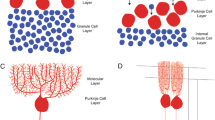
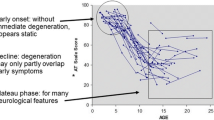
Abstract
Appropriate cellular signaling responses to DNA damage and the ability to repair DNA are fundamental processes that are required for organismal survival. Ataxia-telangiectasia (A-T) is a rare neurodegenerative disease that results from defective DNA damage signaling. Understanding the molecular basis of A-T has provided many critical insights into the cellular response to DNA double-strand breaks (DSBs). A-T is a syndrome that shows pronounced neurodegeneration of the nervous system coincident with immune deficiency, radiosensitivity, and cancer proneness. A-T results from inactivation of the A-T mutated (ATM) kinase, a critical protein kinase that regulates the response to DNA-DSBs by selective phosphorylation of a variety of substrates. Therefore, understanding the ATM signaling program has important biological ramifications for nervous system homeostasis. Underscoring the importance of the DNA-DSBs response in the nervous system are other diseases related to A-T that also result from defects in this signaling pathway. In particular, defects in the DNA damage sensor, the Mre11-RAD50-NBS1 complex, also lead to syndromes with neurological deficits and overlapping phenotypes to A-T. Collectively, these diseases high light the critical importance of appropriate responses to DNA-DSBs to maintain homeostasis in the nervous system.
Similar content being viewed by others
References
Abner C. W. and McKinnon P. J. (2004) The DNA double-strand break response in the nervous system. DNA Repair (Amst.) 3(8–9), 1141–1147.
Allen D. M., van Praag H., Ray J., et al. (2001) Ataxia telangiectasia mutated is essential during adult neurogenesis. Genes Dev. 15(5), 554–566.
Angele S., Romestaing P., Moullan N., et al. (2003) ATM haplotypes and cellular response to DNA damage: association with breast cancer risk and clinical radiosensitivity. Cancer Res. 63(24), 8717–8725.
Bakhshi S., Cerosaletti K. M., Concannon P., et al. (2003) Medulloblastoma with adverse reaction to radiation therapy in nijmegen breakage syndrome. J. Pediatr. Hematol. Oncol. 25(3), 248–251.
Bakkenist C. J. and Kastan M. B. (2003) DNA damage activates ATM through intermolecular autophosphorylation and dimer dissociation. Nature 421(6922), 499–506.
Baptista C. A., Hatten M. E., Blazeski R., and Mason C. A. (1994) Cell-cell interactions influence survival and differentiation of purified Purkinje cells in vitro. Neuron 12(2), 243–260.
Barbieri F., Santoro L., Crisci C., Massini R., Russo E., and Campanella G. (1986) Is the sensory neuropathy in ataxia-telangiectasia distinguishable from that in Friedreich's ataxia? Morphometric and ultrastructural study of the sural nerve in a case of Louis Bar syndrome. Acta Neuropathol. (Berl.) 69(3–4), 213–219.
Barlow C., Hirotsune S., Paylor R., et al. (1996) Atm-deficient mice: a paradigm of ataxia telangiectasia. Cell 86(1), 159–171.
Barnes D. E., Stamp G., Rosewell I., Denzel A., and Lindahl T. (1998) Targeted disruption of the gene encoding DNA ligase IV leads to lethality in embryonic mice. Curr. Biol. 8(25), 1395–1398.
Bassing C. H. and Alt F. W. (2004) The cellular response to general and programmed DNA double strand breaks. DNA Repair (Amst.) 3(8–9), 781–796.
Bekiesinska-Figatowska M., Chrzanowska K. H., Sikorska J., et al. (2000) Cranial MRI in the Nijmegen breakage syndrome. Neuroradiology 42(1), 43–47.
Borges H. L., Chao C., Xu Y., Linden R., and Wang J. Y. (2004) Radiation-induced apoptosis in developing mouse retina exhibits dose-dependent requirement for ATM phosphorylation of p53. Cell Death Differ. 11(5) 494–502.
Borghesani P. R., Alt F. W., Bottaro A., et al. (2000) Abnormal development of Purkinje cells and lymphocytes in Atm mutant mice. Proc. Natl. Acad. Sci. USA 97(7), 3336–3341.
