-
PDF
- Split View
-
Views
-
Cite
Cite
Terry J. Smith, Gregory D. Sempowski, Charles S. Berenson, H. James Cao, Hwai-Shi Wang, Richard P. Phipps, Human Thyroid Fibroblasts Exhibit a Distinctive Phenotype in Culture: Characteristic Ganglioside Profile and Functional CD40 Expression, Endocrinology, Volume 138, Issue 12, December 1997, Pages 5576–5588, https://doi.org/10.1210/endo.138.12.5563
Close - Share Icon Share
Abstract
Fibroblasts from different regions of the human body exhibit substantial phenotypic diversity, some of which relates to the capacity for cross-talk with cells of the immune system. We examine, for the first time, thyroid fibroblast biology in culture. Thyroid explants were placed in culture, and fibroblasts were outgrown and serially passaged. These fibroblasts take on a morphology in culture resembling cells from other anatomic regions. When treated with PGE2, they assume a stellate morphology similar to that of prostanoid-treated orbital fibroblasts. The ganglioside profile exhibited by these cells is distinct from that observed previously in orbital and dermal fibroblasts. They uniformly express Thy-1, a surface glycoprotein. Messenger RNA encoding CD40, a surface receptor found on bone marrow-derived cells, and CD40 protein were expressed constitutively at low levels. Interferon-γ (500 U/ml) treatment for 48–72 h resulted in high levels of surface HLA-DR and CD40 display. When CD40 is engaged with CD40 ligand (CD40L), nuclear factor-κB binding activity is up-regulated as is interleukin (IL)-6 and IL-8 expression. IL-1β treatment up-regulates the expression of IL-1α, IL-1β, and PGE2. These observations suggest that thyroid fibroblasts possess the molecular machinery necessary for cross-talk with immunocompetent cells such as lymphocytes and mast cells through the CD40/CD40L complex, as well as through classic cytokine networks, and to participate potentially in the inflammatory response of the thyroid gland.
SUBSTANTIAL evidence supports the concept of fibroblast diversity. Clear-cut differences have been demonstrated between fibroblasts derived from distant anatomic sites with regard to morphology (1) and morphogenic responses to small molecules (2, 3), and the expression and induction of several genes and their products (4–6), receptors (7), and extracellular matrix (1, 8). Moreover, in some organs, such as the lung, spleen, and orbit, fibroblasts have been successfully sorted into subsets and appear to represent heterogeneous populations of cells with differing functional capacities (9–11). Phenotypic diversity among human fibroblasts may underlie many of the peculiarities, including susceptibility to disease, displayed by specific tissues. The molecular environment confronting parenchymal cells is determined, in part, by neighboring cell types, such as endothelial and vascular smooth muscle cells and fibroblasts. If fibroblasts in each organ are characteristic and distinct, it would appear crucial that insight into the nature of each fibroblast population be gained if the contribution of fibroblast/parenchymal cell interactions is to be understood. Moreover, the potential cross-talk between fibroblasts and components of the immune system may prove an important basis for recruitment of bone marrow-derived cells and the initiation of tissue remodeling.
The thyroid gland serves a unique biological function as the principal iodine-concentrating tissue in mammals. The vast majority of investigations concerning the thyroid have focused on function and disease directly related to epithelial cells. This gland is frequently affected by inflammatory autoimmune diseases, and some of this pathology results in substantial tissue remodeling. Thus it should be of considerable interest to characterize the fibroblast population residing in thyroid tissue with regard to its role in supporting normal glandular function and potentially its contribution to disease susceptibility. Unfortunately, little is currently known about thyroid fibroblasts.
We have defined a number of attributes characteristic of human orbital fibroblasts because of their putative role in the pathogenesis of thyroid-associated ophthalmopathy (TAO). Those cells are particularly susceptible to several actions of inflammatory cytokines. For instance, the T lymphocyte product, leukoregulin, can induce the synthesis of hyaluronan (8) and the expression of plasminogen activator inhibitor type-1 (6) in orbital fibroblasts. The magnitude of these actions is substantially greater than that observed in dermal fibroblasts (6, 8). We have reported that orbital fibroblasts exhibit an exaggerated induction of PG endoperoxide H synthase-2 (PGHS-2), the inflammatory cyclooxygenase (12), and others have shown a similar inducibility of PGHS-2 in synoviocytes from patients with rheumatoid arthritis (13). We hypothesize that this exaggerated inducibility of PGHS-2 in orbital and synovial fibroblasts represents the molecular basis for the inflammatory responses observed in TAO and rheumatoid arthritis, respectively. We have also reported that orbital fibroblasts express a profile of gangliosides that differs considerably from that observed in dermal fibroblasts (14). These glycosphingolipids play important roles in cell recognition and signaling (15). Thus differences in ganglioside expression imply that fibroblasts from distant anatomic regions may engage in interplay with their molecular environment that is cell-specific.
A major conduit for the cross-talk between T and B lymphocytes was identified with the discovery of CD40 and its natural ligand, gp39, also known as CD40L (16). B lymphocytes are triggered by CD40 engagement, often in association with interleukin (IL)-4 (17). This activation represents an important stimulus for B cell proliferation, Ig production, and isotype switching (18–20). CD40 is also expressed by non-B lymphocyte lineage cells such as monocytes, dendritic, Langerhans, epithelial, endothelial cells, and keratinocytes (16, 21–26). Recently, CD40 has been found on synoviocytes and certain fibroblasts from lung and periodontal tissue (27). In these fibroblasts, CD40 is up-regulated by interferon-γ. The biological implications of CD40 expression by fibroblasts is uncertain but might represent an important mechanism through which these cells cross-talk with T lymphocytes and other CD40L-displaying cells.
In this study, we examine, for the first time, several aspects of the human thyroid fibroblast phenotype in vitro. We report here that these cells undergo the dramatic changes in morphology in response to exogenous PGE2 that were observed previously in orbital fibroblasts (2, 3). They express a profile of gangliosides that sets them apart from orbital and dermal fibroblasts. Thyroid fibroblasts express CD40 and, when exposed to CD40L, express high levels of IL-6 and IL-8. These fibroblasts also respond to exogenous IL-1β with regard to an up-regulation of IL-1α and IL-1β expression and the production of PGE2. Thus they may play an important role in the pathogenesis of inflammatory diseases of the thyroid by virtue of their abilities to cross-talk with immunocompetent cells. This can be accomplished directly through the CD40/CD40L signaling conduit as well as through multiple, classic cytokine pathways.
Materials and Methods
Materials
PGE2 was purchased from Sigma (St. Louis, MO). IL-1β was from Biosource (Camarillo, CA), and human recombinant interferon-γ was obtained from Genzyme (Cambridge, MA). Eagle’s medium, FBS, and other culture reagents were supplied by GIBCO (Grand Island, NY), from Hyclone (Logan, UT), or from Life Technologies (Gaithersburg, MD).
