Abstract
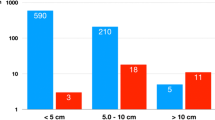
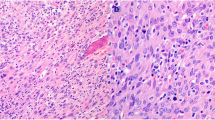
We analyzed a series of adrenocortical neoplasms to compare the clinicopathologic features and the expression of insulin-like growth factor-2 (IGF-2) in adrenocortical adenomas and carcinomas. IGF-2 is a growth factor commonly expressed in many tumors including adrenal cortical and medullary neoplasms. Formalin-fixed paraffin-embedded tissues from 64 adrenocortical adenomas and 67 adrenocortical carcinomas were analyzed. The carcinomas were histologically graded from 1 to 4 based on mitotic activity and necrosis. Tumor weight, size, and follow-up information were obtained by chart review. Expression of IGF-2 was detected by immunohistochemistry with the avidin-biotin-peroxidase complex method and a monoclonal antibody against IGF-2. Adrenocortical carcinomas were larger (mean: 13.1 cm, 787 g) than adenomas (mean: 4.2 cm, 52 g) (p<0.001). In patients with adrenocortical carcinomas, high tumor grade (3 or 4) (p=0.01) was associated with decreased survival. Expression of IGF-2 was higher in adrenocortical carcinomas than in adenomas (p<0.001). These results show that tumor size and weight along with expression of IGF-2 protein are useful features to assist in distinguishing between adrenocortical adenomas and carcinomas, and that high tumor grade is a predictor of survival in adrenocortical carcinomas. However, single immunohistochemical markers such as IGF-2 or single histopathologic features cannot by themselves separate adrenocortical adenomas from carcinomas, and a combination of clinical, gross, and microscopic features are needed to establish the diagnosis in difficult cases.
Similar content being viewed by others
References
Lack EE. Tumors of the adrenal gland and extra-adrenal paraganglia. In: Atlas of tumor pathology. 3rd series Armed Forces Institute of Pathology, Washington, DC. Fascicle 19, 1997.
Hough AJ, Hollifield JW, Page DL, Harmann WH. Prognostic factors in adrenal cortical tumors: a mathematical analysis of clinical and morphologtic data. Am J Clin Pathol 72:390–399, 1979.
Weiss LW. Comparative histologic study of 43 metastasizing and non-metastasizing adrenocortical tumors. Am J Surg Pathol 8:163–169, 1984.
Weiss LM, Medeiros LJ, Vickery AL. Pathologic features of prognostic significance in adrenocortical carcinoma. Am J Surg Pathol 13:202–206, 1989.
Van Slooten HV, Schaberg A, Smeenk D, Moolenaar AJ. Morphologic characteristics of benign and malignant adrenocortical tumors. Cancer 55:766–773, 1985.
De Chiara T, Robertson E, Efstratiadis A. Parental imprinting of the mouse insulin-like growth factor II gene. Cell 64:849–859, 1991.
Rainier S, Johnson L, Dobry C, Ping A, Grundy P, Feinberg A. Relaxation of imprinted genes in human cancer. Nature 362:747–749, 1993.
Voutilainen R. Adrenocortical cells are the site of secretion and action of insulin-like growth factors and TNF-alpha. Horm Metab Res 30:432–435, 1998.
Voutilainen R, Miller WL. Developmental and hormonal regulation of mRNAs for insulin-like growth factor II and steroidogenic enzymes in human fetal adrenals and gonads. DNA 7:9–15, 1988.
Morison I, Becroft D, Taniguchi, Woods C, Reeve A. Somatic overgrowth associated with overexpression of insulin-like growth II. Nat Med 2:311–316, 1996.
Boulle N, Logie A, Gicquel C, Perin L, Le Bouc Y. Increased levels of insulin-like growth factor II (IGF-II) and IGF-binding protein-2 are associated with malignancy in sporadic adrenocortical tumors. J Clin Endocrinol Metab 83:1713–1720, 1998.
Chang HJ, Batts KP, Lloyd RV, Sebo J, Thompson B, Lohse CM, Pankrat SV. Prognostic significance of p27, Ki-67, and topoisomerase IIα expression in clinically non-functioning pancreatic endocrine tumors. Endocr Pathol 11:229–241, 2000.
Voutilainen, Ilvesmaki V, Ariel I, Rachmilewitz J, DeGroot N, Hochberg A. Parallel regulation of parentally imprinted H19 and insulin-like growth factor-II genes in cultured human fetal adrenal cells. Endocrinology 134(5):2051–2056, 1994.
Goshen, Rachmilewitz J, Schneider T, de Groot N, Ariel I, Patli Z, Hochberg A. The expression of the human H19 and IGF-2 genes during human embryogenesis and placental development. Mol eprod Dev 34:374–379, 1993.
Wiedemann HR. Tumours and hemihypertrophy associated with Wiedemann-Beckwith syndrome. Eur J Pediatr 141:129–133, 1983.
Gicquel C, Affin-Sanson ML, Gaston V, Bertagna X, Plouin PF, Schlumberger M, Louvel A, Luton JP, Le Bouc Y. Structural and functional abnormalities at 11p15 are associated with the malignant phenotype in sporadic adrenocortical tumors: study on a series of 82 tumors. J Clin Endocrinol Metab 82:2559–2565, 1997.
Meideiros LA, Weiss LM. New Developments in the pathologic diagnosis of adrenal cortical neoplasms: a review. Am J Clin Pathol 97:73–83, 1992.
Goldblum JR, Shannon R, Kaldjian EP, Thiny M, Davenport R, Thompson N, Lloyd RV. Immunohistochemical assessment of proliferative activity in adrenocortical neoplasm. Mod Pathol 6:663–668, 1993.
El-Naggar AK, Evans DB, Mackay B. Oncocytic adrenal cortical carcinoma. Ultrastruct Pathol 15:549–556, 1991.
Forsthoefel KF. Myxoid adrenal cortical carcinoma: a case report with differential diagnostic considerations. Arch Pathol Lab Med 118:1151–1153, 1994.
Tang CK, Harriman BB, Toker C. Myxoid adrenal cortical carcinoma: a light and electron microscopic study, Arch pathol Lab Med 103:635–638, 1979.
Brown FM, Gaffey TA, Wold LE, Lloyd RV. Myxoid neoplasms of the adrenal cortex: a rare histologic variant. Am J Surg Pathol 24:396–401, 2000.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Erickson, L.A., Jin, L., Sebo, T.J. et al. Pathologic features and expression of insulin-like growth factor-2 in adrenocortical neoplasms. Endocr Pathol 12, 429–435 (2001). https://doi.org/10.1385/EP:12:4:429
Issue Date:
DOI: https://doi.org/10.1385/EP:12:4:429