Burma S. and Chen D. J. (2004) Role of DNA-PK in the cellular response to DNA double-strand breaks. DNA Repair (Amst.) 3(8–9), 909–918.
Caldecott K. W. (2004) DNA single-strand breaks and neurodegeneration. DNA Repair (Amst.) 3(8–9), 875–882.
Carlomagno F., Chang-Claude J., Dunning A. M., and Ponder B. A. (1999) Determination of the frequency of the common 657Del5 Nijmegen breakage syndrome mutation in the German population: no association with risk of breast cancer. Genes Chromosomes Cancer 25(4), 393–395.
Carney J. P., Maser R. S., Olivares H., et al. (1998) The hMre11/hRad50 protein complex and Nijmegen breakage syndrome: linkage of double-strand break repair to the cellular DNA damage response. Cell 93(3), 477–486.
Carson C. T., Schwartz R. A., Stracker T. H., Lilley C. E., Lee D. V., and Weitzman M. D. (2003) The Mrell complex is required for ATM activation and the G(2)/M checkpoint. EMBO J. 22(24), 6610–6620.
Casorelli I., Offman J., Mele L., et al. (2003) Drug treatment in the development of mismatch repair defective acute leukemia and myelodysplastic syndrome. DNA Repair (Amst.) 2(5), 547–559.
Cerosaletti K. M., Morrison V. A., Sabath D. E., Willerford D. M., and Concannon P. (2002) Mutations and molecular variants of the NBS1 gene in non-Hodgkin lymphoma. Genes Chromosomes Cancer 35(3), 282–286.
Chen L., Trujillo K., Ramos W., Sung P., and Tomkinson A. E. (2001) Promotion of Dnl4-catalyzed DNA end-joining by the Rad50/Mre11/Xrs2 and Hdf1/Hdf2 complexes. Mol. Cell 8(5), 1105–1115.
Chrzanowska K. H., (1996) [Microcephaly with chromosomal instability and immunodeficiency—Nijmegen syndrome]. Pediatr Pol 71(3), 223–234.
Chun H. H. and Gatti R. A. (2004) Ataxia-telangiectasia, an evolving phenotype. DNA Repair (Amst.) 3(8–9), 1187–1196.
Ciemins J. J. and Horowitz A. L. (2000) Abnormal white matter signal in ataxia telangiectasia. AJNR Am. J. Neuroradiol. 21(8), 1483–1485.
Cleaver J. E. (2005) Cancer in xeroderma pigmentosum and related disorders of DNA repair. Nat. Rev. Cancer 5(7), 564–573.
Cruz-Martinez A., Barrio M., Gutierrez A. M., and Lopez E. (1977) Abnormalities in sensory and mixed evoked potentials in ataziat elangiectasia. J. Neurol. Neurosurg. Psych. 40(1077), 44–49.
Cybulski C., Gorski B., Debniak T. et al. (2004) NBS1 is a prostate cancer susceptibility gene. Cancer Res. 64(4), 1215–1219.
D'Amours D. and Jackson S. P. (2002) The Mre11 complex: at the crossroads of dna repair and checkpoint signalling. Nat. Rev. Mol. Cell Biol. 3(5), 317–327.
Delia D., Piane M., Buscemi G., et al. (2004) MRE11 mutations and impaired ATM-dependent responses in an Italian family with ataxia-telangiectasia-like disorder. Hum. Mol. Genet. 13(18), 2155–2163.
Difilippantonio S., Celeste A., Fernandez-Capetillo O., et al. (2005) Role of Nbs1 in the activation of the Atm kinase revealed in humanized mouse models. Nat. Cell Biol. 7(7), 675–685.
Digweed M. and Sperling K. (2004) Nijmegen breakage syndrome: clinical manifestation of defective response to DNA double-strand breaks. DNA Repair (Amst.) 3(8–9), 1207–1217.
Distel L., Neubauer S., Varon R., Holter W., and Grabenbauer G. (2003) Fatal toxicity following radio- and chemotherapy of medulloblastoma in a child with unrecognized Nijmegen breakage syndrome. Med. Pediatr. Oncol. 41(1), 44–48.
Dumon-Jones V., Frappart P. O., Tong W. M., et al. (2003) Nbn heterozygosity renders mice susceptible to tumor formation and ionizing radiation-induced tumorigenesis. Cancer Res. 63(21), 7263–7269.