Cell culture
Thyroid fibroblasts were obtained by mincing thyroid tissue explants obtained at thyroidectomy for Graves’ disease or for neoplastic disease, where normal tissue was excised. Orbital fibroblasts were obtained during decompressive surgery for TAO. Dermal fibroblasts were obtained by punch biopsy of normal appearing skin. These activities have been approved by the Institutional Review Board of the Albany Medical College. Explants were cut into small pieces and allowed to attach to the bottom of 60-mm plastic culture plates as described previously (28). They were covered with Eagle’s medium supplemented with FBS (10%), glutamine, and antibiotics and maintained in a 5% CO2, humidified incubator. When fibroblasts emerging from the tissue became numerous, the explants were removed carefully so as to not leave any intact thyroid tissue, which would include thyrocytes and endothelial cells, on the plate. Fibroblast monolayers were disrupted with trypsin/EDTA treatment. These cells exhibit morphologies that are fibroblast-like. The cultures were assessed for contaminating cells by immunostaining for factor VIII, smooth muscle actin, and thyroglobulin. For experiments, cultures were passaged approximately every 2 weeks by gentle treatment with trypsin/EDTA. Some of the cellular material was stored at −70 C. Cells were used between the second and 12th passage.
Phase-contrast microscopy
Fibroblasts were inoculated on sterile 22 × 22 mm glass coverslips at a seeding density of about 104 cells per coverslip. Cells were allowed to attach for 1 day before the initiation of experiments. Cultures were shifted to medium supplemented with 1% FBS without or with the test compounds. After treatment, monolayers were fixed in 2% para-formaldehyde in PBS (pH 8.0). The coverslips were washed twice and mounted on glass slides with glycerol/PBS (1:1). Microscopy was performed with a Nikon Labophot (Nikon Corp., Melville, NY) equipped with a 35-mm camera. Photographs were taken on Kodak Tri-Max 400 film (Eastman Kodak, Rochester, NY). The final magnification was 400×.
Flow cytometric analysis of surface HLA-DR, Thy-1, and CD40 expression by thyroid fibroblasts
Fibroblasts were prepared for flow cytometry by washing dispersed fibroblasts in suspension in PBS with 0.1% sodium azide and 1% BSA. Cells were incubated with either antihuman HLA-DR [L243, 100μ l of hybridoma supernatant, American Type Culture Collection (ATCC), Rockville, MD], anti-Thy-1 (F15–42-1, kindly provided by Dr. John W. Fabre, University of London, London, UK) or anti-CD40 monoclonal antibody (G28–5, 100 μl of hybridoma supernatant, ATCC) for 30 min on ice. After washing, fluorescein isothiocyanate-conjugated (FITC) goat antimouse Ig (1:100 dilution, Jackson Immunoreasearch, West Grove, PA) was added for 30 min on ice. Samples stained only with the secondary antibody served as negative control. Once washed, the cells were resuspended in PBS with 0.1% sodium azide and 1% BSA and were analyzed on a Coulter Elite flow cytometer (Coulter Corp., Hialeah, FL). We have demonstrated that trypsin treatment, used to disrupt fibroblast monolayers, fails to alter the detection of surface HLA-DR, Thy-1, or CD40. Viable cells were gated on the basis of forward light scatter, and the data were analyzed with the Cytologo software program (Coulter). All studies were performed at least three times with representative results being presented.
Immunofluoresence analysis of HLA-DR, CD40, and Thy-1 expression in thyroid fibroblasts
Indirect immunofluorescence analysis of HLA-DR, CD40, and Thy-1 expression in thyroid fibroblasts was achieved by inoculating cells in chamber slides (Nunc, Naperville, IL). They were stained under similar conditions as those examined by flow cytometry with the exception that they were conducted at room temperature. Slides were removed from the chambers, coverslips were applied, and cell layers were visualized under an Olympus BX 40 microscope (Olympus America, Lake Success, NY) equipped with a UPlan-FL (Ph2) 40 × objective.
Ganglioside analysis
Gangliosides were purified from fibroblasts as reported previously (14, 29). Briefly, monolayers were disrupted mechanically and cells rinsed in 0.31 m pentaerythritol and centrifuged. Pelleted cells were continuously inverted for 18 h in chloroform-methanol (1:1, all vol/vol) in glass tubes. Total lipid extracts were rotary evaporated to dryness and applied to diethylaminoethyl-Sephadex A-25 columns (bed vol, 3 ml) in chloroform-methanol-water (30:60:8). Acidic lipids were eluted with chloroform-methanol: 0.8 n sodium acetate (30:60:8), evaporated to dryness, and solubilized in 0.1 n NaOH. Samples were then neutralized to pH 4–5 with 0.1 n HCl and desalted by reverse phase column chromatography (Sep-Pak, Waters Associates, Waltham, MA). Lipids were recovered and applied to Iatrobeads 6RS-8060 (Iatron Laboratories, Tokyo). Less polar lipids were eluted with chloroform-methanol (85:15). Gangliosides were eluted with chloroform-methanol (1:2) and dried, and purity was confirmed on thin layer chromatograms sprayed with resorcinol.
Two-dimensional TLC was performed on each sample, containing 3–5 μg ganglioside-bound sialic acid. Standard markers of murine brain ganglioside were included in each dimension for reference. The solvent system for the first dimension TLC was chloroform-methanol-0.25% KCl (50:45:10), and that of the second was chloroform-methanol-2.5 n NH3 in aqueous 0.025% KCl (50:40:10). After the chromatogram was dried, gangliosides were visualized by spraying with resorcinol reagent and heating uniformly to 92 C. Relative quantification of individual ganglioside peaks was performed (30) with a two-dimensional analytical scanning system (Molecular Dynamics, Sunnyvale, CA). To compensate for differences in the quantity of individual samples, individual peaks are expressed as relative percentages of the total signal. Each chromatogram was loaded with 3–5μ g ganglioside-bound sialic acid. Because of the small quantities of materials, significance of differences in the abundance of the minor peaks could not be assessed. Thus, differences in peaks containing less than 6% of the total (i.e. <0.4 μg sialic acid) were not assigned levels of significance. Mean relative percentages of homologous peaks contained in Table 1 are expressed as the mean ± sem. Statistical differences between groups was determined using Student’s one-tailed t test and confirmed by post hoc ANOVA using Scheffe’s F test.
