Abstract
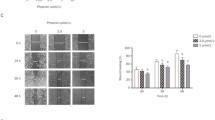
Verticillin A is a diketopiperazine compound which was previously isolated from Amanita flavorubescens Alk (containing parasitic fungi Hypomyces hyalines (Schw.) Tul.). Here, we initially found, by wound healing assay and Transwell assay in vitro, that verticillin A possesses an inhibitory effect against the migration and invasion of the human colon cancer cell. Subsequently, c-mesenchymal-epithelial transition factor (c-Met) was identified as a molecular target of verticillin A by screening key genes related to cell migration. Verticillin A-mediated c-Met suppression is at the transcriptional level. Further study demonstrated that verticillin A suppressed c-MET phosphorylation and decreased c-MET protein level. In addition, verticillin A inhibited the phosphorylation of c-MET downstream molecules including rat sarcoma (Ras)-associated factor (Raf), extracellular signal-regulated kinase (ERK), and protein kinase B (AKT). Overexpression of Erk partially reversed the verticillin A-mediated anti-metastasis action in the human colon cancer cell. More importantly, verticillin A also inhibited cancer cell metastasis in vivo. Thus, verticillin A can significantly inhibit the migration and invasion of colon cancer cells by targeting c-Met and inhibiting Ras/Raf/mitogen-activated extracellular signal-regulated kinase (MEK)/ERK signaling pathways. Therefore, we determined that verticillin A is a natural compound that can be further developed as an anti-metastatic drug in human cancers.
摘要
目的
结肠癌是全球第三大常见癌症,转移是结肠癌患 者死亡的主要原因,因此迫切需要寻找能够抑制 结肠癌转移新方法。在前期研究中,我们证明了 轮枝孢菌素A 在各种类型的癌症中均具有强大 的抗肿瘤增殖作用。在本研究中,我们试图确定 轮枝孢菌素A 对结肠癌细胞的抗转移作用,并阐 明其潜在的分子机制.
创新点
首次发现轮枝孢菌素A 可以有效地抑制结肠癌细 胞在体内外的迁移和侵袭。在分子水平上,沉默 c-Met 显著降低了结肠细胞的侵袭,而轮枝孢菌 素A 正是有效的c-Met 抑制剂; 同时,轮枝孢菌 素A 还可以通过削弱c-MET 磷酸化来抑制 c-MET 下游Ras/Raf/MEK/ERK 信号通路,从而 达到抗肿瘤转移效果.
方法
采用划痕分析和Transwell 法来证明轮枝孢菌素A 对结肠癌细胞体外迁移和侵袭的影响; 采用蛋白 质印迹法来确定c-MET 及其下游分子的表达水 平和活性; 采用实时定量聚合酶链式反应 (qRT-PCR)分析以评估c-Met 的mRNA 表达水 平; 采用RNA 干扰测定用于沉默细胞中c-Met 的表达; 采用质粒瞬时转染以过度表达Erk.
结论
轮枝孢菌素A 显著抑制结肠癌细胞的迁移和侵 袭,抑制c-Met 转录表达,削弱c-MET 磷酸化, 从而抑制其下游Ras/Raf/MEK/ERK 信号通路, 这是轮枝孢菌素A 抗结肠癌细胞转移的主要分 子机制。根据本研究结果,我们确定天然产物轮 枝孢菌素A 具有进一步开发抗癌症转移的巨大 潜力.
