Abstract
Background: Patients with renal cell carcinoma (RCC) do not develop an effective antitumor immune response, despite significant infiltration by lymphocytes. Tumor production of immunosuppressive factors may account for this failure. The object of this study was to investigate the production of immunosuppressive mediators, especially prostaglandin E2 (PGE2), by RCC.
Methods: Peripheral blood mononuclear cells (PBMC) were cocultured with conditioned medium (CM) from human RCC cell lines in the presence or absence of NS-398, a selective cyclooxygenase 2 (COX-2) inhibitor. Supernatants were analyzed for levels of PGE2, interleukin (IL)-10, IL-6, IL-2, interferon-γ, and IL-12. The effects of RCC CM on PBMC proliferation were also examined. The expression of basal and stimulated COX-2 messenger RNA in the cell lines was assessed by reverse transcriptase-polymerase chain reaction.
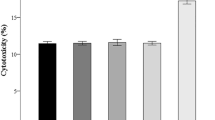
Results: RCC CM significantly increased PGE2 production by PBMC. T-helper type 2 (Th2) cytokine production was also significantly increased. Th1 cytokines were unchanged or decreased. RCC CM increased proliferation of PBMC. Coculture with NS-398 reduced PBMC PGE2 production to below control levels and significantly decreased IL-6 production and PBMC proliferation. NS-398 had no effect on cellular production of IL-10 or Th1 cytokines.
Conclusions:Human RCC inhibits the host antitumor immune response by promoting PGE2 production and Th2 cytokines in PBMC. Selective inhibition of COX-2 may have a role in abrogating this effect.
Similar content being viewed by others
REFERENCES
Landis S, Murray T, Bolden S. Cancer statistics, 1999. Cancer J Clin 1999;49:8–31.
Amato R. Chemotherapy for renal cell carcinoma. Semin Oncol 2000;27:177–86.
Evans R. Macrophage requirement for growth of a murine fibrosarcoma. Br J Cancer 1978;37:1086–9.
Leek R, Lewis C, Whitehouse R, Greenall M, Clarke J, Harris A. Association of macrophage infiltration with angiogenesis and prognosis in invasive breast cancer. Cancer Res 1996;56:4625–9.
Bennett A. The production of prostanoids in human cancers and their implications for tumor progression. Prog Lipid Res 1986;25:539–42.
Goodwin J, Ceuppens J. Regulation of the immune response by prostaglandins. J Clin Immunol 1983;3:295–315.
Oshima M, Dinchuk J, Kargman S. Suppression of intestinal polyposis in Apc delta 716 knockout mice by inhibition of cyclooxygenase 2 (COX-2). Cell 1996;87:803–9.
Hull M, Booth J, Tisbury A, et al. Cyclooxygenase 2 is upregulated and localized to macrophages in the intestine of Min mice. Br J Cancer 1999;79:1399–405.
Eisengart C, Mestre J, Naama H, et al. Prostaglandins regulate melanoma-induced cytokine production in macrophages. Cell Immunol 2000;204:143–9.
Pellegrini P, Berghella A, Beato TD, Cicia S, Adorno D, Casciani C. Disregulation in Th1 and Th2 subsets of CD4+ T cells in peripheral blood of colorectal cancer patients and involvement in cancer establishment and progression. Cancer Immunol Immunother 1996;42:1–8.
Mosman T, Coffman R. Th1 and Th2 cells: different patterns of lymphokine secretion lead to different functional properties. Annu Rev Immunol 1989;7:145–73.
DelPrete G, DeCarli M, Ricci M, Romagnani S. Helper activity for immunoglobulin synthesis of T helper type 1 (Th1) and Th2 human T cell clones: the help of Th 1 clones is limited by their cytolytic capacity. J Exp Med 1991;174:809–13.
Asselin-Paturel C, Echchakir H, Carayol G, et al. Quantitative analysis of Th1, Th2 and TGF-beta 1 cytokine expression in tumor, TIL and PBL of non-small cell lung cancer patients. Int J Cancer 1998;77:7–12.
Ren Z, Pang G, Clancy R, et al. Gastric carcinoma: T cell response and vascularity. Shift of the gastric T-cell response in gastric carcinoma. J Gastroenterol Hepatol 2001;16:142–8.
Snijdewint F, Kalinski P, Wierenga E, Bos J, Kapsenberg M. Prostaglandin E2 differentially modulates cytokine secretion profiles of human T helper lymphocytes. J Immunol 1993;150:5321–9.
Katamura K, Shintaku N, Yamauchi Y, et al. Prostaglandin E2 at priming of naive CD4+ T cells inhibits acquisition of ability to produce IFN-gamma and IL-2, but not IL-4 and IL-5. J Immunol 1995;155:4604–12.
Wu C, Wang K, McDyer J, Seder R. Prostaglandin E2 and dexamethasone inhibit IL-12 receptor expression and IL-12 responsiveness. J Immunol 1998;161:2723–30.
Tineke C, Kraan V, Boeije L, Smeenk R, Wijdenes J, Aarden L. Prostaglandin-E2 is a potent inhibitor of human interleukin 12 production. J Exp Med 1995;181:775–9.
