Abstract
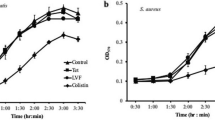
The antimicrobial activity of anthraquinone emodin isolated from Aspergillus awamori WAIR120 (LC032125) culture was investigated against some clinical, phytopathogenic and foodborne pathogenic microorganisms using an agar diffusion method. Among bacterial and fungal strains tested, the highest activity was obtained against Enterococcus faecalis AHR7 as well as Aspergillus niger OC20 with minimal inhibitory emodin concentration of 125 and 85 μg/mL, respectively. Emodin was found to induce morphogenic effects including swelling and elongation of bacterial cell and conidiation decrease, pigmentation loss, and cytoplasmic retraction of fungal cell, as was shown by light microscopy. Additionally, cellular effects were also resulted, in which emodin caused considerable changes in the nature of cell membrane and submicroscopic structure of bacterial and fungal cell, as was shown by transmission electron microscopy. Furthermore, there was an evidence of a disruption of lipid metabolism of fungal cell. These findings thus indicate the future possibility of exploiting emodin as an effective inhibitor of clinical, phytopathogenic and foodborne pathogenic microorganisms.
Similar content being viewed by others
Abbreviations
- HPLC:
-
high performance liquid chromatography
- MIC:
-
minimal inhibitory concentration
- TEM:
-
transmission electron microscopy
- TLC:
-
thin layer chromatography
References
Abd El-Aal M.A. 2012. Opportunistic fungi associated with ocular infections in Sharkia district. Master Thesis, Zagazig University, Zagazig, Egypt.
Agosti G., Birkinshaw J.H. & Chaplen P. 1962. Studies in the biochemistry of micro-organisms. 112. Anthraquinone pigments of strains of Cladosporium fulvum Cooke. Biochem. J. 85: 528–530
Al-Nuri M., Za’tar N.A., Abu-Eid M.A., Hannoun M.A., Al Jondi W.J., Hussein A.I. & Ali-Shtayeh M.S. 1996. Emodin, a naturally occuring anthraquinone: its isolation and spectrophotometric determination in Rumex cyprius Plant. Spectrosc. Lett. 29: 1539–1543
Altschul S.F., Madden T.L., Schäffer A.A., Zhang J., Zhang Z., Miller W. & Lipman D.J. 1997. Gapped BLAST and PSIBLAST: a new generation of protein database search programs. Nucleic Acids Res. 25: 3389–3402
Anke H., Kolthoum I. & Laatsch H. 1980. Metabolic products of microorganisms. 192. The anthraquinones of the Aspergillus glaucus group. II. Biological activity. Arch. Microbiol. 126: 231–236
Auffray Y., Boutibonnes P. & Lemarinier S. 1984. Filamentous forms of Bacillus thuringiensis (Berliner) formed in the presence of genotoxic mycotoxins. Microbiol. Aliments Nutr. 2: 59–67
Barnard D.L., Huffman J.H., Morris J.L., Wood S.G., Hughes B.G. & Sidwell R.W. 1992. Evaluation of the antiviral activity of anthraquinones, anthrones and anthraquinone derivatives against human cytomegalovirus. Antiviral Res. 17: 63–77
Basu S., Ghosh A. & Hazra B. 2005. Evaluation of the antibacterial activity of Ventilago madraspatana Gaertn., Rubia cordifolia Linn. and Lantana camara Linn.: isolation of emodin and physcion as active antibacterial agents. Phytother. Res. 19: 888–894
Boik J.C. 1995. Cancer and Natural Medicine: A Textbook of Basic Science and Clinical Research. Oregon Medical Press, Princeton, MN, USA.
