Abstract
Background: Susceptibility to schizophrenia is determined by interactions between genes and environment, possibly via epigenetic mechanisms. Schizophrenia has been associated with a restrictive epigenome, and histone deacetylase (HDAC) inhibitors have been postulated as coadjuvant agents to potentiate the efficacy of current antipsychotic drugs. We aimed to evaluate global histone posttranslational modifications (HPTMs) and HDAC expression and activity in the dorsolateral prefrontal cortex (DLPFC) of individuals with schizophrenia.
Methods: We used postmortem DLPFC samples of individuals with schizophrenia and controls matched for sex, age, and postmortem interval. Schizophrenia samples were classified into antipsychotic-treated or antipsychotic-free subgroups according to blood toxicology. Expression of HPTMs and HDAC was quantified by Western blot. HDAC activity was measured with a fluorometric assay.
Results: H3K9ac, H3K27ac, and H3K4me3 were globally enhanced in the DLPFC of individuals with schizophrenia (+24%–42%, p < 0.05). HDAC activity (−17%, p < 0.01) and HDAC4 protein expression (−20%, p < 0.05) were downregulated in individuals with schizophrenia. Analyses of antipsychotic-free and antipsychotic-treated subgroups revealed enhanced H3K4me3 and H3K27ac (+24%–49%, p < 0.05) and reduced HDAC activity in the antipsychotic-treated, but not in the antipsychotic-free subgroup.
Limitations: Special care was taken to control the effect of confounding factors: age, sex, postmortem interval, and storage time. However, replication studies in bigger cohorts might strengthen the association between permissive HPTMs and schizophrenia.
Conclusion: We found global HPTM alterations consistent with an aberrantly permissive epigenome in schizophrenia. Further studies to elucidate the significance of enhanced permissive HPTMs in schizophrenia and its association with the mechanism of action of antipsychotic drugs are encouraged.
Introduction
Epigenetics has emerged as the mechanism to explain how environmental factors may have an impact on schizophrenia pathophisiology.1,2 Histone posttranslational modifications (HPTMs) and DNA methylation are the most studied epigenetic mechanisms in human tissue.3 While DNA methylation typically has a repressive impact on gene expression, the effects of HPTMs rely on both the modified residue and the particular type of modification (i.e., acetylation or methylation).4 In general, histone acetylation opens the chromatin and promotes the transcription of the associated gene, whereas histone methylation would affect gene transcription either way depending on the residue. Among all HPTMs writer, reader, and eraser enzymes, histone deacetylases (HDACs) catalyze histone deacetylation reactions, resulting in chromatin condensation and gene repression.
According to prior findings, drugs acting on epigenetic mechanisms may be beneficial in the treatment of schizophrenia.5,6 Thus, HDAC inhibitors have been tested as possible therapeutic agents acting on particular symptoms associated with schizophrenia.7 For example, valproate, one of the most studied antipsychotic augmenting drugs in schizophrenia,8 has shown HDAC inhibitor activity.9 Other HDAC inhibitors have also been suggested for the treatment of schizophrenia; i.e., vorinostat as an antipsychotic enhancer (NCT03263533) and sodium butyrate, postulated to improve cognitive impairment (NCT03010865). Although these clinical studies are currently withdrawn, interest in HDAC inhibitors in schizophrenia research continues.6
However, the status of HPTMs and their regulatory mechanisms in schizophrenia is not clear yet. Most studies have focused on the expression of HPTMs in gene promoters associated with schizophrenia.10–13 Other studies evaluating global changes in HPTMs in schizophrenia have mainly assessed HPTMs in peripheral tissues.14–16 Findings in the latter studies supported the hypothesis that a restrictive epigenome, characterized by reduced permissive and/or increased repressive HPTMs in the brain, may be associated with schizophrenia. However, results in brain tissue have not been conclusive, and a comprehensive study of global HPTMs in schizophrenia brain samples is still lacking. Moreover, these studies did not properly assess the effects of antipsychotic drug treatment on global HPTMs in schizophrenia.
