Abstract
Background
Isolated locoregional recurrences (ILRRs) of breast cancer confer a significant risk for the development of distant metastasis. Management practices and second ILRR events in the Chemotherapy as Adjuvant for LOcally Recurrent breast cancer (CALOR) trial were investigated.
Methods
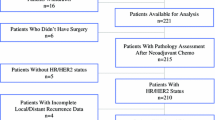
In this study, 162 patients with ILRR were randomly assigned to receive postoperative chemotherapy or no chemotherapy. Descriptive statistics characterize outcomes according to local therapy and the influence of hormone receptor status on subsequent recurrences. Competing risk regression models, Kaplan-Meier estimates, and Cox proportional hazards models were used to evaluate associations between treatment, site of second recurrence, and outcome.
Results
The median follow-up period was 4.9 years. Of the 98 patients who received breast-conserving primary surgery 89 had an ipsilateral-breast tumor recurrence. Salvage mastectomy was performed for 73 patients and repeat lumpectomy for 16 patients. Another eight patients had nodal ILRR, and one patient had chest wall ILRR. Among 64 patients whose primary surgery was mastectomy, 52 had chest wall/skin ILRR, and 12 had nodal ILRR. For 15 patients, a second ILRR developed a median of 1.6 years (range 0.08–4.8 years) after ILRR. All second ILRRs occurred for patients with progesterone receptor-negative ILRR. Death occurred for 7 (47 %) of 15 patients with a second ILRR and 19 (51 %) of 37 patients with a distant recurrence. As shown in the multivariable analysis, the significant predictors of survival after either a second ILRR or distant recurrence were chemotherapy for the primary cancer (hazard ratio [HR], 3.55; 95 % confidence interval [CI], 1.15–10.9; p = 0.03) and the interval (continuous) from the primary surgery (HR, 0.87; 95 % CI, 0.75–1.00; p = 0.05).
Conclusions
Second ILRRs represented about one third of all recurrence events after ILRR, and all were PR-negative. These second ILRRs and distant metastases portend an unfavorable outcome.


Similar content being viewed by others
References
Halverson KJ, Perez CA, Kuske RR, et al. Survival following locoregional recurrence of breast cancer: univariate and multivariate analysis. Int J Radiat Oncol Biol Phys. 1992;23:285–91.
Fowble B, Solin LJ, Schultz DJ, et al. Breast recurrence following conservative surgery and radiation: patterns of failure, prognosis, and pathologic findings from mastectomy specimens with implications for treatment. Int J Radiat Oncol Biol Phys. 1990;19:833–42.
Fisher B, Anderson S, Fisher ER, et al. Significance of ipsilateral breast tumour recurrence after lumpectomy. Lancet. 1991;338:327–31.
Veronesi U, Marubini E, Del Vecchio M, et al. Local recurrences and distant metastases after conservative breast cancer treatments: partly independent events. J Natl Cancer Inst. 1995; 87:19–27.
Wapnir IL, Anderson SJ, Mamounas EP, et al. Prognosis after ipsilateral breast tumor recurrence and locoregional recurrences in five National Surgical Adjuvant Breast and Bowel Project node-positive adjuvant breast cancer trials. J Clin Oncol. 2006;24:2028–37.
Nielsen HM, Overgaard M, Grau C, et al. Locoregional recurrence after mastectomy in high-risk breast cancer–risk and prognosis: an analysis of patients from the DBCG 82 b&c randomization trials. Radiother Oncol. 2006;79:147–55.
Anderson SJ, Wapnir I, Dignam JJ, et al. Prognosis after ipsilateral breast tumor recurrence and locoregional recurrences in patients treated by breast-conserving therapy in five National Surgical Adjuvant Breast and Bowel Project protocols of node-negative breast cancer. J Clin Oncol. 2009;27:2466–73.
Botteri E, Rotmensz N, Sangalli C, et al. Unavoidable mastectomy for ipsilateral breast tumour recurrence after conservative surgery: patient outcome. Ann Oncol. 2009;20:1008–12.
Kuerer HM, Arthur DW, Haffty BG. Repeat breast-conserving surgery for in-breast local breast carcinoma recurrence: the potential role of partial breast irradiation. Cancer. 2004;100:2269–80.
Schuck A, Könemann S, Matthees B, et al. Radiotherapy in the treatment of locoregional relapses of breast cancer. Br J Radiol. 2002;75:663–9.
Siglin J, Champ CE, Vakhnenko Y, et al. Radiation therapy for locally recurrent breast cancer. Int J Breast Cancer. 2012;2012:571946.
Hannoun-Levi J-M, Ihrai T, Courdi A. Local treatment options for ipsilateral breast tumour recurrence. Cancer Treat Rev. 2013;39:737–41.
Maaskant-Braat AJG, Voogd AC, Roumen RMH, et al. Repeat sentinel node biopsy in patients with locally recurrent breast cancer: a systematic review and meta-analysis of the literature. Breast Cancer Res Treat. 2013;138:13–20.
Aebi S, Gelber S, Anderson SJ, et al. Chemotherapy for isolated locoregional recurrence of breast cancer (CALOR): a randomised trial. Lancet Oncol. 2014;15:156–63.