Similar content being viewed by others
Change history
16 April 2022
An Erratum to this paper has been published: https://doi.org/10.1631/jzus.B20e0190
References
Arnold L, Enders J, Thomas SM, 2017. Activated HGF-c-Met axis in head and neck cancer. Cancers, 9(12):169. https://doi.org/10.3390/cancers9120169
Birchmeier C, Birchmeier W, Gherardi E, et al., 2003. Met, metastasis, motility and more. Nat Rev Mol Cell Biol, 4(12):915–925. https://doi.org/10.1038/nrm1261
Budchart P, Khamwut A, Sinthuvanich C, et al., 2017. Partially purified Gloriosa superba peptides inhibit colon cancer cell viability by inducing apoptosis through p53 upregulation. Am J Med Sci, 354(4):423–429. https://doi.org/10.1016/j.amjms.2017.06.005
Chaffer CL, Weinberg RA, 2011. A perspective on cancer cell metastasis. Science, 331(6024):1559–1564. https://doi.org/10.1126/science.1203543
Chiche A, Di-Cicco A, Sesma-Sanz L, et al., 2019. p53 controls the plasticity of mammary luminal progenitor cells downstream of Met signaling. Breast Cancer Res, 21:13. https://doi.org/10.1186/s13058-019-1101-8
Christiano AP, Yoshida BA, Dubauskas Z, et al., 2000. Development of markers of prostate cancer metastasis: review and perspective. Urol Oncol, 5(5):217–223. https://doi.org/10.1016/S1078-1439(00)00070-3
Cohen SA, Yu M, Baker K, et al., 2017. The CpG island methylator phenotype is concordant between primary colorectal carcinoma and matched distant metastases. Clin Epigenetics, 9:46. https://doi.org/10.1186/s13148-017-0347-1
D’Amico L, Belisario D, Migliardi G, et al., 2016. c-MET inhibition blocks bone metastasis development induced by renal cancer stem cells. Oncotarget, 7(29):45525–45537. https://doi.org/10.18632/oncotarget.9997
Du PC, Liang HB, Fu XW, et al., 2019. SLC25A22 promotes proliferation and metastasis by activating MAPK/ERK pathway in gallbladder cancer. Cancer Cell Int, 19:33. https://doi.org/10.1186/s12935-019-0746-9
Engstrom PF, 2012. Ten years of progress in colon cancer therapy. J Natl Compr Canc Netw, 10(5):574–576. https://doi.org/10.6004/jnccn.2012.0058
Fan JX, Zheng DW, Rong L, et al., 2017. Targeting epithelial-mesenchymal transition: metal organic network nanocomplexes for preventing tumor metastasis. Biomaterials, 139:116–126. https://doi.org/10.1016/j.biomaterials.2017.06.007
Fodde R, Smits R, Clevers H, 2001. APC, signal transduction and genetic instability in colorectal cancer. Nat Rev Cancer, 1:55–67. https://doi.org/10.1038/35094067
Gardner FP, Serie DJ, Salomao DR, et al., 2014. c-MET expression in primary and liver metastases in uveal melanoma. Melanoma Res, 24(6):617–620. https://doi.org/10.1097/CMR.0000000000000118
Gholamin S, Fiuji H, Maftouh M, et al., 2014. Targeting c-MET/HGF signaling pathway in upper gastrointestinal cancers: rationale and progress. Curr Drug Targets, 15(14):1302–1311. https://doi.org/10.2174/1389450115666141107105456
Giordano S, di Renzo MF, Olivero M, et al., 1992. The c-met/HGF receptor in human tumours. Eur J Cancer Prev, 1(6):45–50. https://doi.org/10.1097/00008469-199210003-00007
Glodde N, Bald T, van den Boorn-Konijnenberg D, et al., 2017. Reactive neutrophil responses dependent on the receptor tyrosine kinase c-MET limit cancer immunotherapy. Immunity, 47(4):789–802.e9. https://doi.org/10.1016/j.immuni.2017.09.012
Huang KH, Sung IC, Fang WL, et al., 2018. Correlation between HGF/c-Met and Notch1 signaling pathways in human gastric cancer cells. Oncol Rep, 40(1):294–302. https://doi.org/10.3892/or.2018.6447
