Abstract
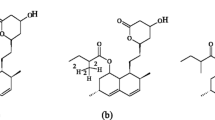
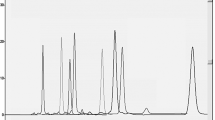
High performance liquid chromatography has been the most popular choice for the determination of atorvastatin. In this study, two-step isocratic chromatography on silica gel 60F254 HPTLC layer and densitometric quantitation at λ = 280 nm was developed for the separation of atorvastatin from plasma constituencies and sodium diclofenac as peak-tracer. The established HPTLC method was validated in terms of LOD/LOQ, linearity, recovery and repeatability. The calibration function of the analyte was linear in the range 101–353.5 ng zone−1 and the correlation coefficient was 0.9969. The limits of detection and quantitation were 30.3 and 101 ng zone−1. The recovery and relative standard deviation obtained from between-days analysis were 97.5–103.0 and 1.7–3.4%.

Similar content being viewed by others
References
Remington (2006) The science and practice of pharmacy, 21st edn. Lippincott Williams & Wilkins, New York, p 1368
Martindale (1999) The complete drug reference, 32nd edn. Pharmaceutical Press, London, p 1268
Kodama Y, Kitta Y, Nakamura T, Takano H, Umetani K, Fujioka D, Saito Y, Kawabata K, Obata J, Mende A, Kobayashi T, Kugiyama K (2006) JACC 48:43–50
Cheng X, Liao YH, Zhang J, Li B, Ge H, Yuan J, Wang M, Lu B, Liu Y, ChengY (2005) Eur J Heart Fail 7:1099–1104
Sever PS, Poulter NR, Dahlöf B, Wedel H (2005) AJC 96:39–44
Newman C, Tsai J, Szarek M, Luo D, Gibson E (2006) AJC 97:61–67
Tousoulis D, Antoniades C, Katsi V, Bosinakou E, Kotsopoulou M, Tsioufis C, Stefanadis C (2006) Int J Cardiol 109:48–52
Waters DD, LaRosa JC, Barter P, Fruchart JC, Gotto AM, Carter R, Breazna A, Kastelein JJP, Grundy SM (2006) JACC 48:1793–1799
Ertürk S, Sevinç Akta E, Ersoy L, Fıçıcıolu S (2003) J Pharm Biomed Anal 33:1017–1023
Borek-Dohalsky V, Huclova J, Barrett B, Nemec B, Ulc I, Jelinek I (2006) Anal Bioanal Chem 386:275–285
Hermann M, Christensen H, Reubsaet JL (2005) Anal Bioanal Chem 382:1242–1249
Bullen WW, Miller RA, Hayes RN (1999) J Am Soc Mass Spectrom 10:55–66
Bahrami G, Mohammadi B, Mirzaeei S, Kiani A (2005) J Chromatogr B 826:41–45
Szepesi G, Nyiredy Sz (1992) J Pharm Biomed Anal 10:1007–1015
Frenczi-Fodor K, Végh Z, Renger B (2006) TrAC 25:778–789
Sherma J (2004) Anal Chem 76:3251–3261
Jamshidi A (2004) J Planar Chromatogr 17:229–232
Poole CF, Poole SK (1995) J Chromatogr A 703:573–612
Jamshidi A, Mobedi H, Ahmad-Khanbeigi F (2006) J Planar Chromatogr 19:223–227
ICH Guideline Q2A, Validation of analytical procedures: definition and terminology (CPMP III/5626/94), 1995, Geneva, Switzerland
ICH Guideline Q2B, validation of analytical procedures: definition and terminology (CPMP III/5626/94), 1995, Geneva, Switzerland
Frenczi-Fodor K, Végh Z, Nagy-Turák A, Renger B, Zeller M (2001) J AOAC Int 84:1265–1276
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jamshidi, A., Nateghi, A.R. HPTLC Determination of Atorvastatin in Plasma. Chroma 65, 763–766 (2007). https://doi.org/10.1365/s10337-007-0228-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1365/s10337-007-0228-4