Region 1). United States Fish and Wildlife Service, Washington, DC, USA. Biological Report 88(26.1).
Relyea, R. A. 2005. The impact of insecticides and herbicides on the biodiversity and productivity of aquatic communities. Ecological Applications 15: 618–27.
Robertson, T. L. and J. S. Weiss. 2005. A comparison of epifaunal communities associated with the stems of salt marsh grasses Phragmites australis and Spartina alterniflora. Wetlands 25: 1–7.
Rooth, J. E., J. C. Stevenson, and J. C. Cornwell. 2003. Increased sediment accretion rates following invasion by Phragmites australis: the role of litter. Estuaries 26: 475–83.
Saltonstall, K. 2002. Cryptic invasion by a non-native genotype of the common reed, Phragmites australis, into North America. Proceedings of the National Academy of Sciences of the USA 99: 2445–49.
Shannon, C. E. and W. Weaver. 1963. The Mathematical Theory of Communication. University of Illinois Press, Urbana, IL, USA.
Sokal, R. R. and F. J. Rohlf. 1995. Biometry, third edition. W. H. Freeman and Company, New York, NY, USA.
Stevenson, R. J. 1996. The stimulation and drag of current. p. 321–40. In R. J. Stevenson, M. L. Bothwell, and R. L. Lowe (eds.) Algal Ecology: Freshwater Benthic Ecosystems. Academic Press, San Diego, CA, USA.
Talley, T. S. and L. A. Levin. 2001. Modification of sediments and macrofauna by an invasive marsh plant. Biological Invasions 3: 51–68.
Templer, P., S. Findlay, and C. Wigand. 1998. Sediment chemistry associated with native and non-native emergent macrophytes of a Hudson River marsh ecosystem. Wetlands 18: 70–78.
Thoma, R. F. 1997. Fish community based assessment of Ohio’s Lake Erie near shore area. Ohio Journal of Science 97: 21.
Trautman, M. B. 1981. The Fishes of Ohio with Illustrated Keys, revised edition. Ohio State University Press in Collaboration with the Ohio Sea Grant Program Center for Lake Erie Area Research, Columbus, OH, USA.
Trexel-Kroll, D. 2002. Succession of floating-leaf to emergent plant communities following reduced water levels in Old Woman Creek Estuary. M.S. Thesis. Miami University, Oxford, OH, USA.
Turner, A. M. and J. C. Trexler. 1997. Sampling aquatic invertebrates from marshes: evaluating the options. Journal of the North American Benthological Society 16: 694–709.
USDA (U.S. Department of Agriculture). 2000. Glyphosate: herbicide information profile. U.S. Forest Service, Pacific Northwest Region, USA.
Varga, I. 2003. Structure and changes of macroinvertebrate community colonizing decomposing rhizome litter of common reed at Lake Ferto/Neusiedler See (Hungary). Hydrobiologia 506-509: 413–20.
Wallwork, J. A. 1976. The Distribution and Diversity of Soil Fauna. Academic Press, London, UK.
Warren, R. S., P. E. Fell, J. L. Grimsby, E. L. Buck, G. C. Rilling, and R. A. Fertik. 2001. Rates, patterns, and impacts of Phragmites australis expansion and effects of experimental Phragmites control on vegetation, macroinvertebrates, and fish within tidelands of the lower Connecticut River. Estuaries 24: 90–107.
Wehr, J. D. and R. G. Sheath. 2003. Freshwater Algae of North America: Ecology and Classification. Academic Press, New York, NY, USA.
Westfall, M. J. and M. L. May. 2006. Damselflies of North America, revised edition. Scientific Publishers, Gainesville, FL, USA.
Whyte, R. S. 1996. The vegetation dynamics of a freshwater estuary on Lake Erie: the Old Woman Creek State Nature Preserve and National Estuarine Research Reserve, Huron, Ohio. Ph.D. Dissertation. Miami University, Oxford, OH, USA.
Whyte, R. S., D. A. Francko, and D. M. Klarer. 1997. Distribution of the floating-leaf macrophyte Nelumbo lutea (American water lotus) in a coastal wetland on Lake Erie. Wetlands 17: 567–73.
Whyte, R. S., D. A. Francko, and D. M. Klarer. 2003. The aquatic vegetation of the Old Woman Creek National Estuarine Research Reserve (Huron, Ohio): a Lake Eriecoastal wetland. The Michigan Botanist 42: 63–84.
Wilcox, K. L., S. A. Petrie, L. A. Maynard, and S. W. Meyers. 2003. Historical distribution and abundance of Phragmites australis at Long Point, Lake Erie, Ontario. Journal of Great Lakes Research 29: 664–80.
Wilkinson, L. 2000. SYSTAT 9. SPSS, Chicago, IL, USA.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kulesza, A.E., Holomuzki, J.R. & Klarer, D.M. Benthic community structure in stands of Typha angustifolia and herbicide-treated and untreated Phragmites australis . Wetlands 28, 40–56 (2008). https://doi.org/10.1672/07-63.1
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1672/07-63.1