Abstract
Background
Retroperitoneal lymphadenectomy (RPLND), chemotherapy, and active surveillance represent treatment options for patients with nonseminomatous germ-cell tumors of the testis (NSGCTT). Our objective was to assess the utilization rate of RPLND in patients with NSGCTT.
Methods
Within the Surveillance, Epidemiology and End Results (SEER) cohort, 4,620 and 1,671 patients with stage I and II NSGCTT were diagnosed between the years 1988 and 2006. Univariable and multivariable logistic regression analyses were performed to test the predictors of RPLND use in respectively patients with stage I and II NSGCTT.
Results
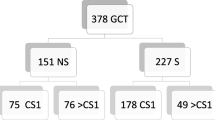
The rate of RPLND according to stage I and II was 27 and 58%, respectively. In patients with stage I disease, the rate of RPLND decreased from 39% in 1988–1995 to 18% in 2004–2006 (P < 0.001), and remained stable for stage II patients (62–56%, P = 0.2). Regional variability existed regarding the rate of RPLND use only in stage I (Utah: 51% vs. Louisiana: 16%). Multivariable analyses performed in stage I NSGCTT revealed that year of diagnosis, SEER registry, and age were significant predictors of RPLND use. However, none of these variables achieved statistical significance within stage II NSGCTT patients.
Conclusions
In patients with stage I NSGCCT, the RPLND utilization rate decreased during the study span. This observation may be ascribed to wider use of surveillance or chemotherapy. No temporal difference was recorded in patients with stage II NSGCCT.




Similar content being viewed by others
References
McKiernan JM, Goluboff ET, Liberson GL, et al. Rising risk of testicular cancer by birth cohort in the United States from 1973 to 1995. J Urol. 1999;162:361-3.
Bray F, Richiardi L, Ekbom A, et al. Trends in testicular cancer incidence and mortality in 22 European countries: continuing increases in incidence and declines in mortality. Int J Cancer. 2006;118:3099–111.
de Wit R, Fizazi K. Controversies in the management of clinical stage I testis cancer. J Clin Oncol. 2006;24:5482–92.
Albers P, Siener R, Krege S, et al. Randomized phase III trial comparing retroperitoneal lymph node dissection with one course of bleomycin and etoposide plus cisplatin chemotherapy in the adjuvant treatment of clinical stage I Nonseminomatous testicular germ cell tumors: AUO trial AH 01/94 by the German Testicular Cancer Study Group. J Clin Oncol. 2008;26:2966–72.
Surveillance, Epidemiology and End Results (SEER). Surveillance Epidemiology and End Results. National Cancer Institute, 2010. Available at http://seer.cancer.gov/.
Du XL, Fang S, Coker AL, et al. Racial disparity and socioeconomic status in association with survival in older men with local/regional stage prostate carcinoma: findings from a large community-based cohort. Cancer. 2006;106:1276-85.
Robert SA, Strombom I, Trentham-Dietz A, et al. Socioeconomic risk factors for breast cancer: distinguishing individual- and community-level effects. Epidemiology. 2004; 15: 442–50.
National Comprehensive Cancer Network Guidelines (NCCN). Testicular cancer. Clinical practice guidelines in oncology. 2010. www.nccn.org.
Hotte SJ, Mayhew LA, Jewett M, et al. Management of stage I non-seminomatous testicular cancer: a systematic review and meta-analysis. Clin Oncol (R Coll Radiol).2010;22: 17–26.
Colls BM. Re: Clinical stage I testis cancer: long-term outcome of patients on surveillance. J Urol. 1999;162:169.
Daugaard G, Petersen PM, Rørth M. Surveillance in stage I testicular cancer. APMIS. 2003;111:76-83; discussion 83–75.
Ernst DS, Brasher P, Venner PM, et al. Compliance and outcome of patients with stage 1 non-seminomatous germ cell tumors (NSGCT) managed with surveillance programs in seven Canadian centres. Can J Urol. 2005;12:2575–80.
Read G, Stenning SP, Cullen MH, et al. Medical Research Council prospective study of surveillance for stage I testicular teratoma. Medical Research Council Testicular Tumors Working Party. J Clin Oncol. 1992;10:1762-8.
Albers P, Siener R, Krege S, et al. Randomized phase III trial comparing retroperitoneal lymph node dissection with one course of bleomycin and etoposide plus cisplatin chemotherapy in the adjuvant treatment of clinical stage I Nonseminomatous testicular germ cell tumors: AUO trial AH 01/94 by the German Testicular Cancer Study Group. J Clin Oncol. 2008;26:2966–72.
Nguyen CT, Fu AZ, Gilligan TD, et al. Defining the optimal treatment for clinical stage I nonseminomatous germ cell testicular cancer using decision analysis. J Clin Oncol. 2010;28:119–25.
Williams SB, McDermott DW, Winston D, et al. Morbidity of open retroperitoneal lymph node dissection for testicular cancer: contemporary perioperative data. BJU Int. 2010;105:918–21.
Acknowledgment
Pierre I. Karakiewicz is partially supported by the University of Montreal Health Center Urology Specialists, Fonds de la Recherche en Santé du Quebec, the University of Montreal Department Of Surgery and the University of Montreal Health Center (CHUM) Foundation.
Author information
Authors and Affiliations
Corresponding author
Additional information
Maxine Sun and Firas Abdollah contributed equally to this paper.
Rights and permissions
About this article
Cite this article
Sun, M., Abdollah, F., Budaüs, L. et al. Trends of Retroperitoneal Lymphadenectomy Use in Patients with Nonseminomatous Germ Cell Tumor of the Testis: A Population-Based Study. Ann Surg Oncol 18, 2997–3004 (2011). https://doi.org/10.1245/s10434-011-1722-3
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-011-1722-3