Abstract
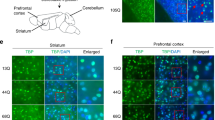
We report the identification of a full-length novel β-spectrin II gene (βSpIIΣ2) in human brain. The βSpIIΣ2 gene has 32 exons encoding an actin-binding domain, followed by 17-spectrin repeats, and a short COOH-terminal regulatory region that lacks the Pleckstrin homology (PH) domain. Pair-wise sequence analysis showed an additional 36 and 28 amino acids located at the NH2 and COOH-terminal regions of βSpIIΣ2, respectively. Northern-blot analysis showed an abundant expression of βSpIIΣ2 transcripts in brain, lung, and kidney. Western-blot analysis confirmed the predicted ∼225 kD molecular size of βSpIIΣ2 protein in these same tissues. In brain, immunofluorescent staining revealed that βSpIIΣ2 was enriched in cerebellar neurons, with specific enrichment in Purkinje cell bodies, but not in dendrites. Of considerable interest, neurofibromatosis type 2 (NF2) gene product schwannomin was found to co-immunoprecipitate with βSpIIΣ2 in cultured Purkinje cells. These results suggest that βSpIIΣ2 may play an important role in the assembly of the specialized plasma membrane domain of Purkinje neurons and that schwannomin may be involved in actin-cytoskeleton organization by interacting with βSpIIΣ2.
Similar content being viewed by others
References
Altschul, S. F. and Gish, W. (1996) Local alignment statistics. Methods Enzymol. 266, 460–480.
Baptista, C. A., Hatten, M. E., Blazeski, R., and Mason, C. A. (1994) Cell-cell interactions influence survival and differentiation of purified Purkinje cells in vitro. Neuron 12, 243–260.
Bashir, R., Britton, S., Strachan, T., Keers, S., Vafiadaki, E., Lako, M., et al. (1998) A gene related to Caenorhabditis elegans spermatogenesis factor fer-1 is mutated in limb-girdle muscular dystrophy type 2B. Nat. Genet. 20, 37–42.
Berghs, S., Aggujaro, D., Dirkx, R. Jr., Maksimova, E., Stabach, P., Hermel, J. M., et al. (2000) betaIV spectrin, a new spectrin localized at axon initial segments and nodes of anvier in the central and peripheral nervous system. J. Cell Biol. 151, 985–1002.
Clark, M. B., Ma, Y., Bloom, M. L., Barker, J. E., Zagon, I. S., Zimmer, W. E., and Goodman, S. R. (1994) Brain alpha erythroid spectrin: identification, compartmentalization, and beta spectrin associations. Brain Res. 663, 223–236.
Claudio, J. O., Lutchman, M., and Rouleau, G. A. (1995) Widespread but cell type-specific expression of the mouse neurofibromatosis type 2 gene. Neuroreport 6, 1942–1946.
Dahlberg, W. K., Little, J. B., Fletcher, J. A., Suit, H. D., and Okunieff, P. (1993) Radiosensitivity in vitro of human soft tissue sarcoma cell lines and skin fibroblasts derived from the same patients. Intl. J. Radiat. Biol. 63, 191–198.
Fagerheim, T., Raeymaekers, P., Tonnessen, F. E., Pedersen, M., Tranebjaerg, L., and Lubs, H. A. (1999) A new gene (DYX3) for dyslexia is located on chromosome 2. J. Med. Genet. 36, 664–669.
Furuya, S., Mitoma, J., Makino, A., and Hirabayashi, Y. (1998) Ceramide and its interconvertible metabolite sphingosine function as indispensable lipid factors involved in survival and dendritic differentiation of cerebellar Purkinje cells. J. Neurochem. 71, 366–377.
Gallagher, P. G. and Forget, B. G. (1998) Hematologically important mutations: spectrin and ankyrin variants in hereditary spherocytosis. Blood Cells Mol. Dis. 24, 539–543.
Goodman, S. R., Zimmer, W. E., Clark, M. B., Zagon, I. S., Barker, J. E., and Bloom, M. L. (1995) Brain spectrin: of mice and men. Brain Res. Bull. 36, 593–606.