RT-PCR
Fibroblasts were either treated with nothing or with interferon-γ (500 U/ml) for 72 h, after which the monolayers were scraped with a rubber policeman, and total cellular RNA was extracted with Tri-Reagent (1 ml per 106 cells, Molecular Research Center, Cincinnati, OH) following the manufacturer’s protocol. RNA was solubilized in nuclease-free water, concentrations were determined spectrophotometrically, and 5 μg of each sample were subjected to reverse transcription with Moloney murine leukemia virus reverse transcriptase (200 U/reaction, Life Technologies) and oligo-dT primer (Pharmacia, Piscataway, NJ) as described previously (31). RT was withheld from replicate samples that served as negative controls (RT−). PCR reactions for CD40 and β-actin included 5 μl of the above generated complementary DNA (cDNA), PCR buffer (5 μl of 10X stock, Boehringer Mannheim, Indianapolis, IN), deoxynucleotides (1μ m each) primers (1 μm each), Taq polymerase (2.5 U, Boehringer Mannheim) in a total volume of 50 μl. The CD40 primer sequences were 3′-CGTACAGTGCCAGCCTTCTTC and 5′-ATGGTTCGTCTGCCTCTG CAG yielding a 330 bp product and the β-actin primers were 3′-CTCCTTAATGTCACG CACGATTTC and 5′-GTGGGGCGCCCAGGCACCA, which generated a 539-bp product. Samples underwent 30 cycles of amplification in a M.J. Research PTC-200 DNA Engine (Watertown, MA), and each cycle included denaturation at 94 C for 30 sec, annealing at 63 C for 30 sec, and primer extension at 72 C for 60 sec. The products were electrophoresed on 2% agarose gels and visualized by ethidium bromide staining. A 100-bp ladder (Life Technologies) was used for size determination.
Electrophoretic mobility shift assay (EMSA) for nuclear factor (NF)-κB activity
Fibroblasts were allowed to proliferate to confluence in 100-mm culture plates covered in medium supplemented with 10% FBS. They were treated with interferon-γ (500 U/ml) or nothing for 72 h. The cultures were then washed extensively and incubated overnight with medium with 1% FBS. Cells were then stimulated for 2 h at 37 C with medium alone, control insect cell membranes containing glutathione S-transferase (GST) or insect cell membranes containing human CD40L, generously provided by Dr. Marilyn Kehry (Boehringer Ingelheim, Ridgefield, CT) prepared as described previously (32). Nuclear extracts were prepared as described (33), and proteins were normalized using a BCA protein assay (Pierce, Rockford, IL). To assess NF-κB binding activity, 2.5 μg of the extract were incubated with 32P-labeled double- stranded oligonucleotide probe representing the consensus sequence for the NF-κB-binding site following the supplier’s instructions (Promega, Madison, WI). Samples were electrophoresed on 4% polyacrylamide gels, and the DNA/protein complexes were visualized by autoradiography. Supershift assays were performed by preincubating the nuclear extracts with anti-p65 antibodies or control rabbit IgG (Santa Cruz Biotechnology, Santa Cruz, CA).
Cytokine expression in response to CD40 engagement with CD40L
Thyroid fibroblasts were seeded into 96-well tissue culture plates (5000 cells per well) and once established were pretreated with nothing or interferon-γ (500 U/ml) for 72 h in medium supplemented with 10% FBS. Monolayers were washed extensively with fresh medium and then stimulated as triplicate wells for 2 h with medium alone, control insect cell membranes containing GST, or human CD40L. Cell-free culture media were harvested, and the levels of IL-6 and IL-8 secreted by the monolayers were determined by specific enzyme-linked immunosorbent assay (ELISA). These were diluted with sample buffer so that the cytokine concentrations would fall in the linear range of the assays. Neither cytokine was detectable in fresh medium without or with the various additives.
With regard to the IL-6 assay, Immulon 3 flat bottom 96-well plates (Dynatech, Chantilly, VA) were coated overnight at 4 C with 2μ g/ml antihuman IL-6 (capture antibody, Pharmingen, San Diego, CA) diluted in 0.1 m sodium bicarbonate (pH 8.2). The plates were then washed three times with PBS/Tween (0.5 ml Tween-20 per liter PBS containing 0.1% sodium azide). Plates were blocked with ELISA buffer (PBS containing 5% BSA (wt/vol), 5% calf serum (vol/vol) and 0.1% sodium azide) for 2 h at room temperature. Plates were washed three times with PBS/Tween. Samples and IL-6 standards (Pharmingen) were incubated in the plates for 2 h at 37 C followed by three washes with PBS/Tween. Detection antibody (2 μg/ml biotinylated antihuman IL-6, Pharmingen) was diluted in ELISA buffer and incubated in the plates for 1 h at room temperature. Plates were washed three times and incubated 1 h at 37 C with streptavidin-alkaline phosphatase (1:500 dilution in ELISA buffer, Southern Biotechnologies, Birmingham, AL). After three washes, plates were incubated with p-nitrophenyl phosphate (1 mg/ml) in DEA buffer (10 μm MgCl2, 30 μm NaN3, and 1% (vol/vol) diethanolamine, pH 9.0). The reaction was stopped with 3 n NaOH, and the optical density of each well was determined at 405 nm with a Dynatech plate reader.
The assay for IL-8 resembled that for IL-6 with the following exceptions. The capture antibody (anti-IL-6, Endogen, Cambridge, MA) was at a concentration of 1 μg/ml. The IL-8 standard was from Biosource, and the biotinylated detection antibody (antihuman IL-8, Endogen) was at a concentration of 0.5 μg/ml. Detection antibody was added 15 min after the addition of sample to the plates and incubated for 1 h at room temperature. All studies were performed at least three times, and results from representative experiments are shown.
Quantification of IL-1α and IL-1β expression and PGE2 production by thyroid fibroblasts
Thyroid fibroblasts for the IL-1α and IL-1β assays were cultured in 24-well plates to confluence in medium supplemented with 10% FBS. Cultures were then shifted to medium with 1% FBS for the next 16 h. IL-1α or IL-1β were added to some of the wells at the times indicated in the figures. After treatment, monolayers were washed extensively with PBS and cells were taken up in lysis buffer containing 20 mm Tris-HCl (pH 7.5), 15 mm 3-[(3-chloamidopropyl)dimethylammonio]-1-propanesulfonic acid, 1 mm EDTA, 10 μm phenylmethylsulfonyl fluoride, and 10 U/ml soybean trypsin inhibitor. Cellular protein (10 μg) was subjected to assay using either IL-1α or IL-1β ELISA kits (Immunotech, Westbrook, ME) according to the instructions of the manufacturer. We have determined that the exogenous IL-1β used in the treatment of cultures does not contribute to the cytokine measured in the cell lysates. With regard to quantification of PGE2 production, confluent fibroblasts in 24-well plates were shifted to medium with 1% FBS for 16 h, and then some received IL-1β (10 ng/ml) for 16 h. Cultures were then shifted to PBS (150 μl/well) for the final 30 min of incubation in the absence or presence of the cytokine. PBS was collected and subjected to RIA as described previously (12) for PGE2 content.
