Abstract
Background
Easily accessible biomarkers are needed for the early identification of individuals at risk of developing Alzheimer’s disease (AD) in large population screening strategies.
Objectives
This study evaluated the potential of plasma β-amyloid (Aβ) biomarkers in identifying early stages of AD and predicting cognitive decline over the following two years.
Design
Total plasma Aβ42/40 ratio (TP42/40) was determined in 83 cognitively normal individuals (CN) and 145 subjects with amnestic mild cognitive impairment (a-MCI) stratified by an FDG-PET AD-risk pattern.
Results
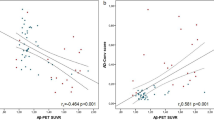
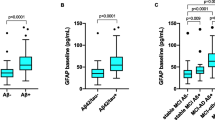
Significant lower TP42/40 ratio was found in a-MCI patients compared to CN. Moreover, a-MCIs with a highrisk FDG-PET pattern for AD showed even lower plasma ratio levels. Low TP42/40 at baseline increased the risk of progression to dementia by 70%. Furthermore, TP42/40 was inversely associated with neocortical amyloid deposition (measured with PiB-PET) and was concordant with the AD biomarker profile in cerebrospinal fluid (CSF).
Conclusions
TP42/40 demonstrated value in the identification of individuals suffering a-MCI, in the prediction of progression to dementia, and in the detection of underlying AD pathology revealed by FDG-PET, Amyloid-PET and CSF biomarkers, being, thus, consistently associated with all the well-established indicators of AD.


Similar content being viewed by others
References
Lacosta AM, Insua D, Badi H, Pesini P, Sarasa M. Neurofibrillary Tangles of Abetax–40 in Alzheimer’s Disease Brains. J Alzheimers Dis 2017;58:661–667.
Brookmeyer R, Johnson E, Ziegler–Graham K, Arrighi HM. Forecasting the global burden of Alzheimer’s disease. Alzheimers Dement 2007 Jul;3:186–191.
Doody RS, Thomas RG, Farlow M, et al. Phase 3 trials of solanezumab for mild–to–moderate Alzheimer’s disease. N Engl J Med 2014 Jan 23;370:311–321.
Salloway S, Sperling R, Fox NC, et al. Two phase 3 trials of bapineuzumab in mild–to–moderate Alzheimer’s disease. N Engl J Med 2014 Jan 23;370:322–333.
Sperling RA, Rentz DM, Johnson KA, et al. The A4 study: stopping AD before symptoms begin? Sci Transl Med 2014 Mar;%19;6:228fs13.
Albert MS, DeKosky ST, Dickson D, et al. The diagnosis of mild cognitive impairment due to Alzheimer’s disease: recommendations from the National Institute on Aging–Alzheimer’s Association workgroups on diagnostic guidelines for Alzheimer’s disease. Alzheimers Dement 2011 May;7:270–279.
Bohnen NI, Djang DS, Herholz K, Anzai Y, Minoshima S. Effectiveness and safety of 18F–FDG PET in the evaluation of dementia: a review of the recent literature. J Nucl Med 2012 Jan;53:59–71.
Fagan AM, Mintun MA, Mach RH, et al. Inverse relation between in vivo amyloid imaging load and cerebrospinal fluid Abeta42 in humans. Ann Neurol 2006 Mar;59:512–519.
Weiner MW, Veitch DP, Aisen PS, et al. The Alzheimer’s Disease Neuroimaging Initiative: a review of papers published since its inception. Alzheimers Dement 2012 Feb;8:S1–68.
Rowe CC, Bourgeat P, Ellis KA, et al. Predicting Alzheimer disease with beta–amyloid imaging: results from the Australian imaging, biomarkers, and lifestyle study of ageing. Ann Neurol 2013 Dec;74:905–913.
Blennow K, Dubois B, Fagan AM, Lewczuk P, de Leon MJ, Hampel H. Clinical utility of cerebrospinal fluid biomarkers in the diagnosis of early Alzheimer’s disease. Alzheimers Dement 2015 Jan;11:58–69.
