Abstract
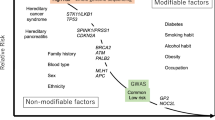
Many factors, including a family history of cancer, have been implicated in the development of pancreatic cancer. Among these factors, germline BRCA2 mutations have been clearly associated with the development of this disease, while mutations in BRCA1 appear to have a limited role. Patients with pancreatic cancer and germline BRCA2 mutations tend to be Ashkenazi Jewish, have a younger than average age of onset, and in many cases, lack family history for breast, ovarian, or pancreatic cancers. In addition, somatic mutations of BRCA2 appear to be rare in tumors of the pancreas. The mechanism by which mutant BRCA2 contributes to development of pancreatic cancers is not well understood. However, it appears that inactivation of several independent functions of BRCA2 including regulation of gene transcription, chromatin remodeling, cell growth, DNA damage repair, and chromosomal instability may provide a pathophysiological basis for the association of BRCA2 mutations and pancreatic cancer.
Similar content being viewed by others
References
Lynch HT, Fursaro L, Lynch JF. Familial pancreatic cancer: a family study. Pancreas 1992;7(5):511–515.
Fernandez E, La Vecchia C, D’Avanzo B, et al. Family history and the risk of liver, gallbladder, and pancreas cancer. Cancer Epidemiol Biomarkers Prev 1994;3(3):209–212.
Falk RT, Pickle LW, Fonthan ET, et al. Life-style risk factors for pancreatic cancer in Louisiana: a case-control study. Am J Epidemiol 1988;128(2):324–336.
Ghadirian P, Bayle P, Simard A, et al. Reported family aggregation of pancreatic cancer within a population-based case-control study in the Francophone community in Montreal, Canada. Int J Pancreatol 1991;10:183–196.
Schenk M, Schwartz AG, O’Neal E, et al. Familial risk of pancreatic cancer. J Natl Cancer Inst 2001;93(8):640–644.
Coughlin SS, Calle EE, Patel AV, et al. Predictors of pancreatic cancer mortality among a large cohort of United States adults. Cancer Causes Control 2000;11(10):915–923.
Goldstein AM, Fraser MC, Struewing JP, et al. Increased risk of pancreatic cancer in melonoma-prone kindreds with P16INK4 mutations. N Engl J Med 1995;333(15):970–974.
Phelan GM, Lancaster JM, Tonin P, et al. Mutation analysis of the BRCA2 gene in 49 site-specific breast cancer families. Nat Genet 1996;13:120–122.
Giadiello FM, Brensiger JD, Termette AC, et al. Very high risk of cancer in familial Peutz-Jeghers syndrome. Gastroenterology 2000;119(6):1447–1453.
Lynch HT, Smyrk T, Lynch JF. Overview of natural history, pathology, molecular genetics and management of HNPCC (Lynch Syndrome). Int J Cancer 1996;69(1):38–42.
Tulinius H, Egilsson V, Olafsdottir GH, et al. Risk of prostate, ovarian, and endometrial cancer among relatives of women with breast cancer. Brit Med J 1992;305(6858):855–857.
The Breast Cancer Linkage Consortium. Cancer risks in BRCA2 mutation carriers. J Natl Cancer Inst 1999;91(15):1310–1316.
Ozcelik H, Schmocker B, DiNicola N, et al. Germline BRCA2 6174delT mutations in Ashkenazi Jewish pancreatic cancer patients. Nat Genet 1997;16:17–18.
Lal G, Liu G, Schmocker B, et al. Inherited predisposition to pancreatic adenocarcinoma: role of family history and germline p16, BRCA1, and BRCA2 mutations. Cancer Res 2000;60:409–416.
Figer A, Irmin L, Geva R, et al. The rate of the 6174delT founder Jewish mutation in BRCA2 in patients with non-colonic gastrointestinal tract tumors in Israel. Brit J Cancer 2001;84(4):478–481.
Brentnall TA. Cancer surveillance of patients from familial pancreatic cancer kindreds. Med Clin North Am 2000;84:707–718.
White K, Held KR, Weber BHF. A BRCA2 germline mutation in familial pancreatic carcinoma. Int J Cancer 2001;91:742–744.
Murphy KM, Brune KA, Griffin C, et al. Evaluation of candidate genes MAP2K4, MADH4, ACVR1B, and BRCA2 in familial pancreatic cancer: deleterious BRCA2 mutations in 17%. Cancer Res 2002;62(13):3789–3793.
Goggins M, Shuttle M, Lu J, et al. Germline BRCA2 gene mutations in patients with apparently sporadic pancreatic carcinomas. Cancer Res 1996;56:5360–5364.
Goggins M, Hruban RH, Kern SE. BRCA2 is inactivated late in the development of pancreatic intraepithelial neoplasia. Am J Pathol 2000;156(5):1767–1771.
Gayther SA, Mangion J, Russell P, et al. Variation of risks of breast and ovarian cancer associated with different germline mutations of BRCA2 gene. Nat Genet 1997;15:103–105.
Thompson D, Easton D. Breast Cancer Linkage Consortium: Variation in cancer risks, by mutation position, in BRCA2 mutation carriers. Am J Hum Genet 2001;68(2):410–419.