We aimed to test the hypothesis that a restrictive epigenome underlies schizophrenia. To this end, we evaluated the global status of most relevant permissive and repressive HPTMs in the postmortem dorsolateral prefrontal cortex (DLPFC) of individuals with schizophrenia compared with controls matched for sex, age, and postmortem interval (PMI). Specifically, we studied permissive (H3K4me3, H3K9ac, H3K27ac) and repressive (H3K9me3 and H3K27me3) HPTMs at histone H3 because all of them had already been associated with schizophrenia pathology.12,14,15,17–19 We also included both permissive H4K16ac and repressive H4K20me3 HPTMs at histone H4. Among them, permissive H4K16ac at the ADRA2A/C gene promoter had also been described as altered in schizophrenia.12 We further addressed global HDAC activity, as well as the expression of class I and II HDAC proteins. We evaluated HDAC1 and HDAC2, which had been broadly studied in schizophrenia20–23 and associated with memory and cognitive processes24,25 as well as HDAC4, a class II HDAC protein implicated in cognitive disorders.26 Potential pharmacological effects of antipsychotic drugs were evaluated following classification of schizophrenia samples into antipsychotic-free and antipsychotic-treated subgroups according to the absence or presence of antipsychotic drugs in the blood at the time of death.
Methods
Postmortem human brain samples
Autopsies were performed in the Basque Institute of Legal Medicine, Bilbao, Spain. The DLPFC samples were carefully dissected and stored at −80°C until assayed. The DLPFC was selected because of its dual role in cognitive functioning and emotional processing, which are both impaired in individuals with schizophrenia.27,28 Schizophrenia diagnosis was confirmed antemortem by a psychiatrist using Diagnostic and Statistical Manual of Mental Disorders, fourth edition (DSM-IV) criteria, as recorded in the deceased individuals’ medical records. Each individual with schizophrenia was matched with a control based on sex, age, PMI, and, whenever possible, storage time (Table 1). Controls had no evidence of any psychiatric or neurologic disorder in their medical records. Full toxicological analyses were performed in blood samples from all individuals to detect the presence of psychotropic drugs, including antipsychotics and ethanol.
Sex, age, postmortem interval, and storage time of the samples of the main cohort included in the study
Complete demographic and methodological characteristics of individuals included in Western blot assays are shown in Appendix 1, Table 1, available at https://www.jpn.ca/lookup/doi/10.1503/jpn.230054/tab-related-content. Owing to limitations in tissue availability, some samples were exhausted during Western blot assays and replaced with DLPFC samples from individuals with similar characteristics in later enzymatic assays. Demographic and methodological characteristics of individuals included in enzymatic assays are described in Appendix 1, Table 2. The study was performed in compliance with legal and ethical policies of human postmortem tissue research studies and with the approval of the UPV/EHU Ethical Committee. Most schizophrenia and control samples included in this study had been included in a previous study from our group.12 In all experiments, schizophrenia and matched control samples were always processed in parallel.
Preparation of total homogenates for Western blot analysis
The DLPFC samples (approximately 100 mg) were homogenized in 10 volumes of ice-cold homogenization buffer (50 mM Tris-HCl, pH 6.8, 1 mM ethylenediaminetetraacetic acid solution, 1% sodium dodecyl sulfate [SDS], and 1% of a protease inhibitor cocktail [P-8340; Sigma-Aldrich]) using an Ultra-Turrax T8 (IKA Labortechnik). Total protein concentration in each sample was quantified using a detergent compatible (DC) protein assay kit (Bio-Rad) with bovine serum albumin (BSA) as a standard. Homogenates were diluted in loading buffer (15% β-mercaptoethanol, 2% SDS, 8% glycerol, 0.01% bromophenol blue), heated at 95°C for 5 minutes, and stored at −80°C in working aliquots.