Hansson O, Zetterberg H, Vanmechelen E, et al. Evaluation of plasma Abeta(40) and Abeta(42) as predictors of conversion to Alzheimer’s disease in patients with mild cognitive impairment. Neurobiol Aging 2010 Mar;31:357–367.
Lopez OL, Kuller LH, Mehta PD, et al. Plasma amyloid levels and the risk of AD in normal subjects in the Cardiovascular Health Study. Neurology 2008 May 6;70:1664–1671.
Lovheim H, Elgh F, Johansson A, et al. Plasma concentrations of free amyloidbeta cannot predict the development of Alzheimer’s disease. Alzheimers Dement 2016 Sep 28;10.
Cosentino SA, Stern Y, Sokolov E, et al. Plasma beta–Amyloid and Cognitive Decline. Arch Neurol 2010 Aug 9.
Mehta PD, Pirttila T, Patrick BA, Barshatzky M, Mehta SP. Amyloid beta protein 1–40 and 1–42 levels in matched cerebrospinal fluid and plasma from patients with Alzheimer disease. Neurosci Lett 2001 May 18;304:102–106.
Chouraki V, Beiser A, Younkin L, et al. Plasma amyloid–beta and risk of Alzheimer’s disease in the Framingham Heart Study. Alzheimers Dement 2015 Mar;11:249–257.
Graff–Radford NR, Crook JE, Lucas J, et al. Association of low plasma Abeta42/Abeta40 ratios with increased imminent risk for mild cognitive impairment and Alzheimer disease. Arch Neurol 2007 Mar;64:354–362.
Lambert JC, Schraen–Maschke S, Richard F, et al. Association of plasma amyloid beta with risk of dementia: the prospective Three–City Study. Neurology 2009 Sep 15;73:847–853.
Fandos N, Perez–Grijalba V, Pesini P, et al. Plasma amyloid beta 42/40 ratios as biomarkers for amyloid beta cerebral deposition in cognitively normal individuals. Alzheimers Dement (Amst) 2017 Sep 12;8:179–187. doi: 10.1016/j. dadm.2017.07.004. eCollection;%2017.:179–187.
Ovod V, Ramsey KN, Mawuenyega KG, et al. Amyloid beta concentrations and stable isotope labeling kinetics of human plasma specific to central nervous system amyloidosis. Alzheimers Dement 2017 Aug;13:841–849.
Nakamura A, Kaneko N, Villemagne VL, et al. High performance plasma amyloid–beta biomarkers for Alzheimer’s disease. Nature 2018 Feb 8;554:249–254.
Lezak MD, Howieson DB, Loring DW. Neuropsychological Assessment, 4th Edition ed. New York: Oxofrd University Press, 2004.
Petersen RC. Mild cognitive impairment as a diagnostic entity. J Intern Med 2004 Sep;256:183–194.
American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, 4th Edition ed. Washington D.C.: American Psychiatric Association, 2000.
Jagust W, Reed B, Mungas D, Ellis W, Decarli C. What does fluorodeoxyglucose PET imaging add to a clinical diagnosis of dementia? Neurology 2007 Aug 28;69:871–877.
Espinosa A, Alegret M, Pesini P, et al. Cognitive Composites Domain Scores Related to Neuroimaging Biomarkers within Probable–Amnestic Mild Cognitive Impairment–Storage Subtype. J Alzheimers Dis 2017;57:447–459.
Nordberg A, Carter SF, Rinne J, et al. A European multicentre PET study of fibrillar amyloid in Alzheimer’s disease. Eur J Nucl Med Mol Imaging 2013 Jan;40:104–114.
Perez–Grijalba V, Fandos N, Canudas J, et al. Validation of Immunoassay–Based Tools for the Comprehensive Quantification of Abeta40 and Abeta42 Peptides in Plasma. J Alzheimers Dis 2016 Aug 10;54:751–762.