Tavtigian SV, Simard J, Rommens J, et al. The complete BRCA2 gene mutations in chromosome 13q-linked kindreds. Nat Genet 1996;12:1–6.
Venkitaraman AR. Cancer Susceptibility and the functions of BRCA1 and BRCA2. Cell 2002;108:171–182.
Marmorstein LY, Ouchi T, Aaronson SA. The BRCA2 gene product functionally interacts with P53 and RAD51. Proc Natl Acad Sci USA 1998;95(23):13,869–13,874.
Milner J, Fuks F, Hughes-Davies L, et al. The BRCA2 activation domain associates with and is phosphorylated by a cellular protein kinase. Oncogene 2000;19(38):4441–4445.
Fuks F, Milner J, Kouzarides T. BRCA2 associates with acetyl transferase activity when bound to P/CAF. Oncogene 1998;17(19):2531–2534.
Marmorstein LY, Kinev AV, Chan GK, et al. A human BRCA2 complex containing a standrard DNA binding component influences cell cycle progression. Cell 2001;104(2):247–257.
Wang SC, Shao R, Pao AY, et al. Inhibition of cancer cell growth by BRCA2. Cancer Res 2002;62(5):1311–1314.
Marston NJ, Richards WJ, Hughes D, et al. Interaction between the product of the breast susceptibility gene BRCA2 and DSS1, a protein conserved from yeast to mammals. Mol Cell Biol 1999;19(7):4533–4542.
Liu J, Yuan Y, Huan J, et al. Inhibition of breast and breast cell growth by BCCIPalpha, an evolutionary conserved nuclear protein that interacts with BRCA2. Oncogene 2001;20(3):336–345.
Kraakman-van der Zwet M, Overkamp WJ, Van Large RE, et al. BRCA2 (XRCC11 deficiency results in radioresistant DNA synthesis and a higher frequency of spontaneous deletions. Mol Cell Biol 2002;22(2):669–679.
Wong AK, Pero R, Ormonde PA, et al. RAD51 interacts with the evolutionary conserved BRC motifs in the human breast cancer susceptibility gene BRCA2. J Biol Chem 1997;272:31,941–31,944.
Davies AA, Masson J, Mcllwraith MJ, et al. Role of BRCA2 in control of the Rad51 recombination and DNA repair protein. Mol Cell 2001;7:273–282.
Moynahan ME, Pierce AJ, Jasin M. BRCA2 is required for homology directed repair of chromosomal breaks. Mol Cell 2001;7:263–272.
Chen CF, Chen PL, Zhong Q, et al. Expression of BRC repeats in breast cancer cells disrupts the BRCA2-Rad51 complex and leads to radiation sensitivity and loss of G(2)/M checkpoint control. J Biol Chem 1999;274(46):32,931–32,935.
Yang H, Jeffrey PD, Miller J, et al, BRCA2 function in DNA binding and recombination from a BRCA2-DSS1-ssDNA structure. Science 2002;297(5588):1837–1848.
Yu VP, Koehler M, Steinlein C, et al. Gross chromsomal rearrangements and genetic exchange between nonhomologous chromosomes following BRCA2 inactivation. Genes Dev 2000;14:1400–1406.
Abbott DW, Freeman ML, Holt J. Double-stranded break repair deficiency and radiation sensitivity in BRCA2 mutant cancer cells. J Natl Cancer Inst 1998;90:6–13.
Shyng-Shiou F, Sou-Ying L, Gang C, et al. BRCA2 is required for ionizing radiation-induced assembly of Rad51 complex in-vivo. Cancer Res 1999;59:3547–3551.
Huichen W, Zhao-Chong Z, Tu-Auh B, et al. Nonhomologous end-joining of ionizing radiation-induced DNA double-stranded breaks in human tumor cells deficient in BRCA1 and BRCA2. Cancer Res 2001;61:270–277.
Thompson LH, Schild D. The contribution of homologous recombination in preserving genome integrity in mammalian cells. Biochimie (Paris) 1999;81:87–105.
Chen PL, Chen CF, Chen Y, et al. The BRC repeats in BRCA2 are critical for Rad51 binding and resistance to methylmethane sulfonate treatment. Proc Natl Acad Sci USA 1998;95(9):5287–5292.
Bogliolo M, Taylor RM, Caldecoltt KW, et al. Reduced ligation during DNA base excision repair supported by BRCA2 mutant cells. Oncogene 2000;19:5781–5787.
Le Page F, Randrianarison V, Marot D, et al. BRCA1 and BRCA2 are necessary for the transcription-coupled repair of the oxidative 8-oxaguanine in human cells. Cancer Res 2000;60(19):5548–5552.
Tutt A, Gabriel A, Bertwistle D, et al. Absence of BRCA2 causes genome instability by chromosome breakage and loss associated with centrosome amplification. Curr Biol 1999;9(19):1107–1110.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Naderi, A., Couch, F.J. BRCA2 and pancreatic cancer. Int J Gastrointest Canc 31, 99–106 (2002). https://doi.org/10.1385/IJGC:31:1-3:99
Issue Date:
DOI: https://doi.org/10.1385/IJGC:31:1-3:99