Preparation of nucleosolic fraction for enzymatic assay
Samples (around 200 mg) were homogenized using an Ultra-Turrax T8 in 10 volumes of ice-cold phosphate-buffered saline (PBS; pH 7.4) containing 0.32 M sucrose and 1% phosphatase (NaF and Na3VO4) and protease inhibitors. Homogenates were centrifuged at 1100 g for 10 minutes at 4°C. Supernatants (S1) containing the cytosol and cell membranes were discarded. The pellets were washed in PBS supplemented with 1% phosphatase and protease inhibitors and centrifuged at 1000 g for 10 minutes at 4°C. These new pellets (P1), containing the nuclear fraction, were again resuspended in assay buffer (50 mM Tris-HCl, pH 8.0, 137 mM NaCl, 2.7 mM KCl, 1 mM MgCl2 · 6H2O, and 0.5 mM DTT) and sonicated (3 pulses of 5 seconds) in an ultrasonic liquid processor (CL188; Qsonica). After 10 minutes of incubation at 4°C, sonicated samples were centrifuged at 16 000 g for 20 minutes at 4°C. Supernatants containing nucleosol fraction (S2) were stored at −80°C. Protein concentration was quantified using a Pierce bicinchoninic acid (BCA) assay kit (Thermo Fisher Scientific) with BSA as a standard.
Western blot analysis
Homogenized samples were thawed and heated at 95°C for 5 minutes. Protein aliquots (20 μg per sample) were loaded onto 10%–15% SDS polyacrylamide gel electrophoresis (PAGE) gels and transferred to nitrocellulose membranes, as described elsewhere.29 Membranes were blocked (1 h at room temperature) with either 5% skimmed milk or 3% BSA in PBS containing 0.1% Tween-20, and subsequently incubated with primary (overnight at 4°C) and corresponding Alexa Fluor-680/800- or horseradish peroxidase–conjugated secondary antibodies (1 h at room temperature). Membrane immunodensity signal was then respectively detected in an Odyssey infrared imaging system (LI-COR Biosciences) or an Amersham Imager 680 (Cytiva Life Sciences) following addition of enhanced chemiluminescence (ECL) Western blot substrate (Thermo Fisher Scientific). A detailed description of primary antibodies and working conditions is provided in Appendix 1, Table 3. All scanned immunoblots were quantified in Image Studio Lite 5.2 (LI-COR Biosciences). Each case–control pair was run in at least 3 independent gels. To control for interassay variability and compare across the different immunoblots, a standard sample (made by pooling equal amounts of all samples) was loaded in triplicate in all gels.
HDAC enzymatic assays
HDAC activity was measured in the nucleosolic fraction (S2) using the fluorometric HDAC substrate Boc-Lys(Ac)-AMC/Boc-Lys-AMC (I-1875 and I-1880, respectively; Bachem), as reported previously30 with minor modifications. In this assay, the acetylated substrate (Boc-Lys(Ac)-AMC) is sequentially cleaved by tissue HDACs and trypsin, yielding the AMC fluorophore. The signal obtained is therefore proportional to the amount of endogenously active HDACs.
Reactions were performed in quadruplicate in 384-well plates. First, 10 μg protein aliquots of the S2 fraction were co-incubated with growing concentrations of the acetylated substrate Boc-Lys(Ac)-AMC (12.5–800 μM) in a final volume of 40 μL, at 37°C for 30 minutes in a plate shaker. HDAC reactions were stopped by adding 40 μL of the stop solution, containing 0.5% of an HDAC inhibitor cocktail (sc-362323; Santa Cruz Biotechnology) and 107 μM trypsin in PBS. Fluorescent signal was measured 15 minutes later using a CLARIOstarPlus microplate reader (BMG Labtech) at 360/460 nm excitation/emission wavelengths. The total amount of cleaved substrate (in μM/min) was interpolated from raw fluorescent signal using a standard curve of deacetylated Boc-Lys-AMC (2–40 μM) loaded in duplicate in all the plates. HDAC inhibitor cocktail inhibited the reaction in a concentration-dependent manner, confirming the specificity of the assay (Appendix 1, Figure 1).