Kurtz JM, Jacquemier J, Amalric R, et al. Is breast conservation after local recurrence feasible? Eur J Cancer. 1991;27:240–4.
Salvadori B, Marubini E, Miceli R, et al. Reoperation for locally recurrent breast cancer in patients previously treated with conservative surgery. Br J Surg. 1999;86:84–7.
Ishitobi M, Komoike Y, Nakahara S, et al. Repeat lumpectomy for ipsilateral breast tumor recurrence after breast-conserving treatment. Oncology. 2011;81:381–6.
Gentilini O, Botteri E, Veronesi P, et al. Repeating conservative surgery after ipsilateral breast tumor reappearance: criteria for selecting the best candidates. Ann Surg Oncol. 2012;19:3771–6.
Kurtz JM. Factors influencing the risk of local recurrence in the breast. Eur J Cancer. 1991;28:660–6.
Ishitobi M, Okumura Y, Nishimura R, et al. Repeat lumpectomy for ipsilateral breast tumor recurrence (IBTR) after breast-conserving surgery: the impact of radiotherapy on second IBTR. Breast Cancer. 2014;21:754–60.
van der Pol CC, van Geel AN, Menke-Pluymers MBE, et al. Prognostic factors in 77 curative chest wall resections for isolated breast cancer recurrence. Ann Surg Oncol. 2009;16:3414–21.
Fine JP, Gray RJ. A proportional hazards model for the subdistribution of a competing risk. J Am Stat Assoc. 2012; 94:496–509.
Mamounas EP, Tang G, Liu Q. The importance of systemic therapy in minimizing local recurrence after breast-conserving surgery: the NSABP experience. J Surg Oncol. 2014;110:45–50.
Perez EA, Romond EH, Suman VJ, et al. Trastuzumab plus adjuvant chemotherapy for human epidermal growth factor receptor 2-positive breast cancer: planned joint analysis of overall survival from NSABP B-31 and NCCTG N9831. J Clin Oncol. 2014;32:3744–52.
Hannoun-Levi J-M, Houvenaeghel G, Ellis S, et al. Partial breast irradiation as second conservative treatment for local breast cancer recurrence. Int J Radiat Oncol Biol Phys. 2004;60:1385–92.
Sedlmayer F, Zehentmayr F, Fastner G. Partial breast re-irradiation for local recurrence of breast carcinoma: benefit and long-term side effects. Breast. 2013;22:S141–S146.
Hannoun-Levi J-M, Resch A, Gal J, et al. Accelerated partial breast irradiation with interstitial brachytherapy as second conservative treatment for ipsilateral breast tumour recurrence: multicentric study of the GEC-ESTRO Breast Cancer Working Group. Radiother Oncol. 2013;108:226–31.
Trombetta M, Hall M, Julian TB. Long-term follow-up of breast preservation by re-excision and balloon brachytherapy after ipsilateral breast tumor recurrence. Brachytherapy. 2014;13:488–92.
Tanis E, van de Velde CJH, Bartelink H, et al. Locoregional recurrence after breast-conserving therapy remains an independent prognostic factor even after an event free interval of 10 years in early-stage breast cancer. Eur J Cancer. 2012;48:1751–6.
Early Breast Cancer Trialists’ Collaborative Group (EBCTCG), McGale P, Taylor C, et al. Effect of radiotherapy after mastectomy and axillary surgery on 10-year recurrence and 20-year breast cancer mortality: meta-analysis of individual patient data for 8135 women in 22 randomised trials. Lancet. 2014;383:2127–35.
Early Breast Cancer Trialists’ Collaborative Group (EBCTCG), Darby S, McGale P, Correa C, et al. Effect of radiotherapy after breast-conserving surgery on 10-year recurrence and 15-year breast cancer death: meta-analysis of individual patient data for 10,801 women in 17 randomised trials. Lancet. 2011;378:1707–16.
Acknowledgments
The CALOR trial was supported in part by Public Service Grants U10-CA-180868, U10-CA-189867, U10-CA-180822, and U24-CA-75362 from the National Cancer Institute, Department of Health and Human Services. The International Breast Cancer Study Group is supported in part by the Swiss Group for Clinical Cancer Research (SAKK), Frontier Science and Technology Research Foundation, Australia and New Zealand Breast Cancer Trials Group, Swedish Cancer Society, Cancer Research Switzerland/Oncosuisse, Cancer Association of South Africa, Foundation for Clinical Research of Eastern Switzerland (OSKK). Spanish participation was funded by Grupo Español de Investigación en Cáncer de Mama (GEICAM), and Dutch participation by BOOG, the Dutch Breast Cancer Trialists’ Group.
Disclosure
None of the authors have any conflicts of interest to report.
Author information
Authors and Affiliations
Consortia
Corresponding author
Additional information
Members of the Chemotherapy as Adjuvant for the LOcally Recurrent (CALOR) Breast Cancer Collaborative Group are listed in the Supplementary Appendix.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Wapnir, I.L., Gelber, S., Anderson, S.J. et al. Poor Prognosis After Second Locoregional Recurrences in the CALOR Trial. Ann Surg Oncol 24, 398–406 (2017). https://doi.org/10.1245/s10434-016-5571-y
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-016-5571-y