Elson A., Wang Y., Daugherty C. J., et al. (1996) Pleiotropic defects inataxia-telangiectasia protein-deficient mice. Proc. Natl. Acad. Sci. USA 93(23), 13,084–13,089.
Falck J., Coates J., and Jackson S. P. (2005) Conserved modes of recruitment of ATM ATR and DNA-PKcs to sites of DNA damage. Nature 434(7033), 605–611.
Farina L., Uggetti C., Ottolini A., et al. (1994) Ataxiatelangiectasia: MR and CT findings. J. Comput. Assist. Tomogr. 18(5), 724–727.
Farr A. K., Shalev B., Crawford T. O., Lederman H. M., Winkelstein J. A., and Repka M. X. (2002) Ocular manifestations of ataxia-telangiectasia. Am. J. Ophthalmol. 134(6), 891–896.
Fernandes N., Sun Y., Chen S., et al. (2005) DNA damage-induced association of ATM with its target proteins requires a protein interaction domain in the N terminus of ATM. J. Biol. Chem. 280(15), 15,158–15,164.
Fernet M., Gribaa M., Salih M. A., Seidahmed M. Z., Hall J., and Koenig M. (2005) Identification and functional consequences of a novel MRE11 mutation affecting 10 Saudi Arabian patients with the ataxia telangiectasia-like disorder. Hum. Mol. Genet. 14(2), 307–318.
Frank K. M., Sharpless N. E., Gao Y., et al. (2000) DNA Ligase IV Deficiency in Mice Leads to Defective Neurogenesis and Embryonic Lethalitivia the p53 Pathway. Mol. Cell 5, 993–1002.
Frappart P. O., Tong W. M., Demuth I., et al. (2005) An essential function for NBS1 in the prevention of ataxia and cerebellar defects. Nat. Med. 11(6), 538–544.
Fukuda T., Sumiyoshi T., Takahashi M. et al. (2001) Alterations of the double-strand break repair gene MRE11 in cancer. Cancer Res. 61(1), 23–26.
Gao Y., Ferguson D. O., Xie W., et al. (2000) Interplay of p53 and DNA-repair protein XRCC4 in tumorigenesis, genomic stability and development. Nature 404(6780) 897–900.
Gao Y., Sun Y., Frank K. M., et al. (1998) A critical role for DNA end-joining proteins in both lymphogenesis and neurogenesis. Cell 95(7), 891–902.
Gatti R. A. and Vinters H. V. (1985) Cerebellar Pathology in Ataxia-Telangiectasia: The significance of Basket Cells, in Kroc Foundation Series, Gatti R. A. and Swift M., eds., Alan R. Liss, Inc., New York, pp. 225–232.
Gatti R. A., Becker-Catania S., Chun H. H., et al. (2001) The pathogenesis of ataxia-telangiectasia. Learning from a Rosetta Stone. Clin. Rev. Allergy Immunol. 20(1), 87–108.
Giannini G., Ristori E., Cerignoli F., et al., (2002) Human MRE11 is inactivated in mismatch repair-deficient cancers. EMBO Rep. 3(3), 248–254.
Goldowitz D. and Hamre K. (1998) The cells and molecules that make a cerebellum. Trends Neurosci. 21(9), 375–382.
Gosink E. C., Chong M. J., and McKinnon P. J. (1999) Ataxia telangiectasia mutated deficiency affects astrocyte growth but not radiosensitivity. Cancer Res. 59(20), 5294–5298.
Guidos C. J., Williams C. J., Grandal I., Knowles G., Huang M. T., and Danska J. S. (1996) V(D)J recombination activates a p53-dependent DNA damage check point in scid lymphocyte precursors. Genes Dev. 10(16), 2038–2054.
Gumy-Pause F., Wacker P., and Sappino A. P. (2004) ATM gene and lymphoid malignancies. Leukemia 18(2), 238–242.
Haber J. E. (2000) Partners and pathways repairing a double-strand break. Trends Genet. 16(6), 259–264.
Hama S., Matsuura S., Tauchi H., et al. (2000) Absence of mutations in the NBS1 gene in B-cell malignant lymphoma patients. Anticancer Res. 20(3B), 1897–1900.
Hatten M. E. and Heintz N. (1995) Mechanisms of Neural Patterning and Specification in the Developing Cerebellum. Annu. Rev. Neurosci. 18, 385–408.
