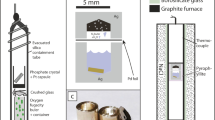
Abstract
Exceptionally low jg 18O values of primary uraninite and pitchblende (i.e. -32 per mil to -19.5 per mil) from Proterozoic unconformity-type uranium deposits in Saskatchewan, Canada, in conjunction with theoretical uraninite-water oxygen isotope fractionation factors suggest that primary uranium mineralization is not in oxygen isotopie equilibrium with clays and silicates. The low jg 18O values have been interpreted to have resulted from the recrystallization of primary uranium mineralization in the presence of modern meteoric fluids having low jg 18O values of ca. -18 per mil. The absence of apparent alteration in many of the uraninite and pitchblende samples requires that the uranium minerals exchange oxygen isotopes with fluids, with only minor disturbances to their original chemical compositions and textures. However, experiments on the interaction between water and natural uraninites, from these deposits, and detailed electron micro-probe analyses of natural uraninite and pitchblende indicate that, in the presence of water, old uraninite rapidly alters to curite (Pb2U5O174H2O). The hydration of uraninite to curite releases uranium and calcium into solution and becquerelite (Ca(UO2)6O4(OH)6H2O) is precipitated. In the presence of Si-saturated waters, uranium silicate minerals, soddyite ((UO2)2(SiO4)2H2O) and kasolite (Pb(UO2)SiO4H2O are precipitated in addition to, curite and schoepite ((UO2)8O2(OH)12(H2O)12). The mineral paragenesis observed in these experiments is similar to sequences observed in oxidized zones in uranium deposits and UO2-water experiments. Therefore, it is unlikely that natural uraninite and pitchblende can simply exchange oxygen with an oxidizing fluid without a concomitant change in phase chemistry or structure, nor will oxidation of uraninite lead to the formation of U3O7. as predicted by theoretical calculations used in natural analogue studies for the disposal of high level nuclear waste.
Similar content being viewed by others
References
R.J. Finch and R.C. Ewing, J. Nucl. Mater., 190, p. 133–156 (1992).
J. Janeczek, R.C. Ewing, V.M. Oversby and L.O. Werme, J. Nucl. Mater., in press (1996).
K. Hattori and S. Halas, Geochim. Cosmochim. Acta, 46, p. 1863–1868 (1982).
H.R. Hoekstra and J.J. Katz, U.S. Geological Survey Professional Paper 300, p. 543–547 (1956).
K. Hattori, K. Muehlenbachs, and D. Morton, Geol. Soc. Amer.-Geol. Soc. Can., Pgm. w Abst, 10, A417 (1978).
T.G. Kotzer and T.K. Kyser, Am. Mineral., 78, p. 1262–1274 (1993).
M. Fayek and T.K. Kyser, accepted, Can. Min (1996).
Y. Zheng, Geochim. Cosmochim. Acta., 55, p. 2299–2307 (1991).
M.R. Wilson and T.K. Kyser, Econ. Geol., 82, p. 1450–1557 (1987).
T.G. Kotzer and T.K. Kyser, Sask. Energy and Mines, Sask. Geol. Surv., Misc. Rep. 90-4, p. 153–157 (1990).
T.G. Kotzer and T.K. Kyser, Chern. Geol., 120, p. 45–89 (1995).
J. Janeczek, R.C. Ewing and L.E. Thomas, J. Nucl. Mater., 207, p. 177–191 (1993).
J.J. Cramer and J.A.T. Smellie, AECL Rep. 10851 (1994).
J.J. Cramer, AECL Rep. 11204 (1995).
J.A.T. Smellie and F. Karlsson, SKB Tech. Rep. TR 96-08, (1996).
J.F.W. Bowles, Chem. Geol., 83, p. 47–53 (1990).
D.J. Wronkiewicz, J.K. Bates, T.J. Gerding, E. Veleckis and B.S. Tani, J. Nucl. Mater., 190, p. 107–127 (1992).
D.E. Grandstaff, Econ. Geol., 71, p. 1493–1506 (1976).
F. Gronvold, J. Inorg. Nucl. Chem., 1, p. 357–370 (1955).
R.M. Berman, Journal of the Mineralogical Society of America, 42, p. 705–731 (1957).
L. Powers and M. Stauffer, Can. J. Earth Sci., 25, p 1945–1954 (1985).
J. Janeczek and R.C. Ewing, R.C., J. Nucl. Mater., 190, p. 128–132 (1992).
J.R. O'Neil, Stable Isotopes in High Temperature Geological Processes, eds. J. W. Valley, H.P. Taylor, J.R. O'Neil (Reviews in Mineralogy, 16, Mineral. Soc. of Amer, 1986), p. 1–37.
S.W. Kieffer, Rev. Geophys. Space Phys., 30-4, p. 827–849 (1982).
R.H. Becker and R.N. Clayton, Geochim. Cosmochim. Acta, 40, p. 1151–1165 (1976).
Acknowledgement
The Natural Sciences and Engineering Research Council of Canada (NSERC) supported this work via operating and equipment grants to T.K. Kyser and a Post-Doctoral Fellowship to M. Fayek. This work was also partially supported by the Basic Energy Science DOE grant DE-FG03-95ER14540. The Authors also wish to thank Drs. Audunr Ludviksson, Leo Archer and Vlad Sopuck for their help in collecting the data for this manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Fayek, M., Kyser, T.K., Ewing, R.C. et al. Uraninite-Water Interactions in an Oxidizing Environment. MRS Online Proceedings Library 465, 1201–1208 (1996). https://doi.org/10.1557/PROC-465-1201
Published:
Issue Date:
DOI: https://doi.org/10.1557/PROC-465-1201