Hayes, N. V., Scott, C., Heerkens, E., Ohanian, V., Maggs, A. M., Pinder, J. C., et al. (2000) Identification of a novel C-terminal variant of beta II spectrin: two isoforms of beta II spectrin have distinct intracellular locations and activities. J. Cell Sci. 113, 2023–2034.
Hazan, J., Davoine, C. S., Mavel, D., Fonknechten, N., Paternotte, C., Fizames, C., et al. (1999) A fine integrated map of the SPG4 locus excludes an expanded CAG repeat in chromosome 2p-linked autosomal dominant spastic paraplegia. Genomics 60, 309–319.
Hirai, H. and Matsuda, S. (1999) Interaction of the C-terminal domain of delta glutamate receptor with spectrin in the dendritic spines of cultured Purkinje cells. Neurosci. Res. 34, 281–287.
Hu, R. J., Watanabe, M., and Bennett, V. (1992) Characterization of human brain cDNA encoding the general isoform of beta-spectrin. J. Biol. Chem. 267, 18,715–18,722.
Hu, R. J., Moorthy, S., and Bennett, V. (1995) Expression of functional domains of beta G-spectrin disrupts epithelial morphology in cultured cells. J. Cell Biol. 128, 1069–1080.
Illarioshkin, S. N., Ivanova-Smolenskaya, I. A., Tanaka, H., Poleshchuk, V. V., Markova, E. D., and Tsuji, S. (1997) Refined genetic location of the chromosome 2p-linked progressive muscular dystrophy gene. Genomics 42, 345–348.
Kermani, S., Gregory-Evans, K., Tarttelin, E. E., Bellingham, J., Plant, C., Bird, A. C., et al. (1999) Refined genetic and physical positioning of the gene for Doyne honeycomb retinal dystrophy (DHRD). Hum. Genet. 104, 77–82.
Kirschner, L. S., Taymans, S. E., Pack, S., Pak, E., Pike, B. L., Chandrasekharappa, S. C., Zhuang, Z., and Stratakis, C. A. (1999) Genomic mapping of chromosomal region 2p15–p21 (D2S378-D2S391): integration of Genemap’98 within a framework of yeast and bacterial artificial chromosomes. Genomics 62, 21–33.
Liu, J., Aoki, M., Illa, I., Wu, C., Fardeau, M., Angelini, C., et al. (1998) Dysferlin, a novel skeletal muscle gene, is mutated in Miyoshi myopathy and limb girdle muscular dystrophy. Nat. Genet. 20, 31–36.
Lombardo, C. R., Weed, S. A., Kennedy, S. P., Forget, B. G., and Morrow, J. S. (1994) Beta II-spectrin (fodrin) and beta I epsilon 2-spectrin (muscle) contain NH2-and COOH-terminal membrane association domains (MAD1 and MAD2). J. Biol. Chem. 269, 29,212–29,219.
Ma, Y., Zimmer, W. E., Riederer, B. M., Bloom, M. L., Barker, J. E., Goodman, S. R., and Goodman, S. M. (1993) The complete amino acid sequence for brain beta spectrin (beta fodrin): relationship to globin sequences. Brain Res. Mol. Brain Res. 18, 87–99.
Malchiodi-Albedi, F., Ceccarini, M., Winkelmann, J. C., Morrow, J. S., and Petrucci, T. C. (1993) The 270 kDa splice variant of erythrocyte beta-spectrin (beta I sigma 2) segregates in vivo and in vitro to specific domains of cerebellar neurons. J. Cell Sci. 106, 67–78.
Marchesi, V. T. and Steers, E., Jr. (1968) Selective solubilization of a protein component of the red cell membrane. Science 159, 203–204.
Martuza, R. L. and Eldridge, R. (1988) Neurofibromatosis 2 (bilateral acoustic neurofibromatosis). N. Engl. J. Med. 318, 684–688.
Mishra, L., Cai, T., Yu, P., Monga, S. P., and Mishra, B. (1999) Elf3 encodes a novel 200-kD beta-spectrin: role in liver development. Oncogene 18, 353–364.
Mishra, L., Cai, T., Levine, A., Weng, D., Mezey, E., Mishra, B., and Gearhart, J. (1998) Identification of elf1, a β-spectrin, in early mouse liver development. Intl. J. Dev. Biol. 41, 747–750.