Results
Human thyroid fibroblasts exhibit typical fibroblast-like morphology in culture and become stellate in response to PGE2
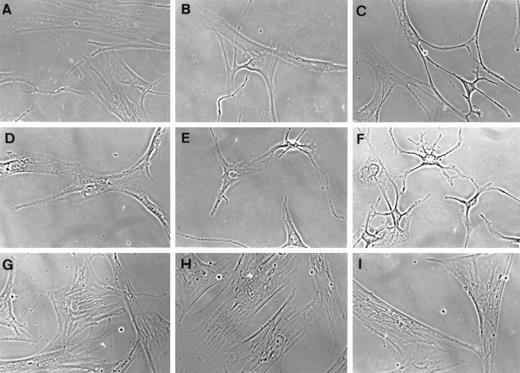
Thyroid fibroblasts from patients with Graves’ disease or derived from histologically normal thyroid tissue proliferate in culture as monolayers. They exhibit many of the characteristics of well differentiated cells such as contact inhibition. They proliferate for a finite number of doublings before becoming senescent. Thyroid fibroblasts express vimentin as assessed by immunostaining. They do not, however, exhibit staining for S-100, factor VIII, MAC 387, smooth muscle actin, or CD34. Moreover, these fibroblasts fail to express throglobulin. Thus the cultures are not contaminated with endothelial, smooth muscle, or thyroid epithelial cells. Cells appear heterogeneous with regard to their native morphologies. The fibroblasts typically have few cytoplasmic projections under control (untreated) conditions (Fig. 1, panel A). When exposed to PGE2 (100 nm, panel B) or 8-br-cAMP (1 mm, panel C) they become stellate. This response is similar to that reported previously in orbital fibroblasts (Fig. 1, panels E and F) (2, 3). In contrast, abdominal wall fibroblasts fail to exhibit an altered cell morphology in response to either compound (panels H and I). Thus the vulnerability of the cytoarchitecture previously recognized in orbital fibroblasts is shared by thyroid cells.
Phase-contrast micrographs of human thyroid (panels A–C), orbital (panels D–F), and dermal (panels G–I) fibroblasts in primary culture. Cells were subpassaged on glass coverslips and allowed to attach in medium supplemented with 10% FBS. They were then shifted to medium containing 1% FBS for 6 h (panels A, D, and G) supplemented with PGE2 (100 nm, panels B, E, and H) or 8-br-cAMP (1 mm, panels C, F, and I).
Thyroid fibroblast gangliosides
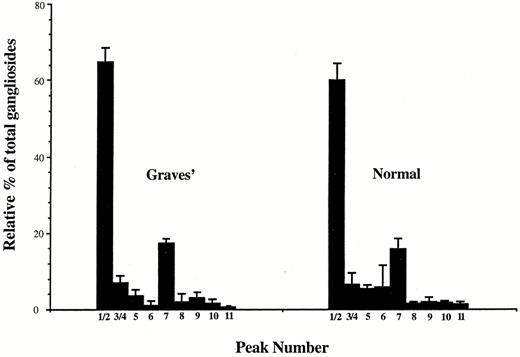
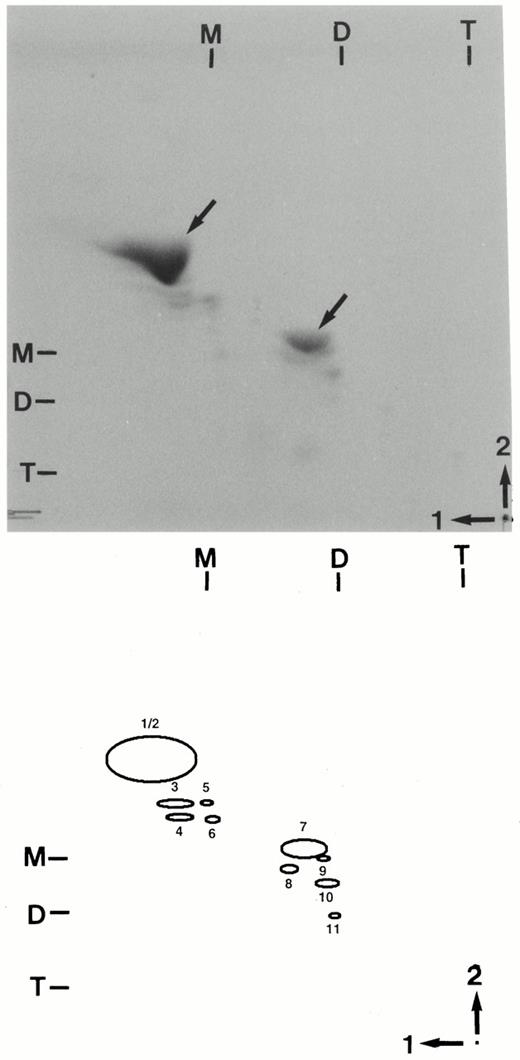
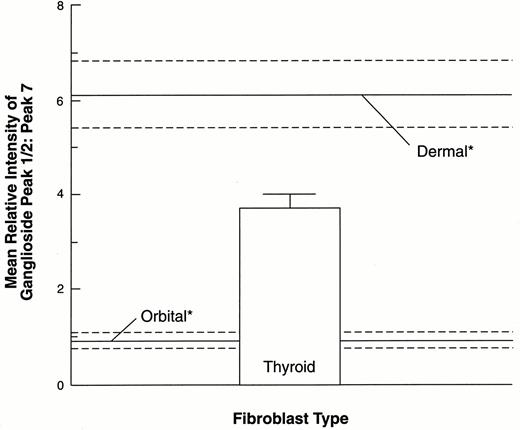
Gangliosides expressed by thyroid fibroblasts were extracted from cultures derived from a total of ten donors: six from patients with Graves’ disease and four strains from apparently normal thyroid tissue. Densitometric measurements of the individual peaks from these culture strains are presented in Fig. 2. Each chromatogram contains 10 to 11 discrete, resorcinol-positive peaks, occupying the same TLC positions, relative to the standards, as gangliosides from orbital and dermal fibroblasts (14). A typical chromatogram is presented in Fig. 3. Some variations occur between individuals with regard to specific ganglioside peaks resolvable on thin layer chromatograms. In some samples, it is not possible to accurately distinguish borders between peaks 1 and 2 and between peaks 3 and 4, although each pair appears as two separate, overlapping peaks. These peaks are designated as peak 1/2 and peak 3/4. There are two predominant ganglioside peaks in every sample from normal and Graves’ thyroids, consistent with what had been reported previously in orbital and dermal fibroblasts (14). The most abundant in all strains examined is peak 1/2. The material migrating in this complex accounts for 62.8 ± 1.4% of the total. This is not significantly different from the fractional contribution of the peak found in dermal fibroblasts (63.9 ± 2.9%) (14). However, as with dermal fibroblasts, it is significantly different from the relative percentage of peak 1/2 of orbital fibroblasts (35.3 ± 2.4%, P < 0.01) (14). Peak 1/2 migrated slightly ahead of the GM1 standards in both dimensions. The other major ganglioside complex in thyroid fibroblasts, peak 7, migrates slightly ahead of GD1a standards in both dimensions and made up 16.8 ± 0.6% of the total. Comparison to the relative percent of peak 7 of dermal (11.1 ± 1.3%) and orbital (36.8± 2.4%) reveals significant differences with both (P< 0.05 and P < 0.01, respectively). The relative abundance of several other peaks varies between the two profiles. The composite ratio of peak 1/2 to peak 7 in all thyroid fibroblast strains is 3.8 ± 0.2 (mean ± sem). As can be seen in Fig. 4, this ratio differs from the ratios found between the two peaks in both the orbital (0.98± 0.1, P < 0.01) and dermal (6.1 ± 0.7, P < 0.7, P < 0.01) fibroblasts (14).