Hilkens G, Snijders A, Vermeulen H, Meide P, Wierenga E, Kapsenberg M. Accessory cell-derived IL-12 and prostaglandin E2 determine the IFN-gamma level of activated human CD4+ T cells. Ann N Y Acad Sci 1996;795:349–50.
Kuroda E, Sugiura T, Zeki K, Yoshida Y, Yamashita U. Sensitivity difference to the suppressive effect of prostaglandin E2 among mouse strains: a possible mechanism to polarize Th2 type response in BALB/c mice. J Immunol 2000;164:2386–95.
Oliver R, Nethersell A, Bottomley J. Unexplained spontaneous regression and alpha-interferon as treatment for metastatic renal cell carcinoma. Br J Urol 1989;63:128–31.
Tykka H, Oravisto K, Lehtonen T, Sarna S, Tallberg T. Active specific immunotherapy of advanced renal-cell carcinoma. Eur Urol 1978;4:250–8.
Finke J. Characterization of a human renal cell carcinoma specific cytotoxic CD8+ T cell line. J Immunother 1992;11:1–11.
Uzzo R, Rayman P, Kolenko V, et al. Renal cell carcinoma-derived gangliosides suppress nuclear factor-kappa B activation in T cells. J Clin Invest 1999;104:769–76.
Meischer S, Stoeck M, Qiao L, Barras C, Barrelet L, VonFliedner V. Preferential clonogenic deficit of CD8-positive T-lymphocytes infiltrating human solid tumors. Cancer Res 1988;48:6992–8.
Alexander J, Kudoh S, Melsop K, et al. T-cells infiltrating renal cell carcinoma display a poor proliferative response even though they can produce interleukin 2 and express interleukin 2 receptors. Cancer Res 1993;53:1380–7.
Tsujii M, Bois RD. Alterations in cellular adhesion and apoptosis in epithelial cells overexpressing prostaglandin endoperoxide synthase 2. Cell 1995;83:493–501.
Tsujii M, Kawano S, Tsujii S, Sawaoka H, Hori M, Bois RD. Cyclooxygenase regulates angiogenesis induced by colon cancer cells. Cell 1998;93:705–16.
Plescia O, Smith A, Grinwich K. Subversion of the immune system by tumor cells and the role of prostaglandins. Proc Natl Acad Sci U S A 1975;72:1848–51.
Bennett A, Charlier E, McDonald A, Simpson J, Stamford I, Zebro T. Prostaglandins and breast cancer. Lancet 1977;2:624–6.
Bennett A. The production of prostanoids in human cancers, and their implications for tumor progression. Prog Lipid Res 1986;25:539–42.
Subbaramaiah K, Zakim D, Weksler B, Dannenberg A. Inhibition of cyclooxygenase: a novel approach to cancer prevention. Proc Soc Exp Biol Med 1997;216:201–10.
Rigas B, Goldman I, Levine L. Altered eicosanoid levels in human colon cancer. J Lab Clin Med 1993;122:518–23.
Tucker O, Dannenberg A, Yang E, et al. Cyclooxygenase 2 is up-regulated in human pancreatic cancer. Cancer Res 1999;59:987–90.
Chan G, Boyle J, Yang E, et al. Cyclooxygenase-2 is up-regulated in squamous cell carcinoma of the head and neck. Cancer Res 1999;59:991–4.
Soslow R, Dannenberg A, Rush D, et al. COX-2 is expressed in human pulmonary, colonic and mammary tumors. Cancer 2000;89:2637–45.
Kulkarni S, Rader J, Zhang F, et al. Cyclooxygenase-2 is overexpressed in human cervical cancer. Clin Cancer Res 2001;7:429–34.
Zeidler R, Csanady M, Gires O, Lang S, Schmitt B, Wollenberg B. Tumor cell-derived prostaglandin E2 inhibits monocyte function by interfering with CCR5 and Mac-1. FASEB J 2000;14:661–8.
Hsieh C, Macatonia S, Tripp C, Wolf S, O’Garra A, Murphy K. Development of Th1 CD4+ T cells through IL-12 produced by Listeria-induced macrophages. Science 1993;260:547–50.
Gately M, Wolitzky A, Quinn P, Chizzonite R. Regulation of human cytolytic lymphocyte responses by interleukin-12. Cell Immunol 1992;143:127–42.
Mehrota P, Win D, Crim J, Mostowski H, Siegel J. Effects of IL-12 on the generation of cytotoxic activity in human CD8+ T lymphocytes. J Immunol 1993;151:2444–52.
Coughlin C, Salhany K, Gee M, et al. Tumor cell responses to IFN-gamma affect tumorigenicity and response to IL-12 therapy and antiangiogenesis. Immunity 1998;9:25–34.
Bellone G, Turletti A, Artusio E, et al. Tumor-associated transforming growth factor-beta and interleukin-10 contribute to a systemic Th2 immune phenotype in pancreatic carcinoma patients. Am J Pathol 1999;155:537–47.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Smyth, G.P., Stapleton, P.P., Barden, C.B. et al. Renal Cell Carcinoma Induces Prostaglandin E2 and T-Helper Type 2 Cytokine Production in Peripheral Blood Mononuclear Cells. Ann Surg Oncol 10, 455–462 (2003). https://doi.org/10.1245/ASO.2003.06.036
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1245/ASO.2003.06.036