Abstract
Background
The prognosis of unresectable gastric cancer is poor. Chemotherapy occasionally converts an initially unresectable gastric cancer to a resectable cancer.
Methods
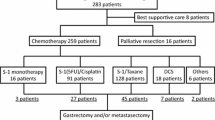
The responses of noncurative factors to initial chemotherapy and the outcomes of additional (conversion) surgery were retrospectively evaluated in 151 patients with unresectable gastric cancer receiving combination chemotherapy with S-1 plus cisplatin or paclitaxel from February 2003 to December 2013.
Results
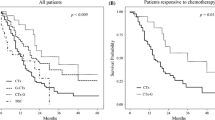
Forty (26 %) of 151 patients underwent conversion surgery. After chemotherapy, R0 resection was accomplished in 32 patients (80 %). The 5-year overall survival (OS) rate among the 40 patients who underwent conversion surgery was 43 % (median survival time, 53 months). The 5-year OS rate in the 111 patients treated with chemotherapy alone was 1 % (median survival time, 14 months). Patients who underwent conversion surgery had significantly longer OS times than patients who underwent chemotherapy alone (P < 0.01). The 5-year OS rate among patients who underwent R0 resection was 49 % (median survival time, 62 months). Patients who underwent R0 resection had significantly longer OS times than those who underwent R1 and R2 resection (P = 0.03). Among patients who underwent conversion surgery, multivariate Cox regression analysis showed that one noncurative factor (odds ratio 0.49; 95 % confidence interval 0.28–0.88; P = 0.02) and R0 resection (odds ratio 0.52; 95 % confidence interval 0.28–0.95; P = 0.03) were significant independent predictors for favorable OS.
Conclusions
Patients with unresectable gastric cancer initially exhibiting one noncurative factor may obtain a survival benefit from chemotherapy and subsequent curative surgery.


Similar content being viewed by others
References
Crew KD, Neugut AI. Epidemiology of gastric cancer. World J Gastroenterol. 2006;12:354–62.
Jemal A, Bray F, Center MM, Ferlay J, Ward E, Forman D. Global cancer statistics. CA Cancer J Clin. 2011;61:69–90.
MacDonald JS, Schein PS, Woolley PV, et al. 5-Fluorouracil, doxorubicin, and mitomycin (FAM) combination chemotherapy for advanced gastric cancer. Ann Intern Med. 1980;93:533–6.
Findlay M, Cunningham D, Norman A, et al. A phase II study in advanced gastro-esophageal cancer using epirubicin and cisplatin in combination with continuous infusion 5-fluorouracil (ECF). Ann Oncol. 1994;5:609–16.
Ohtsu A, Shimada Y, Shirao K, et al. Randomized phase III trial of fluorouracil alone versus fluorouracil plus cisplatin versus uracil and tegafur plus mitomycin in patients with unresectable, advanced gastric cancer: the Japan Clinical Oncology Group Study (JCOG9205). J Clin Oncol. 2003;21:54–9.
Koizumi W, Narahara H, Hara T, et al. S-1 plus cisplatin versus S-1 alone for first-line treatment of advanced gastric cancer (SPIRITS trial): a phase III trial. Lancet Oncol. 2008;9:215–21.
Mochiki E, Ogata K, Ohno T, et al. Phase II multi-institutional prospective randomised trial comparing S-1 + paclitaxel with S-1 + cisplatin in patients with unresectable and/or recurrent advanced gastric cancer. Br J Cancer. 2012;107:31–6.
Okabe H, Ueda S, Obama K, Hosogi H, Sakai Y. Induction chemotherapy with S-1 plus cisplatin followed by surgery for treatment of gastric cancer with peritoneal dissemination. Ann Surg Oncol. 2009;16:3227–36.
Wang Y, Yu YY, Li W, et al. A phase II trial of Xeloda and oxaliplatin (XELOX) neo-adjuvant chemotherapy followed by surgery for advanced gastric cancer patients with para-aortic lymph node metastasis. Cancer Chemother Pharmacol. 2014;73:1155–61.
Kodera Y, Ito S, Mochizuki Y, et al. Long-term follow up of patients who were positive for peritoneal lavage cytology: final report from the CCOG0301 study. Gastric Cancer. 2012;15:335–7.
Nakajima T, Ota K, Ishihara S, et al. Combined intensive chemotherapy and radical surgery for incurable gastric cancer. Ann Surg Oncol. 1997;4:203–8.
Suzuki T, Tanabe K, Taomoto J, et al. Preliminary trial of adjuvant surgery for advanced gastric cancer. Oncol Lett. 2010;1:743–7.
Sobin LH, Gospodarowicz MK, Wittekind CW. TNM classification of malignant tumours. 7th ed. New York: Wiley-Blackwell; 2009.
Japanese Gastric Cancer Association. Japanese classification of gastric carcinoma: third English edition. Gastric Cancer. 2011;14:101–12.
Japanese Gastric Cancer Association. Japanese gastric cancer treatment guidelines, 2010 (ver. 3). Gastric Cancer. 2011;14:113–23.
Koizumi W, Tanabe S, Saigenji K, et al. Phase I/II study of S-1 combined with cisplatin in patients with advanced gastric cancer. Br J Cancer. 2003;89:2207–12.
Chua YJ, Cunningham D. The UK NCRI MAGIC trial of perioperative chemotherapy in resectable gastric cancer: implications for clinical practice. Ann Surg Oncol. 2007;14:2687–90.
Iwasaki Y, Sasako M, Yamamoto S, et al. Phase II study of preoperative chemotherapy with S-1 and cisplatin followed by gastrectomy for clinically resectable type 4 and large type 3 gastric cancers (JCOG0210). J Surg Oncol. 2013;107:741–5.
Yoshida K, Yamaguchi K, Okumura N, et al. The roles of surgical oncologists in the new era: minimally invasive surgery for early gastric cancer and adjuvant surgery for metastatic gastric cancer. Pathobiology. 2011;78:343–52.
Disclosure
The authors declare no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Fukuchi, M., Ishiguro, T., Ogata, K. et al. Prognostic Role of Conversion Surgery for Unresectable Gastric Cancer. Ann Surg Oncol 22, 3618–3624 (2015). https://doi.org/10.1245/s10434-015-4422-6
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-015-4422-6