Hui AY, Meens JA, Schick C, et al., 2009. Src and FAK mediate cell-matrix adhesion-dependent activation of Met during transformation of breast epithelial cells. J Cell Biochem, 107(6):1168–1181. https://doi.org/10.1002/jcb.22219
Jia YT, Dai GY, Wang JX, et al., 2016. c-MET inhibition enhances the response of the colorectal cancer cells to irradiation in vitro and in vivo. Oncol Lett, 11(4):2879–2885. https://doi.org/10.3892/ol.2016.4303
Kim HS, Chon HJ, Kim H, et al., 2018. MET in gastric cancer with liver metastasis: the relationship between MET amplification and Met overexpression in primary stomach tumors and liver metastasis. J Surg Oncol, 117(8):1679–1686. https://doi.org/10.1002/jso.25097
Kiselev VY, Juvin V, Malek M, et al., 2015. Perturbations of PIP3 signalling trigger a global remodelling of mRNA landscape and reveal a transcriptional feedback loop. Nucleic Acids Res, 43(20):9663–9679. https://doi.org/10.1093/nar/gkv1015
Kou LF, Yao Q, Sivaprakasam S, et al., 2017. Dual targeting of L-carnitine-conjugated nanoparticles to OCTN2 and ATB0,+ to deliver chemotherapeutic agents for colon cancer therapy. Drug Deliv, 24(1):1338–1349. https://doi.org/10.1080/10717544.2017.1377316
Kurtzeborn K, Kwon HN, Kuure S, 2019. MAPK/ERK signaling in regulation of renal differentiation. Int J Mol Sci, 20(7):1779. https://doi.org/10.3390/ijms20071779
Lee SJ, Lee J, Park SH, et al., 2018. c-MET overexpression in colorectal cancer: a poor prognostic factor for survival. Clin Colorectal Cancer, 17(3):165–169. https://doi.org/10.1016/j.clcc.2018.02.013
Lino-Silva LS, Guzmán-López JC, Zepeda-Najar C, et al., 2019. Overall survival of patients with colon cancer and a prolonged time to surgery. J Surg Oncol, 119(4):503–509. https://doi.org/10.1002/jso.25354
Liu FY, Liu QQ, Yang DF, et al., 2011. Verticillin A overcomes apoptosis resistance in human colon carcinoma through DNA methylation-dependent upregulation of BNIP3. Cancer Res, 71(21):6807–6816. https://doi.org/10.1158/0008-5472.CAN-11-1575
Lu CW, Yang DF, Sabbatini ME, et al., 2018. Contrasting roles of H3K4me3 and H3K9me3 in regulation of apoptosis and gemcitabine resistance in human pancreatic cancer cells. BMC Cancer, 18:149. https://doi.org/10.1186/s12885-018-4061-y
Mao D, Feng L, Gong H, 2018. The antitumor and immunomodulatory effect of Yanghe decoction in breast cancer is related to the modulation of the JAK/STAT signaling pathway. Evid Based Complement Alternat Med, 2018:8460526. https://doi.org/10.1155/2018/8460526
Mihailidou C, Karamouzis MV, Schizas D, et al., 2017. Cotargeting c-Met and DNA double-strand breaks (DSBs): therapeutic strategies in BRCA-mutated gastric carcinomas. Biochimie, 142:135–143. https://doi.org/10.1016/j.biochi.2017.09.001
Mo HN, Liu P, 2017. Targeting MET in cancer therapy. Chronic Dis Transl Med, 3(3):148–153. https://doi.org/10.1016/j.cdtm.2017.06.002
Oh BY, Park YA, Huh JW, et al., 2018. Prognostic impact of tumor-budding grade in stages 1–3 colon cancer: a retrospective cohort study. Ann Surg Oncol, 25(1):204–211. https://doi.org/10.1245/s10434-017-6135-5
Organ SL, Tsao MS, 2011. An overview of the c-MET signaling pathway. Ther Adv Med Oncol, 3(1_Suppl):S7–S19. https://doi.org/10.1177/1758834011422556
Pan Y, Cheung ST, Tong JHM, et al., 2018. Granulin epithelin precursor promotes colorectal carcinogenesis by activating MARK/ERK pathway. J Transl Med, 16:150. https://doi.org/10.1186/s12967-018-1530-7