Statistical analysis
Protein immunodensity values obtained in Western blot experiments were first normalized with corresponding β-actin values. HPTM immunodensity values were also normalized by histone H3 or H4 immunodensity values, which remained unchanged in schizophrenia (Appendix 1, Figure 2). Normalized values were then calculated in percent change to the ingel standard sample loaded in each gel. The mean across the experimental replicates obtained in at least 3 different gels was taken as the final estimate. HDAC activity was resolved using Michaelis–Menten nonlinear regression kinetics to obtain both maximal enzyme velocity (Vmax) and the KM constant (i.e., the substrate concentration at half-maximal velocity).
Statistical analyses were done in GraphPad Prism version 9.0 (GraphPad Software), SPSS statistics 26.0 (IBM), or InVivoStat software.31 Comparisons between matched case–control experimental groups were done using a 2-tailed paired t test. In HDAC activity experiments, statistical comparison of data fitting to a global or to 2 separate curves in individuals with schizophrenia and controls was done using an extra sum-of-squares F test. Multiple regression analysis using a stepwise regression method was done in the complete control and schizophrenia cohort to assess the contribution of sex, age, PMI, and storage time to the protein immunodensity and HDAC activity. If relevant, statistical differences between means were further studied with the significantly contributing variable as a covariate. Pairwise associations between numerical variables and/or experimental data were studied using a Pearson correlation test (r value) or simple linear regression analysis. Statistical significance was set to p < 0.05.
Results
HPTM immunodensities in the DLPFC of individuals with schizophrenia and controls
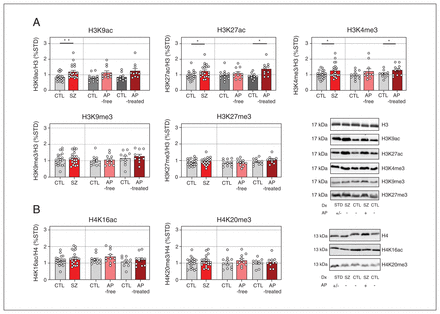
Western blot analyses of DLPFC tissue samples revealed greater immunodensities of H3K9ac (+42%, p = 0.009), H3K27ac (+28%, p = 0.035), and H3K4me3 (+24%, p = 0.042) proteins in individuals with schizophrenia compared with controls (Figure 1A). When individuals with schizophrenia were classified according to the presence (antipsychotic-treated) or absence (antipsychotic-free) of antipsychotics in the blood, H3K27ac (+49%, p = 0.021) and H3K4me3 (+24%, p = 0.043) immunodensities were significantly increased in those who were antipsychotic-treated, but not in those who were antipsychotic-free, compared with their corresponding controls (Figure 1A).
Immunodensity of histone H3 (A) and H4 (B) posttranslational modifications in the dorsolateral prefrontal cortex of individuals with schizophrenia (SZ) and matched controls (CTL). The data shown are mean values of 3–4 independent experiments and are expressed as corrected immunodensity values normalized for β-actin and for either histone H3 or H4 immunodensity as well as to a standard sample (STD) loaded in the same gel. Representative images of studied histone posttranslational modification (HPTM) immunodensities in antipsychotic (AP)-free, AP-treated individuals, and in matched controls are shown. Data correspond to 21 individuals with schizophrenia (11 antipsychoticfree, 10 antipsychotic-treated) and 21 matched controls. *p < 0.05 v. matched controls (2-tailed paired t test). **p < 0.01 v. matched controls (2-tailed paired t test).
Cortical amounts of H3K9me3, H3K27me3, H4K16ac, and H4K20me3 in individuals with schizophrenia did not differ significantly from those in controls, either when all those with schizophrenia were pooled together or when divided into antipsychotic-free of antipsychotic-treated subgroups (Figure 1).
Multiple regression analyses performed to study the possible influence of demographic and methodological variables on HPTM immunodensity showed no influence of nominal (sex) or continuous (age, PMI, storage time) variables.
HDAC activity in the DLPFC of individuals with schizophrenia and controls
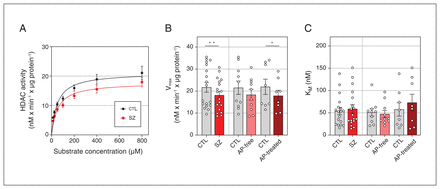
We tested whether the increased acetylation of brain histone H3 may underlie a global deficit in HDAC enzymatic activity associated with schizophrenia. Saturation curves of HDAC activity followed a hyperbolic curve as a function of substrate concentration Boc-Lys(Ac)-AMC, which fitted to the nonlinear Michaelis–Menten equation (schizophrenia r = 0.694, controls r = 0.710; Figure 2A).