Herzog K. H., Chong M. J., Kapsetaki M., Morgan J. I., and McKinnon P. J. (1998) Requirement for Atm in ionizing radiation-induced cell death in the developing central nervous system. Science 280(5366), 1089–1091.
Hoeijmakers J. H. (2001) Genome maintenance mechanisms for preventing cancer. Nature 411(6835), 366–374.
Hopfner K. P., Craig L., Moncalian G., et al. (2002) The Rad50 zinc-hook is a structure joining Mre11 complexes in DNA recombination and repair. Nature 418(6897) 562–566.
Kang J., Bronson R. T., and Xu Y. (2002) Targeted disruption of NBS1 reveals its roles in mouse development and DNA repair. EMBO J. 21(6), 1447–1455.
Kastan M. B., and Bartek J. (2004) Cell-cycle check-points and cancer. Nature 432(7015), 316–323.
Kitagawa R., Bakkenist C. J., McKinnon P. J., and Kastan M. B. (2004) Phosphorylation of SMC1 is a critical downstream event in the ATM-NBS1-BRCA1 pathway. Genes Dev. 18, 1423–1438.
Kwast O. and Ignatowicz R. (1990) Progressive peripheral neuron degeneration in ataxia-telangiectasia: an electrophysiological study in children. Dev. Med. Child Neurol. 32(9), 800–807.
Lakin N. D., Weber P., Stankovic T., Rottinghaus S. T., Taylor A. M., and Jackson S. P. (1996) Analysis of the ATM protein in wild-type and ataxia telangiectasia cells. Oncogene 13(12), 2707–2716.
Lee Y. and McKinnon P. J. (2002) DNA ligase IV suppresses medulloblastoma formation. Cancer Res. 62(22), 6395–6399.
Lee J. H. and Paull T. T. (2004) Direct activation of the ATM protein kinase by the Mre11/Rad50/Nbs1 complex. Science 304(5667), 93–96.
Lee Y., Chong M. J., and McKinnon P. J. (2001) Ataxia telangiectasia mutated-dependent apoptosis after genotoxic stress in the developing nervous system is determined by cellular differentiation status. J. Neurosci. 21(17), 6687–6693.
Lee Y., Barnes D. E., Lindahl T., and McKinnon P. J. (2000) Defective neurogenesis resulting from DNA ligase IV deficiency requires Atm. Genes Dev. 14(20), 2576–2580.
Lees-Miller S. P. and Meek K. (2003) Repair of DNA double strand breaks by nonhomologous end joining. Biochimie 85(11), 1161–1173.
Lefton-Greif M. A., Crawford T. O., Winkelstein J. A., et al. (2000) Oropharyngeal dysphagia and aspiration in patients with ataxia-telangiectasia. J. Pediatr. 136(2), 225–231.
Lelli S., Trevisan C., and Negrin P. (1995) Ataxiateleangectasia: neurophysiological studies in 8 patients. Electromyogr. Clin. Neurophysiol. 35(5), 311–315.
Lewis R. F. and Crawford T. O. (2002) Slow target-directed eye movements in ataxia-telangiectasia. Invest. Ophthalmol. Vis. Sci. 43(3), 686–691.
Lieber M. R., Ma Y., Pannicke U., and Schwarz K. (2003) Mechanism and regulation of human nonhomologous DNA end-joining. Nat. Rev. Mol. Cell Biol. 4(9), 712–720.
Lim D. S., Vogel H., Willerford D. M., Sands A. T., Platt K. A., and Hasty P. (2000) Analysis of ku80-mutant mice and cells with deficient levels of p53. Mol. Cell Biol. 20(11), 3772–3780.
Lin S. Y., Rai R., L. K., Xu Z. X., and Elledge S. J. (2005) BRIT1/MCPH1 is a DNA damage responsive protein that regulates the Brca1-Chk1 pathway, implicating checkpoint dysfunction in microcephaly. Proc. Natl. Acad. Sci. USA 102(42), 15,105–15,109.
Lukas C., Falck J., Bartkova J., Bartek J., and Lukas J. (2003) Distinct spatiotemporal dynamics of mammalian checkpoint regulators induced by DNA damage. Nat. Cell Biol. 5(3), 255–260.