Parizadeh SM, Jafarzadeh-Esfehani R, Fazilat-Panah D, et al., 2019. The potential therapeutic and prognostic impacts of the c-MET/HGF signaling pathway in colorectal cancer. IUBMB Life, 71(7):802–811. https://doi.org/10.1002/iub.2063
Paschall AV, Yang DF, Lu CW, et al., 2015. H3K9 trimethylation silences Fas expression to confer colon carcinoma immune escape and 5-fluorouracil chemoresistance. J Immunol, 195(4):1868–1882. https://doi.org/10.4049/jimmunol.1402243
Pereira SS, Monteiro MP, Costa MM, et al., 2019. MAPK/ERK pathway inhibition is a promising treatment target for adrenocortical tumors. J Cell Biochem, 120(1):894–906. https://doi.org/10.1002/jcb.27451
Printz C, 2017. Some stage III colon cancer patients may need only half of the standard chemotherapy course. Cancer, 123(20):3871. https://doi.org/10.1002/cncr.31042
Ren Y, Cao B, Law S, et al., 2005. Hepatocyte growth factor promotes cancer cell migration and angiogenic factors expression: a prognostic marker of human esophageal squamous cell carcinomas. Clin Cancer Res, 11(17):6190–6197. https://doi.org/10.1158/1078-0432.CCR-04-2553
Saigusa S, Toiyama Y, Tanaka K, et al., 2012. Inhibition of HGF/cMET expression prevents distant recurrence of rectal cancer after preoperative chemoradiotherapy. Int J Oncol, 40(2):583–591. https://doi.org/10.3892/ijo.2011.1200
Sipos F, Galamb O, 2012. Epithelial-to-mesenchymal and mesenchymal-to-epithelial transitions in the colon. World J Gastroenterol, 18(7):601–608. https://doi.org/10.3748/wjg.v18.i7.601
Thayaparan T, Spicer JF, Maher J, 2016. The role of the HGF/Met axis in mesothelioma. Biochem Soc Trans, 44(2):363–370. https://doi.org/10.1042/BST20150252
Wei DY, Geng F, Liang SM, et al., 2017. CAF-derived HGF promotes cell proliferation and drug resistance by upregulating the c-Met/PI3K/Akt and GRP78 signalling in ovarian cancer cells. Biosci Rep, 37(2):BSR20160470. https://doi.org/10.1042/BSR20160470
Xiang C, Chen JX, Fu PF, 2017. HGF/Met signaling in cancer invasion: the impact on cytoskeleton remodeling. Cancers, 9(5):44. https://doi.org/10.3390/cancers9050044
Zewdu A, Lopez G, Braggio D, et al., 2016. Verticillin A inhibits leiomyosarcoma and malignant peripheral nerve sheath tumor growth via induction of apoptosis. Clin Exp Pharmacol, 6(6):221. https://doi.org/10.4172/2161-1459.1000221
Zhang QW, Ye ZD, Shi L, 2019. c-Met kinase inhibitors: an update patent review (2014–2017). Expert Opin Ther Pat, 29(1):25–41. https://doi.org/10.1080/13543776.2019.1552261
Author information
Authors and Affiliations
Contributions
Fei-yan LIU designed study and analyzed data; Fei-yan LIU and Jing-xin LU wrote the manuscript; Qian-qian LIU and Xue-li ZENG performed experiments; Yue-lin GUAN performed plasmid transient transfection; Kai TU provided in vivo study. All authors have read and approved the final version of the manuscript, and, therefore, have full access to all the data in the study and take responsibility for the integrity and security of the data.
Corresponding author
Additional information
Compliance with ethics guidelines
Qian-qian LIU, Xue-li ZENG, Yue-lin GUAN, Jing-xin LU, Kai TU, and Fei-yan LIU declare that they have no conflict of interest.
All institutional and national guidelines for the care and use of laboratory animals were followed.
Rights and permissions
About this article
Cite this article
Liu, Qq., Zeng, Xl., Guan, Yl. et al. Verticillin A inhibits colon cancer cell migration and invasion by targeting c-Met. J. Zhejiang Univ. Sci. B 21, 779–795 (2020). https://doi.org/10.1631/jzus.B2000190
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1631/jzus.B2000190