(A) Histone deacetylase (HDAC) activity in the dorsolateral prefrontal cortex of individuals with schizophrenia (SZ) and matched controls (CTL) adjusted to Michaelis–Menten nonlinear regression model. Values are expressed as converted substrate in nM every minute per μg protein. HDAC activity parameters for (B) maximal velocity (Vmax) and (C) Michaelis constant (KM). Data correspond to 18 individuals with schizophrenia (10 antipsychotic [AP]-free, 8 antipsychotic-treated) and 18 matched controls.*p < 0.05 v. matched controls (2-tailed paired t test). **p < 0.01 v. matched controls (2-tailed paired t test).
HDAC activity differed significantly between individuals with schizophrenia and controls (F2,244 = 8.12, p = 0.004; Figure 2A). Individual analyses of the kinetic parameters found a significant reduction of Vmax in the schizophrenia group compared with matched controls (−17%, p = 0.007; Figure 2B), but no significant differences in KM values (Figure 2C). Following classification by antipsychotic presence or absence in the blood, similar reductions in Vmax values were observed in antipsychotic-free (−15%, p > 0.05) and antipsychotic-treated (−18.5%, p = 0.048) subgroups, but reached statistical significance only in the antipsychotic-treated subgroup when compared with controls (Figure 2B).
Multiple regression analyses were performed to study the effect of potentially confounding variables (sex, age, PMI, and storage time) on the estimated parameters of HDAC activity in postmortem brain samples. Sex had a significant influence on KM (β = 0.523, p = 0.001), with higher values in females than in males (Appendix 1, Figure 2A). For Vmax, a significant model emerged (F2,33 = 8.115, p = 0.001) with the contribution of storage time (β = 0.651) and age (β = 0.491) (see Appendix 1, Figure 2B and 2C for individual correlation plots). Although the case–control paired design had already accounted for these variables, statistical analyses controlling for storage time and age as covariates were further assessed. The analysis confirmed the significant Vmax differences between individuals with schizophrenia and controls (p = 0.015 and p = 0.007, with storage time and age, respectively, as covariates). Decreased Vmax in the antipsychotic-treated subgroup compared with matched controls was also confirmed (p = 0.076 and p = 0.049 for storage time and age, respectively).
HDAC1, HDAC2, and HDAC4 immunodensities in the DLPFC of individuals with schizophrenia and controls
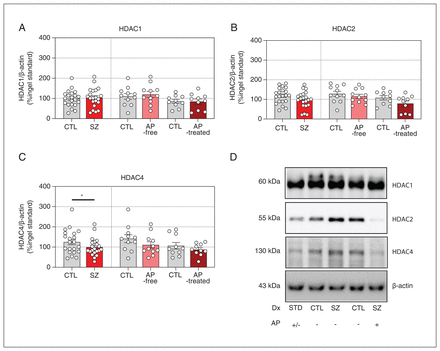
Next, we addressed the possibility that downregulated HDAC activity in individuals with schizophrenia might be due to changes in protein expression levels of these enzymes. Western blot analyses did not reveal significant differences in HDAC1 or HDAC2 immunodensities between the analyzed groups and subgroups. However, cortical amounts of HDAC4 were significantly lower in individuals with schizophrenia than in their matched controls (−20%, p = 0.024). This difference did not hold significance after classification of individuals with schizophrenia into antipsychotic-free and antipsychotic-treated subgroups (Figure 3).
Protein expression levels of (A) histone deacetylase 1 (HDAC1), (B) HDAC2, and (C) HDAC4 in the dorsolateral prefrontal cortex of individuals with schizophrenia (SZ) and matched controls (CTL). The data shown are mean values of 3–4 independent experiments and are expressed as corrected immunodensity values normalized for β-actin as well as to a standard sample (STD) loaded in the same gel. Representative images of studied HDAC immunodensity in antipsychotic (AP)-free and antipsychotic-treated individuals and in matched controls are shown. Data correspond to 21 individuals with schizophrenia (11 antipsychotic-free, 10 antipsychotic-treated) and 21 matched controls. *p < 0.05 v. matched controls (2-tailed paired t test).