Malandrini A., Guazzi G. C., Alessandrini C., and Federico A. (1990) Peripheral nerve involvement in ataxia telangiectasia: histological and ultrastructural studies of peroneal nervebiopsy in two cases. Clin. Neuropathol. 9(3), 109–114.
Maser R. S., Zinkel R., and Petrini J. H. (2001) An alternative mode of translation permits production of a variant NBS1 protein from the common Niimegen breakage syndrome allele. Nat. Genet. 27(4), 417–421.
McConnell M. J., Kaushal D., Yang A. H., et al. (2004) Failed clearance of aneuploid embryonic neural progenitor cells leads to excess aneuploidy in the Atm-deficient but not the Trp53-deficient adult cerebral cortex. J. Neurosci. 24(37) 8090–8096.
McKinnon P. J. (2004) ATM and ataxia telangiectasia. EMBO Rep. 5(8), 772–776.
Moreno-Herrero F., de Jager M., Dekker N. H., Kanaar R., Wyman C., and Dekker C. (2005) Mesoscale conformational changes in the DNA-repair complex Rad50/Mre11/Nbs1 upon binding DNA. Nature 437(7057), 440–443.
Motoyama N. and Naka K. (2004) DNA damage tumor suppressor genes and genomic instability. Curr. Opin. Genet. Dev. 14(1), 11–16.
Nacht M., Strasser A., Chan Y. R., et al. (1996) Mutations in the p53 and SCID genes cooperate in tumorigenesis. Genes Dev. 10(16), 2055–2066.
Nick McElhinny S. A., Snowden C. M., McCarville J., and Ramsden D. A. (2000) Ku recruits the XRCC4-ligase IV complex to DNA ends. Mol. Cell Biol. 20(9), 2996–3003.
Nowak-Wegrzyn A., Crawford T. O., Winkelstein J. A., Carson K. A., and Lederman H. M. (2004) Immunodeficiency and infections in ataxia-telangiectasia. J. Pediatr. 144(4), 505–511.
O'Driscoll M., Cerosaletti K. M., Girard P. M., et al. (2001) DNA ligase IV mutations identified in patients exhibiting developmental delay and immunodeficiency. Mol. Cell 8(6), 1175–1185.
Paula-Barbosa M. M., Ruela C., Tavares M. A., Pontes C., Saraiva A., and Cruz C. (1983) Cerebellar Cortex Ultrastructure in Ataxia-telangiectasia. Ann. Neurol. 13, 297–302.
Perry J. and Kleckner N. (2003) The ATRs ATMs, and TORs are giant HEAT repeat proteins. Cell 112(2), 151–155.
Petrini J. H. and Stracker T. H. (2003) The cellular response to DNA double-strand breaks: defining the sensors and mediators. Trends Cell Biol. 13(9), 458–462.
Pitts S. A., Kullar H. S., Stankovic T., et al. (2001) hMRE11: genomic structure and a null mutation identified in a transcript protected from nonsensemediated mRNA decay. Hum. Mol. Genet. 10(11), 1155–1162.
Resnick I. B., Kondratenko I., Pashanov E., et al. (2003) 657 del5 mutation in the gene for Nijmegen breakage syndrome (NBS1) in a cohort of Russian children with lymphoid tissue malignancies and controls. Am. J. Med. Genet. 120A(2), 174–179.
Saar K., Chrzanowska K. H., Stumm M., et al. (1997) The gene for the ataxia-telangiectasia variant, Nijmegen breakage syndrome, maps to a 1-cM interval on chromosome 8q21. Am. J. Hum. Genet. 60(3), 605–610.
Sardanelli F., Parodi R., Ottonello C. et al. (1995) Cranial MRI in ataxia-telangiectasia. Neuroradiology 37, 77–82.
Sedelnikova O. A., Pilch D. R., Redon C., and Bonner W. M. (2003) Histone H2AX in DNA damage and repair. Cancer Biol. Ther. 2(3), 233–235.
Sedgwick R. P. and Boder E. (1991) Ataxia-Telangiectasia, in Handbook of Clinical Neurology, Vinken P., Bruyn G., and Klawans H., eds., Elsevier, New York, pp. 347–423.
Sekiguchi J., Ferguson D. O., Chen H. T., et al. (2001) Genetic interactions between ATM and the nonhomologous end-joining factors in genomic stability and development. Proc. Natl. Acad. Sci. USA 98(6), 3243–3248.