Carmo E.S., de Oliveira Lima E., de Souza E.L. & de Sousa F.B. 2008. Effect of Cinnamomum zeylancium blume essential oil on the growth and morphogenesis of some potentially pathogenic Aspergillus species. Braz. J. Microbiol. 39: 91–97
Chang C.H., Lin C.C., Yang J.J., Namba T. & Hattori M. 1996. Antiinflammatory effects of emodin from Ventilago leiocarpa. Am. J. Chin. Med. 24: 139–142
Chang M., Wang J., Tian F., Zhang Q. & Ye B. 2010. Antibacterial activity of secondary metabolites from Aspergillus awamori F12 isolated from rhizospheric soil of Rhizophora stylosa Griff. Acta Microbiol. Sin. 50: 1385–1391
Cheng Y.W. & Kang J.J. 1998. Emodin-induced muscle contraction of mouse diaphragm and the involvement of Ca2+ influx and Ca2+ release from sarcoplasmic reticulum. Br. J. Pharmacol. 123: 815–820
Chukwujekwu J.C., Coombes P.H., Mulholland D.A. & van Staden J. 2006. Emodin, an antibacterial anthraquinone from the roots of Cassia occidentalis. S. Afr. J. Bot. 72: 295–297
de Barros I.B., de Souza-Daniel J.F., Pinto J.P., Rezendo M.I., Filho R.B. & Ferreira D.T. 2011. Phytochemical and antifungal activity of anthraquinones and root and leaf extracts of Coccoloba mollis on phytopathogens. Braz. Arch. Biol. Technol. 54: 535–541
Donnelly D.M.X. & Sheridan M.H. 1986. Anthraquinones from Trichoderma polysporum. Phytochem. 25: 2303–2304
Ezzat S.M., El-Sayed E.A., Abou El-Hawa M.I. & Ismaiel A.A. 2007. Morphological and ultrastructural studies for the biological action of penicillic acid on some bacterial species. Res. J. Microbiol. 2: 303–314
Ghosh A.C., Manmade A. & Demain A.L. 1977. Toxins from Penicillium islandicum Sopp, pp. 625–638. In: Rodricks J.V., Hesseltine C.W. & Mehlman M.A. (eds) Mycotoxins in Human and Animal Health. Pathotox, Chicago.
Ismaiel A.A., Ali A.E.S. & Enan G. 2014. Incidence of Listeria in Egyptian meat and dairy samples. Food Sci. Biotechnol. 23: 179–185
Ismaiel A.A., Bassyouni R.H., Kamel Z. & Gabr S.M. 2016. Detoxification of patulin by kombucha tea culture. CyTA J. Food 14: 271–279
Ismaiel A.A. & Papenbrock J. 2014. The effects of patulin from Penicillium vulpinum on seedling growth, root tip ultrastructure and glutathione content of maize. Eur. J. Plant Pathol. 139: 497–509
Ismaiel A.A., Rabie G.H., Kenawey S.E.M. & Abd El-Aal M.A. 2012. Efficacy of aqueous garlic extract on growth, aflatoxin B1 production, and cyto-morphological aberrations of Aspergillus flavus, causing human ophthalmic infection: topical treatment of A. flavus keratitis. Braz. J. Microbiol. 43: 1355–1364
Izhaki I. 2002. Emodin — a secondary metabolite with multiple ecological functions in higher plants. New Phytol. 155: 205–217
Kögl F. & Postowsky J.J. 1925. Untersuchungen über Pilzfarb stoffe. II. Über die Farbstoffe des blutroten Hautkorpfes (Dermocybe sanquinea Wulf.). Justus Liebigs Ann. Chem. 444: 1–7
Le Van T. 1984. Emodin a fungal metabolite and the effects of emodin on the growth of some soil microorganisms. Acta Agr. Silv. Ser. Agraria 23: 235–242
Lu C., Wang H., Lv W., Xu P., Zhu J., Xie J., Liu B. & Lou Z. 2011. Antibacterial properties of anthraquinones extracted from rhubarb against Aeromonas hydrophila. Fish Sci. 77: 375–384
Lu Y.Y., Zhang J.L. & Qian J.M. 2008. The effect of emodin on VEGF receptors in human colon cancer cells. Cancer Biother. Radiopharm. 23: 222–228
Masuda T. & Ueno Y. 1984. Microsomal transformation of emodin into a direct mutagen. Mutat. Res. 125: 135–144
Moubasher A.H. 1993. Soil Fungi in Qatar and Other Arab Countries. The Centre for Scientific and Applied Research, Doha, Qatar.
