Abstract
Background
Alterations of the p16 gene are common in human cancers, but their roles in thyroid cancers have not been clearly defined. The aim of the present study was to investigate the clinicopathological roles of the p16 gene in papillary thyroid carcinoma (PTC).
Methods
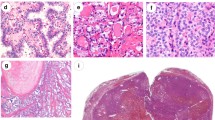
p16 gene alterations were investigated in 44 patients with PTC (9 men, 35 women) by immunohistochemistry, reverse transcriptase–polymerase chain reaction and methylation-specific polymerase chain reaction. The findings were correlated with their clinicopathological features.
Results
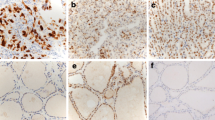
p16 protein expression, mRNA alterations, and promoter methylation were detected in 89% (n = 39), 77% (n = 33), and 41% (n = 18) of patients with PTC, respectively. There was no marked relationship between p16 protein expression, mRNA alteration, and promoter methylation. In follicular variant of PTC (FVPTC), there was a frequent lack of p16 protein expression and promoter methylation. PTCs showing p16 promoter methylation were often associated with a high AMES (age, metastasis to distant sites, extrathyroidal invasion, size) risk group and advanced pTNM (tumor–lymph node–metastasis) stages.
Conclusions
p16 gene alterations are common and correlate with histological features and biological aggressiveness in PTC, suggesting that they might play an important role in its pathogenesis.




Similar content being viewed by others
References
Larsen CJ. Contribution of the dual coding capacity of the p16INK4a/MTS1/CDKN2 locus to human malignancies. Prog Cell Cycle Res 1997; 3:109–24
Oue N, Mitani Y, Motoshita J, et al. Accumulation of DNA methylation is associated with tumour stage in gastric cancer. Cancer 2006; 106:1250–9
Mihic-Probst D, Mnich CD, Oberholzer PA, et al. p16 expression in primary malignant melanoma is associated with prognosis and lymph node status. Int J Cancer 2006; 118:2262–8
Sabah M, Cummins R, Leader M, Kay E. Loss of p16INK4A expression is associated with allelic imbalance/loss of heterozygosity of chromosome 9 p16 in microdissected synovial sarcomas. Virchows Arch 2005; 447:842–8
Tsirigotis P, Pappa V, Labropoulos S, et al. Mutational and methylation analysis of the cyclin-dependent kinase 4 inhibitor (p16INK4A) gene in chronic lymphocytic leukemia. Eur J Haematol 2006; 76:230–6
Auerkari EI. Methylation of tumour suppressor genes p16 (INK4a), p16 (Kip1) and E-cadherin in carcinogenesis. Oral Oncol 2006; 42:5–13
Lam AK, Lo CY, Lam KS. Papillary carcinoma of thyroid: a 30-yr clinicopathological review of the histological variants. Endocr Pathol 2005; 16:323–30
Ciampi R, Nikiforov YE. Alterations of the BRAF gene in thyroid tumours. Endocr Pathol 2005; 16:163–72
Lam KY, Lo CY, Leung PS. High prevalence of RET proto-oncogene activation (RET/PTC) in papillary thyroid carcinomas. Eur J Endocrinol 2002; 147:741–5
Jarzab B, Wloch J, Wiench M. Molecular changes in thyroid neoplasia. Folia Histochem Cytobiol 2001; 39(Suppl 2):26–7
LiVolsi VA, Albores-Saavedra J, Asa SL, et al. Papillary carcinoma. In: DeLellis RA, Llyod RV, Heitz PU, Eng C, eds. World Health Organization Classification of Tumours: Pathology and Genetics—Tumours of Endocrine Organs. Lyon: IARC, 2004:57–66
Cady B, Sedgwick MD, Meissner WA, Wool MS, Salzman FA, Werber J. Risk factor analysis in differentiated thyroid cancer. Cancer 1979; 43:810–20
Jukkola A, Bloigu R, Ebeling T, Salmela P, Blanco G. Prognostic factors in differentiated thyroid carcinomas and their implications for current staging classifications. Endocr Relat Cancer 2004; 11:571–9
Lo CY, Chan WF, Lam KY, Wan KY. Optimizing the treatment of AMES high-risk papillary thyroid carcinoma. World J Surg 2004; 28:1103–9