Relative percentages of thyroid fibroblast gangliosides on thin layer chromatograms. Chromatograms, including the one displayed in Fig. 3, were scanned with a densitometer. Values were expressed as a percentage of the total measured ganglioside present in each sample. Bars indicate the means for each peak± sd for Graves’ (n = 6) and normal (n = 4). Minor peaks 6 and 11 could not be reliably detected in every sample. Thus, for peak 11 of normal fibroblasts, n = 2 and for peaks 6 and 11 of Graves’ fibroblasts, n = 3.
Two-dimensional TLC of human thyroid fibroblast gangliosides. Gangliosides were extracted from thyroid fibroblasts and 3–5 μg ganglioside-bound sialic acid were analyzed on two-dimensional TLC plates as described in Materials and Methods. The chromatogram was generated using gangliosides from an individual donor. The schematic diagram of the plate indicates individual peaks referred to in Fig. 2. The origin is indicated by the dot in the lower right corner. Chromatographic positions of murine brain ganglioside standards are indicated across the top (first solvent) and in the left (second solvent) margins of the chromatogram and designated M (GM1), D (GD1a), and T (GT1b) according to the nomenclature of Svennerholm (59).
Comparison of the ratios of mean relative intensities of ganglioside peak 1/2 to peak 7 in thyroid, orbital, and dermal fibroblasts. Ratios of the means of volumes of ganglioside peak 1/2 to peak 7 were calculated from densitometric measurements of chromatograms from each individual strain for each class of fibroblast. Each datum point represents the ratios expressed as the mean ± sem. Measurements from orbital and dermal fibroblasts (*) were reported previously (14). The data from thyroid fibroblasts derive from 10 different strains. The ratio found in thyroid fibroblasts differs from that in orbital (P < 0.01) and dermal (P < 0.01) fibroblasts.
Overall uniformity exhibited by the individual culture strains with regard to specific gangliosides is remarkable. However, significant variability exists in the individual chromatograms with regard to the peaks of the monosialoganglioside region (peaks 1 through 6). Two of the strains derived from patients with Graves’ disease and one from normal thyroid lack some or all of the minor peaks designated 5 and 6. To rule out differences in the amount of material loaded in these samples as accounting for our failure to detect relatively minor peaks, we intentionally overloaded the TLC’s for these strains and were still unable to visualize them (data not shown). With regard to the disialoganglioside region (peaks 7 through 11), no such variation is observed among the 10 strains analyzed. Moreover, statistical analysis of the individual peaks fails to disclose any systematic differences between strains from patients with Graves’ disease and those without thyroid autoimmunity.
Human thyroid fibroblasts express Thy-1 uniformly
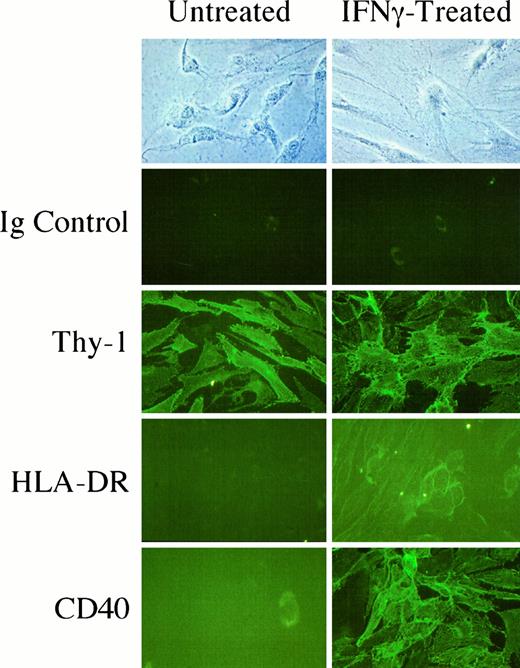
Dermal fibroblasts uniformly express Thy-1, a glycoprotein on the surface of the cell (9). In contrast, orbital fibroblasts derived from the connective/adipose tissue are comprised of cells that express Thy-1 and those that do not (9). The fraction of cells expressing Thy-1 in orbital cultures ranges from 54–71% in the strains examined. Moreover, the Thy-1 expression status of human fibroblasts is faithfully retained over prolonged time in culture. Thyroid fibroblasts resemble dermal fibroblasts with regard to a uniform surface display of Thy-1. Flow cytometric analysis of intact thyroid fibroblasts confirms this near-uniformity. There is a homogeneous fluorescent signal on the surfaces of the cells under both unstimulated and interferon-γ-treated culture conditions (Fig. 5). It would appear from both immunofluorescent studies and flow cytometry that Thy-1 expression is invariant with respect to interferon-γ treatment.
In situ staining of primary thyroid fibroblasts for the surface expression of Thy-1, HLA-DR, and CD40. Monolayers were incubated without or with interferon-γ (500 U/ml) for 72 h and then stained with anti-Thy-1 (F15–421-5), antihuman HLA-DR, anti-CD40, or Ig isotype antibody (as a control). Cultures were then incubated with FITC-conjugated secondary goat antimouse antibody. Final magnification, ×132.
Thyroid fibroblasts treated with interferon-γ express surface HLA-DR
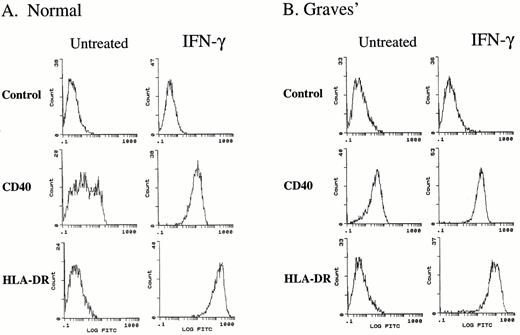
Thyroid fibroblasts do not express detectable levels of HLA-DR under basal culture conditions. This is assessed by indirect immunofluorescence (Fig. 5) and with immunostaining followed by flow cytometry (Fig. 6). Addition of interferon-γ (500 U/ml) for 72 h, however, results in a strong induction of the protein on the surface of the fibroblasts. It would appear that this response occurs uniformly among the fibroblast population. This result is consistent with our findings in orbital and dermal fibroblasts where interferon-γ induced HLA-DR (34). The expression of HLA-DR in thyroid fibroblasts treated with interferon-γ was dramatic in cultures derived from both Graves’ and normal thyroids, suggesting that the disease was not a major determinant of response.
Flow cytometric analysis of thyroid fibroblasts for surface expression of Thy-1, CD40, and HLA-DR. Representative flow cytometric analysis of thyroid fibroblasts from (A) normal and (B) Graves’ disease-derived thyroid tissue are shown. Cells were stained with anti-HLA-DR, anti-CD40, or an isotype control primary antibody followed by a FITC-conjugated secondary antibody. Fibroblasts were treated without or with interferon-γ (500 U/ml) for 72 h.