Mueller S.O., Schmitt M., Dekant W., Stopper H., Schlatter J., Schreier P. & Lutz W.K. 1999. Occurrence of emodin, chrysophanol and physcion in vegetables, herbs and liquors. Genotoxicity and anti-genotoxicity of the anthraquinones and of the whole plants. Food Chem. Toxicol. 37: 481–491
Natori S., Sato F. & Udagawa S. 1965. Anthraquinone metabolites of Talaromyces avellanens (Thom et Turreson), C.R. Benjamin and Preussia multispora (Saito et Minoura) Cain. Chem. Pharm. Bull. 13: 385–389
Panichayupakaranant P., Sakunpak A. & Sakunphueak A. 2009. Quantitative HPLC determination and extraction of anthraquinones in Senna alata leaves. J. Chromatogr. Sci. 47: 197–200
Peng J., Song Z. & Ma C. 2008. Emodin studies on pharmacokinetics and distribution in rat liver after Polygonum cuspidatum Sieb. et Zucc. extract administration. World Sci. Technol. 10: 64–67
Shia C.S., Hou Y.C., Tsai S.Y., Huieh P.H., Leu Y.L. & Chao P.D. 2010. Differences in pharmacokinetics and ex vivo antioxidant activity following intravenous and oral administrations of emodin to rats. J. Pharm. Sci. 99: 2185–2195
Shibata S., Shoji J., Ohta A. & Watanable M. 1957. Metabolic products of fungi. XI. Some observations on the occurrence of skyrin and rugulosin in mold metabolites with reference to structural relationships between penicilliopsin and skyrin. Chem. Pharm. Bull. 5: 380–383
Shibata S. & Udagawa S. 1963. Metabolic products of fungi. XIX. Isolation of rugulosin from Penicillium brunneum Udagawa. Chem. Pharm. Bull. 11: 402–403
Shieh D.E., Chen Y.Y., Yen M.H., Chiand L.C. & Lin C.C. 2004. Emodin-induced apoptosis through p53-dependent pathway in human hepatoma cells. Life Sci. 74: 2279–2290
Singh U.P., Singh K.P., Singh S.P., Ram S.N. & Pandey V.B. 1992. Effect of emodin isolated from Rhamnus triquetra on spore germination of some fungi. Fitopatol. Bras. 17: 420–422
Turner W.B. & Aldridge D.C. 1983. Fungal Metabolites II. London, Academic Press.
Ubbink-Kok T., Anderson J.A. & Konings W.N. 1986. Inhibition of electron transfer and uncoupling effects by emodin and emodinanthrone in Escherichia coli. Antimicrob. Agents Chemother. 30: 147–151
Wang C., Zhang D., Ma H. & Liu J. 2007. Neuroprotective effects of emodin-8-O-beta-D-glucoside in vivo and in vitro. Eur. J. Pharmacol. 577: 58–63
Wang H.H. 1993. Antitrichomonal action of emodin in mice. J. Ethnopharmacol. 40: 111–116
Wang H.H. & Chung J.G. 1997. Emodin-induced inhibition of growth and DNA damage in the Helicobacter pylori. Curr. Microbiol. 35: 262–266
Wang W., Zhou Q., Liu L. & Zou K. 2012. Anti-allergic activity of emodin on IgE-mediated activation in RBL-2H3 cells. Pharmacol. Rep. 64: 1216–1222
Wells J.M., Cole R.J. & Kirksey J.W. 1975. Emodin, a toxic metabolite of Aspergillus wentii isolated from weevil-damaged chestnuts. Appl. Microbiol. 30: 26–28
Xue J., Ding W. & Liu Y. 2010. Anti-diabetic effects of emodin involved in the activation of PPARgamma on high-fat dietfed and low dose of streptozotocin-induced diabetic mice. Fitoterapia 81: 173–177
Yamazaki M., Maebayashi Y. & Miyaki K. 1971. The isolation of secalonic acid A from Aspergillus ochraceus cultured on rice. Chem. Pharm. Bull. 19: 199–201
Zhang L., Lau Y.K., Xi L., Hong R.L., Kim D.S., Chen C.F., Hortobagyi G.N., Chang C. & Hung M.C. 1998. Tyrosine kinase inhibitors, emodin and its derivative repress HER-2/neu-induced cellular transformation and metastasis-associated properties. Oncogene 16: 2855–2863
Acknowledgements
The authors thank Dr. Basma Hamdy Amin, TEM Unit, Regional Center for Mycology and Biotechnology, Al-Azhar University, Cairo, Egypt, for excellent technical assistance during the processing steps of TEM. The work employed herein was supported in part by the Department of Botany and Microbiology, Faculty of Science, Zagazig University, Egypt.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ismaiel, A.A., Rabie, G.H. & Abd El-Aal, M.A. Antimicrobial and morphogenic effects of emodin produced by Aspergillus awamori WAIR120. Biologia 71, 464–474 (2016). https://doi.org/10.1515/biolog-2016-0067
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1515/biolog-2016-0067