Multiple regression analysis further assessed the potential effects of demographic and methodological variables on HDAC enzyme brain expression. HDAC1 immunodensity was potentially influenced by PMI (β = −0.332, p = 0.032) (Appendix 1, Figure 3). The effect of PMI on HDAC1 immunodensity may have been a result of including 2 samples with higher PMI (48 h and 58 h). Removal of these values eliminated the association between PMI and HDAC1 (p = 0.154). No other variables had significant effects on brain immunodensities of HDAC1, HDAC2, or HDAC4.
Association between HDAC activity and HPTMs and HDAC densities
Finally, we studied the possible association of altered HPTM and HDAC immunodensities with KM and Vmax values obtained from HDAC activity assays (Table 2). Pearson correlation analysis of data from all included individuals showed a negative association between HDAC1, HDAC2, and HDAC4 protein expression and KM values that were significant for HDAC1 and HDAC2, reflecting the dependency between HDAC protein expression and the performance of the deacetylation reaction. The KM values also correlated with H3K9me3 (data from all included individuals), and with H3K27ac and H4K16ac (data from controls). Interestingly, we observed a significant negative correlation between H3K27ac and H4K16ac and the velocity of the reaction depicted by the Vmax value in controls. However, in individuals with schizophrenia, only the association between KM and HDAC2 expression remained significant.
Pairwise associations between KM and Vmax coefficients estimated from global HDAC enzymatic activity assays and the Western blot targets
Discussion
We evaluated the status of histone-level epigenetic mechanisms in portmortem brain samples from individuals with schizophrenia. In contrast to the suggested restrictive epigenome hypothesis of schizophrenia,14,15 our results showed that HPTMs favouring gene expression, including H3K4me3, H3K9ac, and H3K27ac, are elevated in the DLPFC of individuals with schizophrenia, likely due to reduced expression of HDAC4 and to the dysregulation of the overall HDAC enzymatic activity. These data suggest an open chromatin in the cortex of individuals with schizophrenia, especially in those who were taking antipsychotics at the time of death.
In line with our results, a recent chromatin immunoprecipitation study followed by sequencing (ChIP-seq) in the DLPFC of individuals with schizophrenia found that the majority of differential H3K4me3 and H3K27ac peaks were actually elevated in both neuronal and glial nuclei of the schizophrenia cohort.18 Enhanced H2A acetylation in the DLPFC of individuals with schizophrenia32 as well as increases in permissive H3S10 phosphorylation in blood cells of individuals with schizophrenia have also been described recently.16 On the other hand, a seminal study assessing overall changes in HPTM levels in postmortem brain samples from individuals with schizophrenia found no alteration in any of the studied marks (including H3K4me3 and H3K9ac).33 As mentioned, some other evidence indicates a restrictive epigenome in schizophrenia.14 Increased levels of both repressive H3K9me2 and histone methyltransferases (HMTs) were observed in the parietal cortex and in circulating lymphocytes of individuals with schizophrenia,14 these findings being later ascribed to men but not to women.34 Decreased levels of permissive histone acetylation, such as H3K9/K14ac, were also found in lymphocytes of patients with schizophrenia.15 Contrasting data across the studies may be attributed to differences in the analyzed tissues (e.g., blood cells v. postmortem brain tissue), selected brain areas (parietal v. prefrontal cortices), the studied HPTMs, sex, and antipsychotic medication. In addition, other factors, including poor dietary habits, weight gain, smoking, and comorbid substance use, usually associated with schizophrenia35,36 might be able to influence epigenetics.37 However, neither the present nor previous studies controlled for any of these factors. Notably, the majority of DLPFC samples, in which we observed enhanced expression of permissive H3K9ac, H3K27ac, and H3K4me3 in schizophrenia, came from men.