Shiloh Y. (1997) Ataxia-telangiectasia and the Nijmegen breakage syndrome: related disorders but genes apart. Annu Rev. Genet. 31, 635–662.
Shiloh Y. (2003) ATM and related protein kinases: safeguarding genome integrity. Nat. Rev. Cancer 3(3), 155–168.
Soares H. D., Morgan J. I., and McKinnon P. J. (1998) Atm expression patterns suggest a contribution from the peripheral nervous system to the phenotype of ataxia-telangiectasia. Neuroscience 86(4), 1045–1054.
Spring K., Ahangari F., Scott S. P., et al. (2002) Mice heterozygous for mutation in Atm, the gene involved in ataxia-telangiectasia, have heightened susceptibility to cancer. Nat. Genet. 32(1), 185–190.
Stanulla M., Stumm M., Dieckvoss B. O., et al. (2000) No evidence for a major role of heterozygous deletion 657 del5 within the NBS1 gene in the pathogenesis of non-Hodgkin's lymphoma of childhood and adolescence. Br. J. Haematol. 109(1), 117–120.
Stewart G. S., Last J. I., Stankovic T., et al. (2001) Residual ataxia telangiectasia mutated protein function in cells from ataxia telangiectasia patients, with 5762ins137 and 7271T→G mutations, showing a less severe phenotype. J. Biol. Chem. 276(32), 30,133–30,141.
Stewart G. S., Maser R. S., Stankovic T., et al. (1999) The DNA double-strand break repair gene hMRE11 is mutated in individuals with anataxia-telangiectasia-like disorder. Cell 99(6), 577–587.
Stumm M., von Ruskowsky A., Siebert R., et al. (2001) No evidence for deletions of the NBS1 gene in lymphomas. Cancer Genet. Cytogenet. 126(1), 60–62.
Sun Y., Jiang X., Chen S., Fernandes N., and Price B. D. (2005) Arole for the Tip60 histone acetyltransferase in the acetylation and activation of ATM. Proc. Natl. Acad. Sci. USA 102(37), 13,182–13,187.
Tauchi H., Kobayashi J., Morishima K., et al. (2002) Nbs1 is essential for DNA repair by homologous recombination in higher vertebrate cells. Nature 420(6911), 93–98.
Tavani F., Zimmerman R. A., Berry G. T., Sullivan K., Gatti R., and Bingham P. (2003) Ataxia-telangiectasia: the pattern of cerebellar atrophy on MRI. Neuroradiology 45(5), 315–319.
Taylor A. M. and Byrd P. J. (2005) Molecular pathology of ataxia telangiectasia. J. Clin. Pathol. 58(10), 1009–1015.
Taylor A. M., Groom A., and Byrd P. J. (2004) Ataxia-telangiectasia-like disorder (ATLD)-its clinical presentation and molecular basis. DNA Repair (Amst.) 3(8–9) 1219–1225.
Taylor G. M., O'Brien H. P., Greaves M. F., Ravetto P. F., and Eden O. B. (2003) Correspondence re: Varon R. et al., Mutations in the Nijmegen breakage syndrome gene (NBS1) in childhood acute lymphoblastic leukemia. Cancer/Res. 63(19), 6563–6564; author reply 6565.
Theunissen J. W., Kaplan M. I., Hunt P. A., et al. (2003) Checkpoint failure and chromosomal instability without lymphomagenesis in Mre11 (ATLD1/ATLD1) mice. Mol. Cell 12(6), 1511–1523.
Thorstenson Y. R., Roxas A., Kroiss R., et al. (2003) Contributions of ATM mutations to familial breast and ovarian cancer. Cancer Res. 63(12), 3325–3333.
Uziel T., Lerenthal Y., Moyal L., Andegeko Y., Mittelman L., and Shiloh Y. (2003) Requirement of the MRN complex for ATM activation by DNA damage. EMBO J. 22(20), 5612–5621.
Valerie K. and Povirk L. F. (2003) Regulation and mechanisms of mammalian double-strand break repair. Oncogene 22(37), 5792–5812.
van den Bosch M., Bree R. T., and Lowndes N. F. (2003) The MRN complex: coordinating and mediating the response to broken chromosomes. EMBO Rep. 4(9), 844–849.
van der Burgt I., Chrzanowska K. H., Smeets D., and Weemaes C. (1996) Nijmegen breakage syndrome. J. Med. Genet. 33(2), 153–156.
van Gent D. C., Hoeijmakers J. H., and Kanaar R. (2001) Chromosomal stability and the DNA double-stranded break connection. Nat. Rev. Genet. 2(3), 196–206.