Thyroid fibroblasts express CD40 messenger RNA (mRNA) and protein
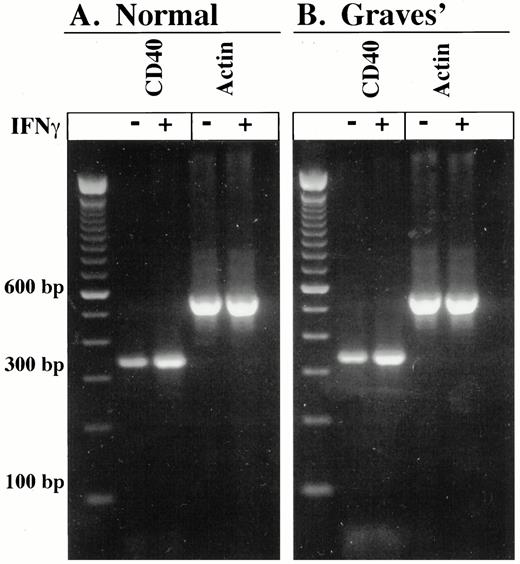
Thyroid fibroblasts were examined for the expression of CD40. Previous studies have disclosed that some primary human fibroblasts, such as those from the lung and gingiva express the antigen while certain dermal fibroblasts do not (31). We found that, in contrast to HLA-DR, CD40 protein is expressed by thyroid fibroblasts under basal culture conditions. Histograms demonstrating background staining (Control) and CD40 expression in untreated fibroblasts are shown in Fig. 6. The log fluorescence intensity of staining is plotted vs. cell number for each sample analyzed. Fibroblasts from Graves’ and normal tissue express CD40. We confirmed this expression of the protein by assessing CD40 mRNA levels by RT-PCR in untreated thyroid fibroblasts as well as those treated with interferon-γ (Fig. 7). Equivalent amounts of RNA were reverse transcribed, and the cDNA generated was then amplified for 30 cycles with primers specific for CD40 and β-actin. Thyroid fibroblasts derived from normal and Graves’ tissue express CD40 mRNA under basal conditions. Exposure to interferon-γ appears to up-regulate the mRNA levels in both types of fibroblasts; however, the RT-PCR was not quantitative. This issue was assessed further by examining the levels of surface CD40 protein display. They are found to be increased substantially by interferon-γ (Figs. 5 and 6). When stimulated with the cytokine for 72 h, there is a dramatic 10-fold up-regulation of CD40 protein in thyroid fibroblasts as assessed by flow cytometry.
Expression of human CD40 and β-actin mRNA in normal (A) and Graves’ disease-derived thyroid fibroblasts (B). Cells were treated without or with interferon-γ (500 U/ml) for 48 h. Total cellular RNA was isolated, and equivalent amounts were reverse-transcribed into cDNA. The cDNA was amplified with specific primer sets for human CD40 and β-actin as described in Materials and Methods. PCR products representing CD40 (330 bp) and β-actin (539 bp) were resolved on ethidium bromide-stained 2% agarose gels.
CD40 engagement with CD40 ligand results in the activation of NF-κB in thyroid fibroblasts
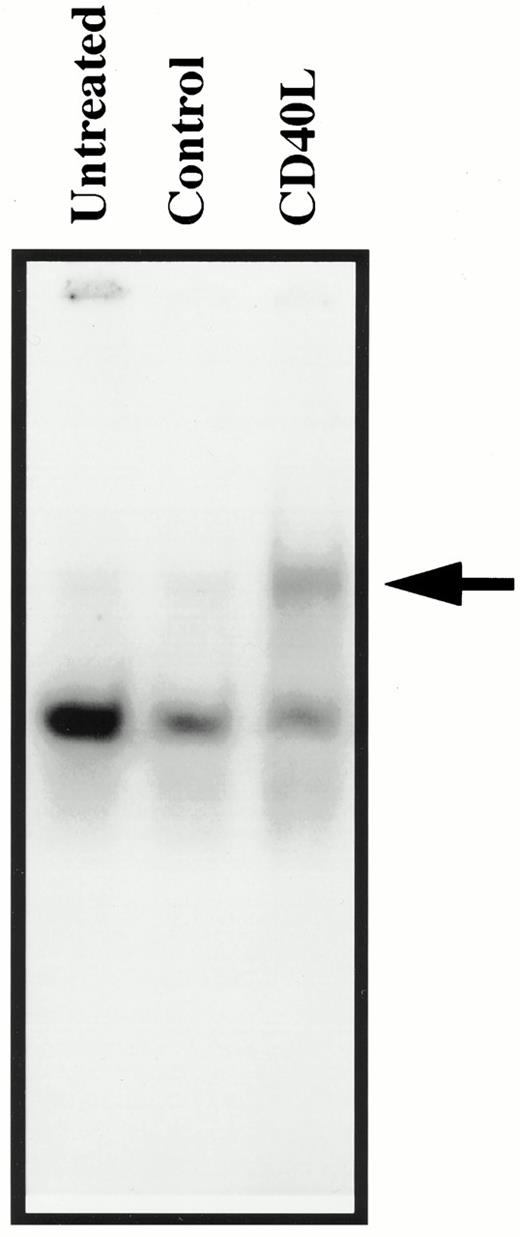
Having demonstrated the display of CD40 on the surface of the thyroid fibroblast, we next assessed whether this determinant was functional with regard to its engagement with CD40L. When CD40/CD40L complex formation occurs on the surface of B lymphocytes, many of the resulting molecular events are mediated through the activation of NF-κB (35). We therefore performed a series of EMSAs on the nuclear extracts of thyroid fibroblasts that had been pretreated with interferon-γ to up-regulate surface CD40 display. Cells were stimulated for 2 h with control insect membranes or those containing CD40L. Nuclear proteins were then incubated with a 32P-labeled double-stranded oligonucleotide probe representing the consensus sequence for the NF-κB-binding site. As the electrophoretic pattern shown in Fig. 8 demonstrates, there is a substantial up-regulation of NF-κB-binding activity. The activity is similar to that elicited by tumor necrosis factor-α, an established activator of NF-κB (data not shown). Identity of the protein/DNA complex was established by adding 200-fold excess of unlabeled NF-κB oligonucleotide, which quenched the signal, as expected. A supershift EMSA was performed by preincubating the reaction mixture with anti-p65 (RelA) polyclonal antiserum. Addition of the antibody resulted in the supershift of the NF-κB/DNA complex, confirming the presence of the p65-transactivating subunit (data not shown). Addition of preimmune serum failed to supershift the complex. These results indicate that thyroid fibroblasts preincubated with interferon-γ display functional CD40 on their surface and that engagement with CD40L results in the activation and nuclear translocation of NF-κB.
Insect cell membranes containing human CD40L trigger nuclear mobilization of NF-κB in thyroid fibroblasts. Interferon-γ-pretreated thyroid fibroblasts were stimulated with either control insect membranes or with membranes containing CD40L for 2 h. Nuclear extracts were prepared and subjected to EMSA using a 32P-labeled oligonucleotide probe for NF-κB as described in Materials and Methods. The arrow indicates the shifted protein/DNA complex induced by CD40L.