Unfortunately, treatment effects are often neglected in postmortem brain studies. Here, we observed that the increase in H3K27ac and H3K4me3 reached statistical significance in antipsychotic-treated, but not in antipsychotic-free individuals, which may indicate a regulatory effect of antipsychotic drugs on HPTMs. However, although alterations were more easily observed in those with detectable antipsychotic drug levels in blood, we cannot discard past use of antipsychotic drugs in antipsychotic-free individuals, which may have affected HPTMs. The effect of antipsychotic treatment may also underlie the differences between the present and previous studies reporting lack of alterations33 or a restrictive epigenome14 in the postmortem brain of individuals with schizophrenia. Notably, previous studies addressed potential medication effects based on prescription records,34 which may contrast with lack of adherence to treatment among patients with schizophrenia.38
Preclinical studies have shed some light into this question. Relevant animal models in schizophrenia have shown decreased H3K4me339 and H3K9ac in the brain.40–42 However, reduced H3K9ac in a mouse model based on chronic phencyclidine administration was attenuated by clozapine pretreatment.40 In addition, reports on the cellular mechanism by which antipsychotics such as haloperidol41 and risperidone42 induce permissive HPTMs have also been published. Thus, together with our results, preclinical studies support the idea that antipsychotic treatment might be able to regulate HPTMs toward an open chromatin state.
Altered HDAC enzyme activity or expression may explain the enhanced histone acetylation observed in schizophrenia. Indeed, correlation analysis of HPTM immunodensities and HDAC activity showed that in controls, global HDAC activity was significantly associated with H3K27ac and H4K16ac levels. However, the significance of these associations was lost in the schizophrenia cohort, emphasizing the disruption of this regulatory epigenetic mechanism in schizophrenia. This dysregulation may be associated with the reduced HDAC4 protein expression found in the DLPFC of individuals with schizophrenia. HDAC4 is particularly abundant in brain tissue compared with other HDAC family members and has been associated with cognitive impairment in several mental disorders.26 Although HDAC4 shows little enzymatic activity, it has been attributed a scaffolding function bringing together substrate and other HDAC-containing complexes.43 Taken together, reduced HDAC4 protein expression may cause an overall downregulation of HDAC enzymatic activity, which may ultimately result in the histone hyperacetylation observed in the DLPFC of individuals with schizophrenia.
To our knowledge, this is the first study to evaluate HDAC activity in human postmortem brain tissue. For this purpose, we adapted the methodology from Reddy and colleagues30 and obtained a reliable method whose performance was dependent on HDAC protein expression. An important point in postmortem brain tissue studies is to keep under control the effect of confounding demographic and methodological factors.44 According to our results, HDAC activity is influenced by age, sex, and storage time of the samples. In all cases, the effect of these confounding factors was prevented by careful matching of schizophrenia and control samples.
Previous postmortem brain studies in schizophrenia have reported inconsistent data regarding HDAC gene or protein expression. Increased HDAC120,21,45 as well as unaltered HDAC1 and reduced HDAC2 gene expression,23 or unaltered HDAC1, HDAC2, and HDAC410 protein expression have been reported in brains of individuals with schizophrenia. Interestingly, a recent study with a HDAC1, HDAC2, HDAC3, and HDAC6 radiotracer [11C]Martinostat revealed reduced HDAC expression in the DLPFC of individuals with schizophrenia compared with controls.22
Like the reported HPTM alterations, downregulated HDAC activity rate was observed in antipsychotic-treated, but not in antipsychotic-free individuals with schizophrenia. Prior studies also addressed the pharmacological effects of antipsychotic medication on HDAC expression in postmortem brain from individuals with schizophrenia, with contrasting results. For example, HDAC1 gene45 and HDAC2 protein10 expression were increased in antipsychotic-treated individuals with schizophrenia. Another study analyzing brain HDAC1 and HDAC2 gene expression in a large cohort of individuals with schizophrenia revealed no significant influence of antipsychotic treatment.23 Thus, the effect of antipsychotic treatment on HDAC activity and protein expression does not seem completely clear, and present observations of increased histone acetylation and reduced HDAC activity in antipsychotic-treated individuals with schizophrenia may involve other mechanisms. Actually, histone acetylation is tightly regulated by histone acetyltransferase (HAT) and HDAC enzymes. Among others, CREB-binding protein (CBP) complex enhances histone acetylation, as it recruits HAT and other required components for acetylation.46 In this context, a meta-analysis of postmortem brain studies concluded that schizophrenia is associated with a greater expression of CREB1 and CREBBP transcripts,47 in line with the enhanced permissive HPTMs observed herein.