Varon R., Gosse-Brun S., Bignon Y. J., Sperling K., and Uhrhammer N. (2002) Nijmegen breakage syndrome gene (NBS1) is not the tumor suppressor gene at 8q21.3 involved in colorectal carcinoma. Oncol. Rep. 9(4), 709–711.
Varon R., Seemanova E., Chrzanowska K., et al. (2000) Clinical ascertainment of Nijmegen breakage syndrome (NBS) and prevalence of the major mutation, 657del5, in three Slav populations. Eur. J. Hum. Genet. 8(11), 900–902.
Vinters H. V., Gatti R. A., and Rakic P. (1985) Sequence of Cellular Events in Cerebellar Ontogeny Relevant to Expression of Neuronal Abnormalities in Ataxia-Telangiectasia, in Ataxia-telangiectasia: Genetics, Neuropathology, and Immunology of a degenerative Disease of Childhood, Gatti R. A. and Swift M., eds., Alan R. Liss, Inc., New York, pp. 233–235.
Ward I. and Chen J. (2004) Early events in the DNA damage response. Curr. Top. Dev. Biol. 63, 1–35.
Weemaes C. M., Smeets D. F., Horstink M., Haraldsson A., and Bakkeren J. A. (1993) Variants of Nijmegen breakage syndrome and ataxia telangiectasia. Immunodeficiency 4(1–4), 109–111.
Weemaes C. M., Hustinx T. W., Scheres J. M., van Munster P. J., Bakkeren J. A., and Taalman R. D. (1981) A new chromosomal instability disorder: the Nijmegen breakage syndrome. Acta Paediatr. Scand. 70(4), 557–564.
West S. C. (2003) Molecular views of recombination proteins and their control. Nat. Rev. Mol. Cell Biol. 4(6), 435–445.
Williams R. S. and Tainer J. A. (2005) A nanomachine for making ends meet: MRN is a flexing scaffold for the repair of DNA double-strand breaks. Mol. Cell 19(6), 724–726.
Williams B. R., Mirzoeva O. K., Morgan W. F., Lin J., Dunnick W., and Petrini J. H. (2002) A murine model of nijmegen breakage syndrome. Curr. Biol. 12(8), 648–653.
Wiltzius J. J., Hohl M., Fleming J. C., and Petrini J. H. (2005) The Rad50 hook domain is a critical determinant of Mre11 complex functions. Nat. Struct. Mol. Biol. 12(5), 403–407.
Woods C. G. (2004) Human microcephaly. Curr. Opin. Neurobiol. 14(1), 112–117.
Xiao Y. and Weaver D. T. (1997) Conditional gene targeted deletion by Cre recombinase demonstrates the requirement for the double-strand break repair Mre11 protein in murine embryonic stem cells. Nucleic Acids Res. 25(15), 2985–2991.
Xu X., Lee J., and Stern D. F. (2004) Microcephalin is a DNA damage response protein involved in regulation of CHK1 and BRCA1. J. Biol. Chem. 279(33), 34,091–34,094.
Xu Y., Ashley T., Brainerd E. E., Bronson R. T., Meyn M. S., and Baltimore D. (1996) Targeted disruption of ATM leads to growth retardation, chromosomal fragmentation during meiosis, immune defects, and thymic lymphoma. Genes Dev. 10, 2411–2422.
You Z., Chahwan C., Bailis J., Hunter T., and Russell P. (2005) ATM activation and its recruitment to damaged DNA require binding to the C terminus of Nbs1. Mol. Cell Biol. 25(13), 5363–5379.
Zhu J., Petersen S., Tessarollo L., and Nussenzweig A. (2001) Targeted disruption of the Nijmegen breakage syndrome gene NBS1 leads to early embryonic lethality in mice. Curr. Biol. 11(2), 105–109.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Frappart, PO., McKinnon, P.J. Ataxia-telangiectasia and related diseases. Neuromol Med 8, 495–511 (2006). https://doi.org/10.1385/NMM:8:4:495
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1385/NMM:8:4:495