Abstract
Background: Concurrent chemoradiotherapy (CCRT) for locally advanced rectal cancer is an important modality for curative resection, but tumors show wide spectrum response. The purpose of this study was to investigate any correlation among related genetic mutations, proliferative index, and tumor response after CCRT.
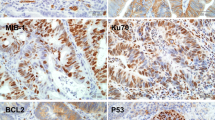
Methods: This study included 23 patients with rectal cancer, who were preoperatively staged as at least T3 N1 or T4 (determined by transrectal ultrasonography and MRI). Enrolled patients were given 5-FU 450 mg/m2/day and leucovorin 20 mg/m2/day intravenously for 5 days during weeks 1 and 5 of radiotherapy (45–54 Gy). Surgical resection was performed 4 weeks after completion of the scheduled treatment. Tumor response was classified as CR (complete response), PR (partial response: 50% diminution of tumor volume and downstaging), and NR (no response). Paraffin-embedded tissue obtained before chemoradiotherapy was studied by immunohistochemical staining for p53, BCL-2, and Ki-67. The extent of tumor response was correlated with proliferative activity and was measured by immunostaining Ki-67 proliferative antigen and the expression of p53 and BCL-2 oncoproteins.
Results: All patients were resectable. CR was obtained in 4 patients, PR in 10 patients, and NR in 9 patients. The p53 mutation was noted in 16 patients: NR in 5 patients, PR in 9 patients, and CR in 2 patients (P = .638). BCL-2 expression was noted in 11 patients: NR in 4 patients, PR in 3 patients, and CR in 4 patients (P = .799). The Ki-67 labeling index was NR: 615.4 ± 47.2; PR: 663.2 ± 20.4; and CR: 765.5 ± 58.3 (CR + PR vs. NR, P = .029).
Conclusions: Immunohistochemical expression of p53 and BCL-2 does not correlate with tumor response after CCRT, but Ki-67 labeling may be a useful parameter for radiosensitive tumors selected for CCRT.
Similar content being viewed by others
REFERENCES
Kim NK, Sohn SK, Min JS, Sung JS, Noh JK. Preoperative concurrent chemoradiotherapy in locally advanced rectal cancer. J Korean Society Coloproctology 2000; 16: 93–8.
Minsky BD, Cohen AM, Kemeny N, et al. Combined modality therapy of rectal cancer: decreased acute toxicity with the peroperative approach. J Clin Oncol 1992; 10: 1218–24.
Charis RS, Tuler DS, Anscher MS, et al. Preoperative radiation and chemotherapy in the treatment of adenocarcinoma of the rectum. Ann Surg 1995; 221: 778–87.
Willet CG, Hagan M, Daley W, Warland G, Shellito PC, Compton CC. Changes in tumor proliferation of rectal cancer induced by preoperative 5-Fluorouracil and irradiation. Dis Colon Rectum 1998; 41: 62–7.
FuCG, Tominaga O, Nagawa H, et al. Role of p53 and p21/WAF1 detection in patient selection for preoperative radiotherapy in rectal cancer patients. Dis Colon Rectum 1998; 41: 68–74.
Sakakura C, Koide K, Ichikawa D, et al. Analysis of histological therapeutic effect, apoptosis rate and p53 status after combined treatment with radiation, hyperthermia and 5-fluorouracil suppositories for advanced rectal cancer. Br J Cancer 1998; 77: 159–66.
Fan S, EL-Deiry S, Boe I, et al. p53 gene mutations are associated with decreased sensitivity of human lymphoma cells to DNA damaging agents. Cancer Res 1994; 54: 5824–30.
Namba H, Hara T, Tukazaki T, et al. Radiation-induced G1 arrest is selectively mediated by the p53-WAF/CIP1 pathway in human thyroid cells. Cancer Res 1995; 55: 2075–80.
Lowe SW, Schmit EM, Smith EW, Osborne BA, Jacks T. p53 is required for radiation induced apoptosis in mouse thymocytes. Nature 1993; 362: 847–52.
Lowe SW, Bodis S, McClatchey A, et al. p53 status and the efficacy of cancer therapy I vivo. Science 1994; 266: 807–10.