Armes JE, Lourie R, de Silva M, et al. Abnormalities of the RB1 pathway in ovarian serous papillary carcinoma as determined by overexpression of the p16 (INK4A) protein. Int J Gynecol Pathol 2005; 24:363–8
Cui X, Shirai Y, Wakai T, Yokoyama N, Hirano S, Hatakeyama K. Aberrant expression of pRb and p16 (INK4), alone or in combination, indicates poor outcome after resection in patients with colorectal carcinoma. Hum Pathol 2004; 35:1189–95
O’Neill CJ, McCluggage WG. p16 expression in the female genital tract and its value in diagnosis. Adv Anat Pathol 2006; 13:8–15
Dray M, Russell P, Dalrymple C, et al. p16 (INK4a) as a complementary marker of high-grade intraepithelial lesions of the uterine cervix. I: experience with squamous lesions in 189 consecutive cervical biopsies. Pathology 2005; 37:112–24
Boltze C, Zack S, Quednow C, Bettge S, Roessner A, Schneider-Stock R. Hypermethylation of the CDKN2/ p16INK4A promotor in thyroid carcinogenesis. Pathol Res Pract 2003; 199:399–404
Ferenc T, Lewinski A, Lange D, et al. Analysis of p161NK4A protein expression in follicular thyroid tumours. Pol J Pathol 2004; 55:143–8
Schagdarsurengin U, Gimm O, Hoang-Vu C, Dralle H, Pfeifer GP, Dammann R. Frequent epigenetic silencing of the CpG island promoter of RASSF1A in thyroid carcinoma. Cancer Res 2002; 62:3698–701
Elisei R, Shiohara M, Koeffler HP, Fagin JA. Genetic and epigenetic alterations of the cyclin-dependent kinase inhibitors p16INK4b and p16INK4a in human thyroid carcinoma cell lines and primary thyroid carcinomas. Cancer 1998; 83:2185–93
Schulte KM, Staudt S, Niederacher D, et al. Rare loss of heterozygosity of the MTS1 and MTS2 tumor suppressor genes in differentiated human thyroid cancer. Horm Metab Res 1998; 30:549–54
Ivan M, Wynford-Thomas D, Jones CJ. Abnormalities of the p16INK4A gene in thyroid cancer cell lines. Eur J Cancer 1996; 32A:2369–70
Calabro V, Strazzullo M, La Mantia G, et al. Status and expression of the p16INK4 gene in human thyroid tumors and thyroid-tumor cell lines. Int J Cancer 1996; 67:29–34
Yane K, Konishi N, Kitahori Y, et al. Lack of p16/CDKN2 alterations in thyroid carcinomas. Cancer Lett 1996; 101:85–92
Jones CJ, Shaw JJ, Wyllie FS, Gaillard N, Schlumberger M, Wynford-Thomas D. High frequency deletion of the tumour suppressor gene p16INK4a (MTS1) in human thyroid cancer cell lines. Mol Cell Endocrinol 1996; 116:115–9
Yane K, Konishi N, Kitahori Y, Hiasa Y, et al. Lack of p16/CDKN2 alterations in thyroid carcinomas. Cancer Lett 1996; 101:85–92
Lang BH, Lo CY, Chan WF, Lam AK, Wan KY. Classical and follicular variant of papillary thyroid carcinoma: a comparative study on clinicopathologic features and long-term Outcome. World J Surg 2006; 30:752–8
Lloyd RV, Erickson LA, Casey MB, et al. Observer variation in the diagnosis of follicular variant of papillary thyroid carcinoma. Am J Surg Pathol 2004; 28:1336–40
Lo CY, Lam KY, Leung PP, Luk JM. High prevalence of cyclooxygenase 2 expression in papillary thyroid carcinoma. Eur J Endocrinol 2005; 152:545–50
Lam AK, Montone KT, Nolan KA, Livolsi VA. Ret oncogene activation in papillary thyroid carcinoma: prevalence and implication on the histological parameters. Hum Pathol 1998; 29:565–8
Acknowledgments
Supported by CRCG Seeding Grant 2003–2004 of University Research Committee. The authors acknowledge the staff of Departments of Surgery and Pathology in Queen Mary Hospital in facilitating the project.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lam, A.K.Y., Lo, C.Y., Leung, P. et al. Clinicopathological Roles of Alterations of Tumor Suppressor Gene p16 in Papillary Thyroid Carcinoma. Ann Surg Oncol 14, 1772–1779 (2007). https://doi.org/10.1245/s10434-006-9280-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-006-9280-9