Ohara, O., Ohara, R., Yamakawa, H., Nakajima, D., and Nakayama, M. (1998) Characterization of a new beta-spectrin gene which is predominantly expressed in brain. Brain Res. Mol. Brain Res. 57, 181–192.
Prchal, J. T., Morley, B. J., Yoon, S. H., Coetzer, T. L., Palek, J., Conboy, J. G., and Kan, Y. W. (1987) Isolation and characterization of cDNA clones for human erythrocyte beta-spectrin. Proc. Natl. Acad. Sci. USA 84, 7468–7472.
Riederer, B. M., Zagon, I. S., and Goodman, S. R. (1986) Brain spectrin (240/235) and brain spectrin (240/235E): two distinct spectrin subtypes with different locations within mammalian neural cells. J. Cell Biol. 102, 2088–2097.
Rouleau, G. A., Merel, P., Lutchman, M., Sanson, M., Zucman, J., Marineau, C., et al. (1993) Alteration in a new gene encoding a putative membrane-organizing protein causes neuro-fibromatosis type 2. Nature 363, 515–521.
Sanger, F., Nicklen, S., and Coulson, A. R. (1977) DNA sequencing with chain-terminating inhibitors. Proc. Natl. Acad. Sci. USA 74, 5463–5467.
Scoles, D. R., Huynh, D. P., Morcos, P. A., Coulsell, E. R., Robinson, N. G., Tamanoi, F., and Pulst, S. M. (1998) Neurofibromatosis 2 tumour suppressor schwannomin interacts with betaII-spectrin. Nat. Genet. 18, 354–359.
Shapiro, M. B. and Senapathy, P. (1987) RNA splice junctions of different classes of eukaryotes: sequence statistics and functional implications in gene expression. Nucleic Acids Res. 15, 7155–7174.
Solimena, M., Dirkx, R., Jr., Hermel, J. M., Pleasic-Williams, S., Shapiro, J. A., Caron, L., and Rabin, D. U. (1996) ICA 512, an autoantigen of type I diabetes, is an intrinsic membrane protein of neurosecretory granules. EMBO J. 15, 2102–2114.
Stabach, P. R. and Morrow, J. S. (2000) Identification and characterization of beta V spectrin, a mammalian ortholog of Drosophila beta H spectrin. J. Biol. Chem. 275, 21,385–21,395.
Stankewich, M. C., Tse, W. T., Peters, L. L., Ch’ng, Y., John, K. M., Stabach, P. R., et al. (1998) A widely expressed betaIII spectrin associated with Golgi and cytoplasmic vesicles. Proc. Natl. Acad. Sci. USA 95, 14,158–14,163.
Trofatter, J. A., MacCollin, M. M., Rutter, J. L., Murrell, J. R., Duyao, M. P., Parry, D. M., et al. (1993) A novel moesin-, ezrin-, radixin-like gene is a candidate for the neurofibromatosis 2 tumor suppressor. Cell 75, 826.
Zhang, X. and Bennett, V. (1996) Identification of O-linked N-acetylglucosamine modification of ankyrinG isoforms targeted to nodes of Ranvier. J. Biol. Chem. 271, 31,391–31,398.
Zhou, D., Lambert, S., Malen, P. L., Carpenter, S., Boland, L. M., and Bennett, V. (1998) AnkyrinG is required for clustering of voltage-gated Na channels at axon initial segments and for normal action potential firing. J. Cell Biol. 143, 1295–1304.
Zucman-Rossi, J., Legoix, P., Der Sarkissian, H., Cheret, G., Sor, F., Bernardi, A., et al. (1998) NF2 gene in neurofibromatosis type 2 patients. Hum. Mol. Genet. 7, 2095–2101.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Chen, Y., Yu, P., Lu, D. et al. A novel isoform of beta-spectrin II localizes to cerebellar Purkinje-cell bodies and interacts with neurofibromatosis type 2 gene product schwannomin. J Mol Neurosci 17, 59–70 (2001). https://doi.org/10.1385/JMN:17:1:59
Accepted:
Issue Date:
DOI: https://doi.org/10.1385/JMN:17:1:59