Xia F, Wang X, Wang YH, et al. Altered p53 status correlates with differences in sensitivity to radiation induced mutation and apoptosis in two closely related human lymphoblast lines. Cancer Res 1995; 55: 12–5.
Lee JM Bernstein A. p53 mutations increase resistance to ionizing radiation. Proc Natl Acad Sci U S A 1993; 90: 5742–46.
Brachman DG, Beckett M, Graves D, Haraf D, Vokes E, Weichselbaum RR. p53 mutation does not correlate with radiosensitivity in 24 head and neck cancer cell line. Cancer Res 1993; 53: 3667–9.
Clarke AR, Purdie CA, Harrison DJ, et al. Thymocytic apoptosis induced by p53 -dependent and independent pathways. Nature 1993; 362: 849–52.
Slichenmeyer WJ, Nelson WG, Slebos RJ, Kastan MB. Loss of a p53 associated G1 checkpoint does not decrease cell survival following DNA damage. Cancer Res 1993; 53: 4164–8.
Lanza G Jr, Cavazzini L, Borghi L, Ferretti S, Buccoliero F, Rubbini M. Immunohistochemical assessment of growth fractions in colorectal adenocarcinomas with monoclonal antibody ki-67. relation to clinical and pathologic variables. Path Res Pract 1990; 186: 608–18.
DuM, Singh N, Husseuin A, Isaacson PG, Pan L. Positive correlation between apoptotic and proliferative indices in gastrointestinal lymphomas of mucosa-associate lymphoid tissue (MALT). J Pathol 1996; 178: 379–84.
Fukunaga-Johnson N, Ryan JJ, Wicha M, Nunez G, Clarke MF. Bcl-2 protects murine erythroleukemiacells from p53-dependent and independent radiation-induced cell death. Carcinogenesis 1995; 16: 1761–7.
Bronner MP, Culin C, Reed JC, Furth EE. The bcl-2 proto-oncogene and the gastrointestinal epithelial tumor and progression model. Am J Pathol 1995; 146: 20–26.
Reed JC. Bcl-2: prevention of apoptosis as a mechanism of drug resistance. Hematol Oncol Clin North Am 1995; 9: 451–73.
Oefner D, Riehemann K, Maier H, et al. Immunohistochemically detectable bcl-2 expression in colorectal carcinoma: correlation with tumor stage and patient survival. Br J Cancer 1995; 72: 981–5.
Leek RD, Kaklamamis L, Pezzella, Gatter KC, Harris AL. Bcl-2 in normal human breast and carcinoma, associated with estrogen receptor positive, epidermal growth factor receptor -negative tumor and in situ cancer. Br J Cancer 1994; 69: 135–9.
Pezzella F, Turley H, Kuzu I, et al. Bcl-2 protein in non-small cell lung carcinoma. N Engl J Med 1993; 329: 690–4.
Krajewska M, Moss SF, Krajewski S, Song K, Holt PR, Reed JC. Elevated expression of Bcl-x and reduced Bak in primary colorectal adenocarcinoma. Cancer Res 1996; 56: 2422–7.
Flohil CC, Janssen PA, Bosman F. Expression of bcl-2 protein in hyperplastic polyps, adenomas, and carcinomas of the colon. J Pathol 1996; 178: 393–7.
Reed John C. Bcl-2 family proteins: Strategies for overcoming chemoresistance in Cancer. Apoptosis: pharmacological implications and therapeutic opportunities. Advances In Pharmacology, vol 41 New York: Academic Press, 1997 501–6.
Charalambous GK, Gomatos IP, Konstadoulakis MM, et al. Protein expression of bax, bcl-2 and p53 in patients with Non-Hodgkin’s Gastric Lymphoma: prognostic significance. World J Surg 2000; 24: 608–14.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kim, N.K., Park, J.K., Lee, K.Y. et al. p53, BCL-2, and Ki-67 Expression According to Tumor Response After Concurrent Chemoradiotherapy for Advanced Rectal Cancer. Ann Surg Oncol 8, 418–424 (2001). https://doi.org/10.1007/s10434-001-0418-5
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s10434-001-0418-5