Thyroid fibroblasts respond to CD40 ligand engagement with regard to cytokine expression
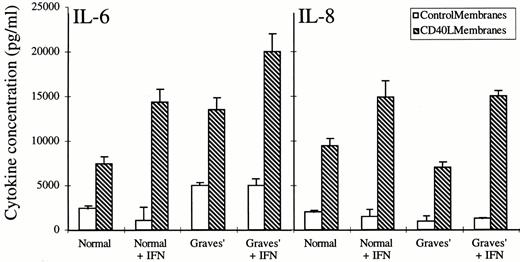
We next determined whether engagement of CD40 on thyroid fibroblasts with CD40L leads to alterations in fibroblast cytokine expression. IL-6 and IL-8 are important proinflammatory cytokines, the promoter regions of which contain NF-κB sites. Moreover, the expression of both IL-6 and IL-8 genes can be transactivated by p65-containing dimers (36–38). IL-6 is an important trigger for the activation of B lymphocytes (39) and is a fibroblast autocrine growth factor (40). IL-8 is a powerful chemoattractant molecule for neutrophils and T lymphocytes (41). We therefore examined the influence of CD40 activation on IL-6 and IL-8 expression in thyroid fibroblasts. Confluent monolayers were incubated in the absence or presence of interferon-γ (500 U/ml) for 72 h to up-regulate surface CD40 display. Cultures were then treated with fresh medium with insect membranes containing GST (control) or CD40L for 72 h, and then the media were collected for cytokine quantification using ELISA assays. CD40 engagement with CD40L in these cells provokes a substantial increase in IL-6 release into the culture medium. The magnitude of this response is 3.1-fold and 2.5-fold in normal and Graves’ fibroblasts, respectively, as Fig. 9 (left panel) suggests. Priming the cultures with interferon-γ (500 U/ml) pretreatment enhances the impact of CD40 engagement, as expected. After interferon-γ pretreatment, the increases are 15-fold and 4-fold in the two types of fibroblasts. The effects of interferon-γ on basal IL-6 expression are negligible. IL-8 is a chemoattractant and thus potentially of importance in the trafficking of neutrophils and T lymphocytes to the thyroid in inflammatory states. It is expressed by both bone marrow-derived and nonimmunocompetent cells at sites of tissue injury and inflammation (41–43). As was the case with regard to IL-6, thyroid fibroblasts express low levels of IL-8 constitutively, and mRNA encoding the cytokine is detectable by RT-PCR (data not shown). Il-8 synthesis increases dramatically in thyroid fibroblasts after CD40 activation (Fig. 9, right panel). CD40L elicits a 5.7-fold and 30.4-fold increase in IL-8 production in normal and Graves’ thyroid fibroblasts not pretreated with interferon-γ. As can be seen, pretreatment of the cultures with the cytokine enhances substantially the impact of CD40 engagement. The magnitude of the induction is 15.5-fold and 30-fold above baseline in the respective pretreated culture strains. Thus CD40 engagement in thyroid fibroblasts from Graves’ and normal tissue results in substantial increases in IL-6 and IL-8 expression.
Activation of thyroid fibroblasts from normal tissue and from Graves’ disease by CD40 engagement results in the up-regulation of IL-6 and IL-8 expression. Fibroblasts were pretreated without or with interferon-γ (500 U/ml) for 72 h, after which insect membranes containing GST (control) or human CD40L were added for 72 h. Media were harvested and assayed for IL-6 and IL-8 using a specific double-determinant ELISA. Data are expressed as the mean± sd (n = 3).
Thyroid fibroblasts express IL-1α and IL-1β in response to exogenous IL-1
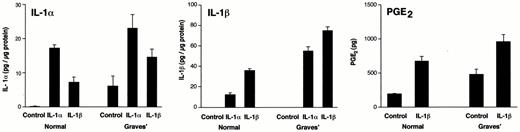
A wide variety of fibroblasts have been shown to express either of the IL-1 gene products. Some, such as certain lung fibroblasts, express only IL-1α (44). We examined thyroid fibroblasts for basal and cytokine-stimulated IL-1α and IL-1β production. As Fig. 10 suggests, IL-1α is expressed in untreated Graves’ fibroblasts but is at the limits of detection in the cultures from normal thyroid tissue. In contrast, IL-1β is undetectable in both types of untreated fibroblasts. When the cultures were exposed to exogenous IL-1, the levels of both IL-1α and IL-1β are up-regulated substantially. The levels of IL-1α achieved after 16 h of IL-1α and IL-1β exposure are 17.3 ± 0.9 and 7.2 ± 1.6 pg/μg protein in fibroblasts from normal tissue (100-fold and 42-fold increases, respectively) and 23.1 ± 4 and 14.6 ± 2.4 pg/μg protein (3.8-fold and 2.4-fold, respectively) in cultures of Graves’ fibroblasts. With regard to IL-1β expression, IL-1α and IL-1β induce the cytokine’s levels to 12.5 ± 1.7 and 35.9 ± 2 pg/μg protein, respectively, in normal fibroblasts and 55 ± 4.3 and 74.6 ± 3.7 pg/μg protein, respectively, in Graves’ cultures. Thus fibroblasts from Graves’ and normal thyroid tissue exhibit substantial expression of IL-1α and IL-1β when treated with exogenous IL-1.
Treatment of thyroid fibroblasts with exogenous IL-1 results in the up-regulation of IL-1α (panel A) and IL-1β (panel B) expression and PGE2 production (panel C). These were assayed as described in Materials and Methods. The concentration of exogenous IL-1α and IL-1β was 10 ng/ml, and the treatment period was 16 h. Data are presented as the mean ± sem of triplicate cultures.
Thyroid fibroblasts synthesize PGE2 in response to IL-1β
Human fibroblasts in culture display considerable variability with regard to levels of basal and inducible PGE2 production (12, 13, 45). Moreover, this prostanoid plays an important role as a determinant of the tissue immunological environment (46). We therefore assessed PGE2 synthesis in thyroid fibroblasts. As the data in Fig. 10 demonstrate, these cells produce detectable levels of the prostanoid under basal culture conditions. PGE2 levels were 192 ± 10 pg in normal fibroblasts and 480 ± 74 pg in fibroblasts from Graves’ tissue. When the cultures are treated for 16 h with IL-1β (10 ng/ml), PGE2 levels were increased modestly to 672 ± 70 pg and 960 ± 100 pg in the two cell types, respectively (3.5-fold and 2-fold increases). These values are similar to those observed in dermal fibroblasts where control levels of PGE2 are 120 ± 10 pg and those after treatment with IL-1β for 16 h increase to 1200 ± 15 pg. In contrast, PGE2 levels in orbital cultures are 44 ± 8.7 pg before and 3000 ± 74 pg after IL-1β treatment.
