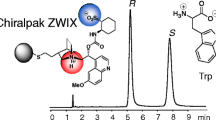
Abstract
The liquid chromatography enantiomeric separation of a series of 17 chiral sulfoxides was systematically investigated using multimodal elution with the new synthetic polymeric stationary phases P-CAP, P-CAP DP and DEAVB. The sulfoxide series was composed of aryl alkyl sulfoxides, benzoimidazole sulfoxides and the drugs modafinil, albendazole sulfoxide, omeprazole, lansoprazole, pantoprazole and rabeprazole. This work examines the effectiveness of the polymeric chiral stationary phases for the separation of chiral sulfoxides and describes the superiority of DEABV for these separations in three different elution modes. The first ever reversed phase enantiomeric separations on these columns is demonstrated.









Similar content being viewed by others
References
Lin G, Li Y, Chan ASC (2001) Introduction. In: Principles and applications of asymmetric synthesis. Wiley, New York, pp 7–8
Rayner DR, Gordon AJ, Mislow K (1968) J Am Chem Soc 90:4854–4860
Fernandez I, Khiar N (2003) Chem Rev 103:3651–3705
Harrison PWB, Kenyon J, Phillips H (1926) J Chem Soc pp 2079–2090
Kuesters E, Gerber G (1997) Chromatographia 44:91–96
Anderson JL, Ding J, McCulla RD, Jenks WS, Armstrong DW (2002) J Chromatogr A 946:197–208
Balmer K, Persson B-A, Lagerstroem P-O (1994) J Chromatogr A 660:269–273
Berthod A, Xiao TL, Liu Y, Jenks WS, Armstrong DW (2002) J Chromatogr A 955:53–69
Matlin SA, Tiritan ME, Cass QB, Boyd DR (1996) Chirality 8:147–152
Chankvetadze B, Yamamoto C, Okamoto Y (2001) J Chromatogr A 922:127–137
Cass QB, Batigalhia F (2003) J Chromatogr A 987:445–452
Belaz KRA, Coimbra M, Barreiro JC, Montanari CA, Cass QB (2008) J Pharm Biomed Anal 47:81–87
Belaz KRA, Cass QB, Oliveira RV (2008) Talanta 76:146–153
Cass QB, Ferreira Galatti T (2008) J Pharm Biomed Anal 46:937–944
Zhong Q, Han X, He L, Beesley TE, Trahanovsky WS, Armstrong DW (2005) J Chromatogr A 1066:55–70
Gasparrini F, Misiti D, Villani C (2003) WO 2003079002, p 25
Gasparrini F, Misiti D, Villani C, Rompietti R (2003) In: Abstracts of the 15th international symposium on chirality (ISCD-15), Shizuoka, Japan, p 132
Han X, He L, Zhong Q, Beesley TE, Armstrong DW (2006) Chromatographia 63:13–23
Han X, Wang C, He L, Beesley TE, Armstrong DW (2007) Anal Bioanal Chem 387:2681–2697
Montanari MLC, Cass QB, Andricopulo AD, Leitao A, Montanari CA (2005) Anal Chim Acta 545:33–45
Montanari MLC, Cass QB, Leitao A, Andricopulo AD, Montanari CA (2006) J Chromatogr A 1121:64–75
Batigalhia F, Zaldini-Hernandes M, Ferreira AG, Malvestiti I, Cass QB (2001) Tetrahedron 57:9669–9676
Bonato PS, Paias FO (2004) J Braz Chem Soc 15:318–323
Cirilli R, Ferretti R, Gallinella B, De Santis E, Zanitti L, La Torre F (2008) J Chromatogr A 1177:105–113
Cass QB, Degani ALG, Cassiano NM, Pedrazolli J (2002) J Chromatogr B 766:153–160
Cass QB, Lima VV, Oliveira RV, Cassiano NM, Degani ALG, Pedrazzoli J (2003) J Chromatogr B 798:275–281
Acknowledgments
This study was supported by the Fundação de Amparo a Pesquisa do Estado de São Paulo (FAPESP). The authors also acknowledge the Conselho Nacional de Desenvolvimento Científico e Técnologico (CNPq) and the manufactures for the donations of several chiral sulfoxides. We gratefully acknowledge the support from the National Institute of Health (GM053825-11 and GM053825-12) for this work.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lourenço, T.C., Armstrong, D.W. & Cass, Q.B. Enantiomeric Resolution of a Chiral Sulfoxide Series by LC on Synthetic Polymeric Columns with Multimodal Elution. Chroma 71, 361–372 (2010). https://doi.org/10.1365/s10337-009-1460-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1365/s10337-009-1460-x