Perez–Grijalba V, Pesini P, Allue JA, et al. Abeta1–17 is a major amyloid–beta fragment isoform in cerebrospinal fluid and blood with possible diagnostic value in Alzheimer’s disease. J Alzheimers Dis 2015;43:47–56.
Hixson JE, Vernier DT. Restriction isotyping of human apolipoprotein E by gene amplification and cleavage with HhaI. J Lipid Res 1990 Mar;31:545–548.
van OM, Hofman A, Soares HD, Koudstaal PJ, Breteler MM. Plasma Abeta(1–40) and Abeta(1–42) and the risk of dementia: a prospective case–cohort study. Lancet Neurol 2006 Aug;5:655–660.
Abdullah L, Luis C, Paris D, et al. Serum Abeta levels as predictors of conversion to mild cognitive impairment/Alzheimer disease in an ADAPT subcohort. Mol Med 2009 Nov;15:432–437.
Yaffe K, Weston A, Graff–Radford NR, et al. Association of plasma betaamyloid level and cognitive reserve with subsequent 19;305:261–266.
Okereke OI, Xia W, Selkoe DJ, Grodstein F. Ten–year change in plasma amyloid beta levels and late–life cognitive decline. Arch Neurol 2009 Oct;66:1247–1253.
Koyama A, Okereke OI, Yang T, Blacker D, Selkoe DJ, Grodstein F. Plasma amyloid–beta as a predictor of dementia and cognitive decline: a systematic review and meta–analysis. Arch Neurol 2012 Jul;69:824–831.
Rembach A, Watt AD, Wilson WJ, et al. Plasma amyloid–beta levels are significantly associated with a transition toward Alzheimer’s disease as measured by cognitive decline and change in neocortical amyloid burden. J Alzheimers Dis 2014;40:95–104.
Janelidze S, Stomrud E, Palmqvist S, et al. Plasma beta–amyloid in Alzheimer’s disease and vascular disease. Sci Rep 2016 May 31;6:26801. doi: 10.1038/srep26801.:26801.
Shaw LM, Vanderstichele H, Knapik–Czajka M, et al. Cerebrospinal fluid biomarker signature in Alzheimer’s disease neuroimaging initiative subjects. Ann Neurol 2009 Apr;65:403–413.
Toledo JB, Vanderstichele H, Figurski M, et al. Factors affecting Abeta plasma levels and their utility as biomarkers in ADNI. Acta Neuropathol 2011 Oct;122:401–413.
Vanderstichele H, Van KE, Hesse C, et al. Standardization of measurement of beta–amyloid(1–42) in cerebrospinal fluid and plasma. Amyloid 2000 Dec;7:245–258.
Roher AE, Esh CL, Kokjohn TA, et al. Amyloid beta peptides in human plasma and tissues and their significance for Alzheimer’s disease. Alzheimers Dement 2009 Jan;5:18–29.
Poljak A, Crawford JD, Smythe GA, et al. The Relationship Between Plasma Abeta Levels, Cognitive Function and Brain Volumetrics: Sydney Memory and Ageing Study. Curr Alzheimer Res 2016;13:243–255.
Figurski MJ, Waligorska T, Toledo J, et al. Improved protocol for measurement of plasma beta–amyloid in longitudinal evaluation of Alzheimer’s Disease Neuroimaging Initiative study patients. Alzheimers Dement 2012 Jul;8:250–260.
Insel PS, Mattsson N, Mackin RS, et al. Biomarkers and cognitive endpoints to optimize trials in Alzheimer’s disease. Ann Clin Transl Neurol 2015 May;2:534–547.
Author information
Authors and Affiliations
Consortia
Corresponding author
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Pérez-Grijalba, V., Romero, J., Pesini, P. et al. Plasma Aβ42/40 Ratio Detects Early Stages of Alzheimer’s Disease and Correlates with CSF and Neuroimaging Biomarkers in the AB255 Study. J Prev Alzheimers Dis 6, 34–41 (2019). https://doi.org/10.14283/jpad.2018.41
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.14283/jpad.2018.41