Discussion
We have begun to characterize the thyroid fibroblast, a cell type receiving little attention previously. Fibroblast diversity is only now being appreciated, and the vast array of regulatory molecules they express suggests that they may play important roles in normal tissue function and underlie disease susceptibility. Because fibroblasts are common to both the orbit and thyroid, we have begun to consider what attributes might be unique to these fibroblasts. Such shared characteristics might prove the basis, in part, for the association between the two anatomic regions in Graves’ disease. Thyroid fibroblasts exhibit a profile of phenotypic attributes that appears distinct from other fibroblasts. Like lung, orbital, and gingival fibroblasts, they express CD40. On the other hand, they express Thy-1 uniformly as do dermal fibroblasts (9), although they are probably a heterogeneous population of cells. With regard to prostanoid production, they are considerably less active in synthesizing and releasing PGE2 under cytokine-stimulated culture conditions than are orbital cells. This suggests that their role in inflammatory responses may differ from that of orbital fibroblasts.
We hypothesize that differences in fibroblast phenotype reflect peculiarities in function and susceptibility to disease manifested by the tissues from which the cells derive. Moreover, we believe that fibroblast subtypes, as have been demonstrated in the lung (10), spleen (11), and orbit (9), may serve different roles in tissue repair and normal remodeling. This notion of fibroblast specialization differs from the prevailing concept of more or less equivalence among these cells. Substantial evidence, some dating 30 yr, suggests that the site of derivation is a crucial determinant of fibroblast behavior in vitro. By virtue of their extensive biosynthetic repertoires, fibroblasts almost certainly impact on the normal function of neighboring cells. This occurs through characteristic extracellular matrix elaboration and by releasing small regulatory molecules, such as growth factors and cytokines, into the extracellular space. Thus differences in fibroblast responses to molecular cues could account for variations in wound healing and inflammatory responses observed in dissimilar tissues.
We have demonstrated previously that a major departure in fibroblast phenotype, related to the anatomic region of derivation, relates to the profile of gangliosides they express (14). Thyroid fibroblasts exhibit gangliosides in proportions that differ from those in other fibroblast types. They do not appear to display novel, abundant glycolipids not found in the other fibroblasts. Among the abundant gangliosides, it is peak 7 material that differs most among fibroblast strains. Gangliosides are sialic acid-containing glycosphingolipids with diverse biological functions (47). Their putative roles in human autoimmunity (48) make the observations concerning diversity among fibroblasts, with respect to ganglioside expression, particularly interesting. Because gangliosides play important roles in cell signaling (15, 47), differences in expression may represent important and cell-specific recognition determinants. The presence of antibodies against the Forssman glycolipid (Gb5) in patients with Graves’ disease and Hashimoto’s thyroiditis (49) and the apparent alterations in ganglioside content in membranes collected from whole thyroid glands (50) suggest potential relevance of these findings in fibroblasts to thyroid autoimmunity. It is intriguing that the variability in relative ganglioside abundance is restricted to the monosialogangliosides. The material in peaks 5 and 6 may represent ganglioside species similar to those in peaks 3 and 4, differing perhaps in the lengths of fatty acyl chains of the ceramide moiety. Interestingly, neuroblastoma gangliosides with shorter fatty acyl chains are considerably more active immunologically than are those with longer chains (51).
We have demonstrated that thyroid fibroblasts treated with interferon-γ express detectable cell-surface HLA-DR (Fig. 5). Thus these fibroblasts are capable of antigen presentation in the setting of inflammation. Thyroid fibroblasts also display uniformly high levels of CD40 on their surface when treated with interferon-γ. Moreover, they respond to CD40L with regard to a marked up-regulation of IL-6 and IL-8 expression. We hypothesize that the expression of CD40 in fibroblasts underlies, in some way, the propensity for the thyroid to become inflamed. This unexpected result suggests a previously unrecognized pathway whereby resident fibroblasts can receive molecular information from cells expressing CD40L, such as T lymphocytes and mast cells, both of which have been implicated in the pathogenesis of autoimmune thyroid diseases. Mast cells are considered normal residential cells in thyroid tissue (52). Thus it is possible that cross-talk between mast cells and fibroblasts through CD40 occurs under physiological conditions as well.
Our finding that IL-6 is strongly expressed in CD40-engaged fibroblasts may be of considerable relevance to the pathogenesis of autoimmunity of the thyroid such as Graves’ disease and Hashimoto’s thyroiditis. IL-6 is a potent activator of B lymphocytes and has been shown to enhance B cell proliferation and Ig production (39). Moreover, it has been suggested that IL-6 acts as an autocrine growth factor in fibroblasts (40). Serum IL-6 and soluble IL-6 receptor have been reported to be elevated in Graves’ disease (53, 54), and elevated levels have been associated with toxic nodular goiter. The mRNAs encoding IL-6, as well as IL-2, IL-4, IL-5, IL-10, and interferon-γ, have been demonstrated by RT-PCR in glands affected by both Hashimoto’s thyroiditis and Graves’ disease (55). With regard to Graves’ disease, the IL-6 synthesis occurring in fibroblasts could contribute substantially to the proliferation and activation of intrathyroidal B lymphocytes. This would potentially enhance thyroid-stimulating Ig production. IL-8 is also strongly up-regulated through CD40 engagement. IL-8 is an important chemattractant for neutrophils and T lymphocytes (41, 42, 56). The ability for CD40 activation to induce IL-6 and IL-8 expression defines a previously unidentified circuit for the coordination by the fibroblast of both B and T lymphocyte activities within the gland. IL-1α and IL-1β are also highly inducible in thyroid fibroblasts. IL-1 is thought to be an important coactivational molecule with IL-2 in Th1 lymphocytes (57), and thus the IL-1β released by the thyroid fibroblasts is of potential importance as a determinant of intrathyroidal lymphocyte function and cytokine production.
The current observations cast the thyroid fibroblast in a previously unrecognized and potentially central role in the pathogenesis of thyroid autoimmunity. Coupled with our recent (unpublished) finding that thyroid and orbital fibroblasts also express IL-16, a CD4+-specific chemoattractant (58), these fibroblasts may well engage in diverse sets of molecular communication with immunocompetent cells and thus orchestrate and amplify the local immune response. We suggest that thyroidal fibroblasts might function as an integral component of the immune system. Insight into the scope of involvement of these cells in states of health and disease in this tissue, as well as other endocrine organs, awaits further studies.
Acknowledgements
The authors wish to thank Dr. Timothy Jennings, Department of Pathology, Albany Medical College, for performing several immunostaining assays and Dr. Lawrence Robinson, Department of Surgery in Albany, for providing thyroid tissue. We appreciate the technical assistance of Heather Meekins and Chantal Turner.
References
Author notes
These studies were supported in part by NIH Grants EY-08976 and EY-11708 (to T.J.S.) and DE-11309, DE-11047, HL-56002, and CA-11198 (to R.P.P.); by a Merit Review award from the Department of Veterans Affairs (to T.J.S. and C.S.B.); and by the Rochester Area Pepper Center.