Abstract
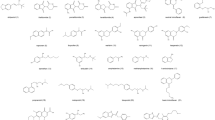
Six phenylcarbamate derivatives of cellulose and amylose having both an electron-withdrawing fluoro group and an electron-donating methyl group on the phenyl moieties were synthesized, and their chiral recognition abilities were evaluated as chiral stationary phases for high-performance liquid chromatography. The resolving abilities were compared with those of chloro-methylphenylcarbamate derivatives of cellulose and amylose. The chiral recognition abilities of fluoro-methylphenylcarbamates of cellulose and amylose were dependent on the position of the substituents. The cellulose derivatives with fluoro and methyl groups at the meta and para positions showed higher resolving power than the ortho- and meta-disubstituted derivative as observed for the chloro-methylphenylcarbamate derivatives of cellulose. On the other hand, the ortho-substituted derivative of amylose, tris(5-fluoro-2-methylphenylcarbamate), showed high chiral recognition as well as the meta- and para-disubstituted derivatives. The different chiral recognition was discussed on the basis of IR and CD spectroscopic data.
Similar content being viewed by others
Article PDF
References
S. Ahuja, in “Chiral Separation by Liquid Chromatography (ACS symposium Series, No. 471),” American Chemical Society, Washington, D.C., 1991.
W. H. Pirkle and T. C. Pochapsky, Chem. Rev., 89, 347 (1989).
D. R. Taylor and K. Maher, J. Chromatogr. Sci., 30, 67 (1992).
S. G. Allenmark, in “Chromatographic Enantioseparation,” Ellis Horwood, Chichester, 1988, Chapter 7, p 90.
Y. Okamoto and Y. Kaida, J. Chromatogr. A., 666, 403 (1994).
A. Ishikawa and T. Shibata, J. Liq. Chromatogr., 16, 859 (1993).
J. Dingenen, in “A Practical Approach to Chiral Separations by Liquid Chromatography,” G. Subramanian, Ed., VCH Publishers, New York, 1994, Chapter 6, p 115.
Y. Okamoto, M. Kawashima, and K. Hatada, J. Chromatogr., 363, 173 (1986).
B. Chankvetadze, E. Yashima, and Y. Okamoto, J. Chromatogr. A, 670, 39 (1994).
B. Chankvetadze, E. Yashima, and Y. Okamoto, Chem. Lett., 617 (1993).
B. Chankvetadze, E. Yashima, and Y. Okamoto, J. Chromatogr. A, 694, 101 (1995).
Y. Kaida and Y. Okamoto, Bull. Chem. Soc. Jpn., 65, 2286 (1992).
E. Yashima, M. Yamada, and Y. Okamoto, Chem. Lett., 579 (1994).
E. Yashima, M. Yamada, Y. Kaida, and Y. Okamoto, J. Chromatogr. A, 694, 347 (1995).
Y. Okamoto and Y. Kaida, unpublished results.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Yashima, E., Yamamoto, C. & Okamoto, Y. Enantioseparation on Fluoro-Methylphenylcarbamates of Cellulose and Amylose as Chiral Stationary Phases for High-Performance Liquid Chromatography. Polym J 27, 856–861 (1995). https://doi.org/10.1295/polymj.27.856
Issue Date:
DOI: https://doi.org/10.1295/polymj.27.856
Keywords
This article is cited by
-
Cellulose phenylcarbamate-derived hybrid bead-type chiral packing materials for efficient chiral recognition
Cellulose (2021)
-
Stereoselective interactions of chiral dipeptides on amylose based chiral stationary phases
Science China Chemistry (2015)
-
New approach for chiral separation: from polysaccharide-based materials to chirality-responsive polymers
Science China Chemistry (2014)