One of the key questions that emerges from our study is to ascertain the genes affected by the enhanced permissive HPTMs in schizophrenia. Recent epigenomic works have shown HPTM- and cell-type-specific alterations.18 Enriched H3K4me3 peaks have been found at a major histocompatibility complex locus on chromosome 6 in bulk brain tissue of individuals with schizophrenia,48 but histone peaks were unaltered in neuronal cells.19 Altered H3K27ac has been reported in both bulk tissue and neuronal cells, with dysregulated peaks in genes associated with immune response, among others.19 Moreover, some of the H3K4me3 and H3K27ac alterations would be specifically altered in either antipsychotic-treated or antipsychotic-free individuals.18
Limitations
An important point in the present and other studies performed in postmortem brain tissue is to keep under control the effect of possible confounding factors.44 Although we took special care to control for sex, age, PMI, and storage time, the effect of other factors such as time from diagnosis and past antipsychotic treatment could not be explored. Limited access to this kind of information as well as to other potentially important factors in epigenetics is a limitation of postmortem brain studies that might be addressed in replication studies in bigger cohorts.
Conclusion
In recent decades, HDAC inhibitors have been postulated as a coadjuvant treatment of current antipsychotic drugs in schizophrenia and other mental disorders.49,50 However, the present results are not compatible with the restrictive epigenome hypothesis in schizophrenia and do not fully support the use of HDAC inhibitors in combination with antipsychotic drugs. Furthermore, significant alterations observed in antipsychotic-treated but not in antipsychoticfree individuals with schizophrenia suggest that enhanced permissive HPTMs could actually be induced by antipsychotic drugs. Finally, owing to the possible involvement of the class IIa enzyme HDAC4 in schizophrenia, the specific role of different HDAC isoforms and selectivity of HDAC inhibitors should be optimized before this coadjuvant therapy is advanced into clinical practice.
Acknowledgements
The study was supported by the Spanish Ministry of Science and Innovation (MCIN/AEI/FEDER Funds; grant RTI2018-094414-A-I00, PID2022-1378480B-I00, and RYC-2016-19282 to A. Ramos-Miguel) and the Basque Government (grants IT1211/19 and IT1512/22). O. Martínez-Peula is a recipient of a predoctoral fellowship from the University of the Basque Country (UPV/EHU, 19/306). The authors thank staff members of the Basque Institute of Legal Medicine for their cooperation.
Footnotes
Competing interests: G. Rivero is a member of the Committee for the Evaluation of New Drugs in Primary Health Care (Basque Government) and a member of the Ethical Committee of UPV/EHU. No other competing interests were declared.
Contributors: L.F. Callado, J.J. Meana, G. Rivero and A. Ramos-Miguel designed the study. O. Martínez-Peula and B. Morentin acquired the data, which O. Martínez-Peula, G. Rivero, and A. Ramos-Miguel analyzed. O. Martínez-Peula, G. Rivero, and A. Ramos-Miguel wrote the article. All of the authors revised it critically for important intellectual content, gave final approval of the version to be published, and agreed to be accountable for all aspects of the work.
- Received April 4, 2023.
- Revision received September 28, 2023.
- Accepted November 3, 2023.
This is an Open Access article distributed in accordance with the terms of the Creative Commons Attribution (CC BY-NC-ND 4.0) licence, which permits use, distribution and reproduction in any medium, provided that the original publication is properly cited, the use is noncommercial (i.e., research or educational use), and no modifications or adaptations are made. See: https://creativecommons.org/licenses/by-nc-nd/4.0/