Abstract
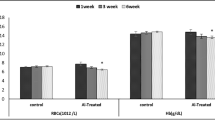
The present study was undertaken to examine possible aluminum (Al) accumulation in the brain of rats and to investigate whether subchronic exposure to the metal leads to behavioral and neurophysiological changes in both treated and control groups. Each of the groups consisted of 10 animals. Aluminum chloride (AlCl3) at a low (50 mg/kg/d) or high (200 mg/kg/d) dose was applied to male Wistar rats by gavage for 8 wk. Al-free water by gavage was given to the control group throughout the experiment. Behavioral effects were evaluated by open-field (OF) motor activity and by acoustic startle response (ASR). Electrophysiological examination was done by recording spontaneous activity and sensory-evoked potentials from the visual, somatosensory, as well as auditory cortex. The Al content of each whole brain was determined by electrothermal atomic absorption spectrophotometry. Subchronic Al exposure slightly caused some changes in the evoked potentials and electrocorticograms and in the OF and ASR performance, but these results were not statistically significant. The brain Al levels of the control and the low and high dose of Al-exposed groups were measured as 0.717±0.208 µg/g (wet weight), 0.963±0.491 µg/g (wet weight) and 1.816±1.157 µg/g (wet weight), respectively.
Similar content being viewed by others
References
P. O. Ganrot, Metabolism and possible health-effects of aluminium, Environ. Health Perspect. 65, 363–441 (1986).
R. Massey and D. Taylor, Aluminium in Food and the Environment, Special Publication No. 73, Royal Society of Chemiistry, London (1988).
IPCS, Environmental Health Criteria 194, Aluminium, WHO, Geneva (1997).
K. A. Winship, Toxicity of aluminium: a historical review, Part 2, Adv. Drug React. Toxical Rev. 12, 177–211 (1993).
R. J. Boegman and L. A. Bates, Neurotoxicity of aluminum, Can. J. Physiol. Pharmacol. 62, 1010–1014 (1984).
D. R. C. McLachlan, Aluminum and Alzheimer’s disease, Neurobiol. Aging 7, 525–532 (1986).
W. A. Banks and A. J. Kastin, Aluminum-induced neurotoxicity: alterations in membrane function at the blood-brain barrier, Neurosci. Biobehav. Rev. 13, 47–53 (1989).
J. C. Murray, C. M. Tanner, and S. M. Sprague, Aluminum neurotoxicity: a reevaluation, Clin. Neuropharmacol. 14, 179–185 (1991).
R. A. Amstrong, J. Anderson, J. D. Cowburn, et al., Aluminium administered in drinking water but not in the diet influences biopterin metabolism in the rodent, Biol. Chem. Hoppe-Seyler 373, 1075–1078 (1992).
R. T. Erasmus, J. Savory, M. R. Wills, et al., Aluminum neurotoxicity in experimental animals, Ther. Drug Monit. 15, 588–592 (1993).
U. De Boni, A. Otvos, J. W. Scott, et al., Neurofibrillary degeneration induced by systemic aluminum, Acta Neuropath. (Berl.) 35, 285–294 (1976).
W. A. Banks and A. J. Kastin, Aluminium increases permeability of the blood-brain barrier to labelled dsip and β-endorphin: possible implications for senile and dialysis dementia, Lancet ii, 1227–1229 (1983).
G. Sahin, I. Varol, A. Temizer, et al., Determination of aluminum levels in the kidney, liver, and brain of mice treated with aluminum hydroxide, Biol. Trace Element Res. 41, 129–135 (1994).
R. L. Commissaris, J. J. Cordon, S. Sprague, et al., Behavioral changes in rats after chronic aluminum and parathyroid hormone administration, Neurobehav. Toxicol. Teratol. 4, 403–410 (1982).
G. Sahin, T. Taskin, K. Benli, et al., Impairment of motor coordination in mice after ingestion of aluminum chloride, Biol. Trace Element Res. 50, 79–85 (1995).
G. M. Berlyne, R. Yagil, J. Ben Ari, et al., Aluminium toxicity in rats, Lancet i, 564–567 (1972).
A. C. Alfrey, G. R. LeGendre, and W. D. Kaehny, The dialysis encephalopathy syndrome, New Engl. J. Med. 294, 184–188 (1976).
A. I. Arieff, J. D. Cooper, D. Amstrong, et al., Dementia, renal failure, and brain aluminum, Ann. Intern. Med. 90, 741–747 (1979).
I. Dési, Neurological investigation of pesticides in animal experiments, Neurobehav. Toxicol. Teratol. 5, 503–517 (1983).
C. Cutrufo, S. Caroli, P. Delle Femmine, et al., Experimental aluminium encephalopathy quantitative EEG analysis of aluminium bioavailability, J. Neurol. Neurosurg. Psychiatry 47, 204–206 (1984).
T. L. Petit, G. B. Biederman, and P. A. McMullan, Neurofibrillary degeneration, dentric dying back, and learning-memory deficits after aluminum administration: implications for brain aging, Exp. Neurol. 67, 152–162 (1980).
V. Bernuzzi, D. Desor, and P. R. Lehr, Effects of prenatal aluminum exposure on neuromotor maturation in the rat, Neurobehav. Toxicol. Teratol. 8, 115–119 (1989).
B. M. Thorne, T. Donohoe, K. Lin, et al., Aluminum ingestion and behavior in the longevans rat, Physiol. Behav. 36, 63–67 (1986).
P. I. Otezia, C. L. Keen, B. Han, et al., Aluminum accumulation and neurotoxicity in Swiss-Webster mice after long-term dietary exposure to aluminum and citrate, Metabolism 42, 1296–1300 (1993).
G. S. Zubenko and I. Hanin, Cholinergic and noradrenergic toxicity of intraventricular aluminum chloride in the rat hippocampus, Brain Res. 498, 381–384 (1989).
S. Gaytan-Garcia, H. Kim, and M. J. Strong, Spinal motor neuron neuroaxonal spheroids in chronic aluminum neurotoxicity contain phosphatase-resistant high molecular weight neurofilament, Toxicology 108, 17–24 (1996).
S. Kumar, Biophasic effects of aluminium on cholinergic enzyme of rat brain, Neurosci. Lett. 248, 121–123 (1998).
J. R. McDermott, A. I. Smith, M. K. Ward, et al., Brain-aluminium concentration in dialysis encephalopathy, Lancet i, 901–904 (1978).
D. R. C. McLachlan, W. J. Lukiw, and T. P. A. Kruck, New evidence for an active role of aluminum in Alzheimer’s disease, Can. J. Neurol. Sci. 16, 490–497 (1989).
G. L. Wenk and K. L. Stemmer, The influence of ingested aluminum upon neorepinephrine and dopamine levels in the rat brain, Neurotoxicology 2, 347–353 (1981).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Baydar, T., Papp, A., Aydin, A. et al. Accumulation of aluminum in rat brain. Biol Trace Elem Res 92, 231–244 (2003). https://doi.org/10.1385/BTER:92:3:231
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1385/BTER:92:3:231