Abstract
Iontophoresis is a widely used drug delivery technique that has been used clinically to improve permeation through the skin for drugs and other actives in topical formulations. It is however not commonly used for the treatment of nail diseases despite its potential to improve transungual nail delivery. Instead, treatments for nail diseases are limited to relatively ineffective topical passive permeation techniques, which often result in relapses of nail diseases due to the thickness and hardness of the nail barrier resulting in lower permeation of the actives. Oral systemic antifungal agents that are also used are often associated with various undesirable side effects resulting in low patient compliance. This review article discusses what is currently known about the field of transungual iontophoresis, providing evidence of its efficacy and practicality in delivering drug to the entire surface of the nail for extended treatment periods. It also includes relevant details about the nail structure, the mechanisms of iontophoresis, and the associated in vitro and in vivo studies which have been used to investigate the optimal characteristics for a transungual iontophoretic drug delivery system. Iontophoresis is undoubtedly a promising option to treat nail diseases, and the use of this technique for clinical use will likely improve patient outcomes.
Graphical abstract




Similar content being viewed by others
References
Aggarwal R, Targhotra M, Kumar B, Sahoo PK, Chauhan MK. Treatment and management strategies of onychomycosis. J Mycol Med. 2020;30(2):100949.
Schons KR, Beber AA, Beck Mde O, Monticielo OA. Nail involvement in adult patients with plaque-type psoriasis: prevalence and clinical features. An Bras Dermatol. 2015;90(3):314–9.
Drake LA, Scher RK, Smith EB, Faich GA, Smith SL, Hong JJ, et al. Effect of onychomycosis on quality of life. J Am Acad Dermatol. 1998;38(5 Pt 1):702–4.
Lubeck DP, Patrick DL, McNulty P, Fifer SK, Birnbaum J. Quality of life of persons with onychomycosis. Qual Life Res. 1993;2(5):341–8.
Baran R. The burden of nail psoriasis: an introduction. Dermatology. 2010;221(Suppl 1):1–5.
Elewski BE. Onychomycosis: pathogenesis, diagnosis, and management. Clin Microbiol Rev. 1998;11(3):415–29.
Crawford F, Young P, Godfrey C, Bell-Syer SE, Hart R, Brunt E, et al. Oral treatments for toenail onychomycosis: a systematic review. Arch Dermatol. 2002;138(6):811–6.
Kreijkamp-Kaspers S, Hawke K, Guo L, Kerin G, Bell-Syer SE, Magin P, et al. Oral antifungal medication for toenail onychomycosis. Cochrane Database Syst Rev. 2017;7(7):Cd010031.
Del Rosso JQ. The role of topical antifungal therapy for onychomycosis and the emergence of newer agents. J Clin Aesthet Dermatol. 2014;7(7):10–8.
Shahi S, Deshpande S. Iontophoresis: an approach to drug delivery enhancement. Int J Pharm Sci Res. 2017;8(10):4056.
Feng X, Xiong X, Ran Y. Efficacy and tolerability of amorolfine 5% nail lacquer in combination with systemic antifungal agents for onychomycosis: a meta-analysis and systematic review. Dermatol Ther. 2017;30(3).
Radtke MA, Beikert FC, Augustin M. Nail psoriasis - a treatment challenge. J Dtsch Dermatol Ges. 2013;11(3):203–19 quiz 20.
Thatai P, Khan AB. Management of nail psoriasis by topical drug delivery: a pharmaceutical perspective. Int J Dermatol. 2020;59(8):915–25.
Gratieri T, Cunha-Filho M, Gelfuso GM. Overcoming hurdles in iontophoretic drug delivery: is skin the only barrier? - an update. Ther Deliv. 2019;10(4):211–4.
Dragicevic N, Maibach HI. Percutaneous penetration enhancers physical methods in penetration enhancement. Springer Berlin / Heidelberg: Berlin, Heidelberg; 2017.
James MP, Graham RM, English J. Percutaneous iontophoresis of prednisolone--a pharmacokinetic study. Clin Exp Dermatol. 1986;11(1):54–61.
Nair AB, Vaka SR, Murthy SN. Transungual delivery of terbinafine by iontophoresis in onychomycotic nails. Drug Dev Ind Pharm. 2011;37(10):1253–8.
Nair AB, Kim HD, Chakraborty B, Singh J, Zaman M, Gupta A, et al. Ungual and trans-ungual iontophoretic delivery of terbinafine for the treatment of onychomycosis. J Pharm Sci. 2009;98(11):4130–40.
Delgado-Charro MB. Iontophoretic drug delivery across the nail. Expert Opin Drug Deliv. 2012;9(1):91–103.
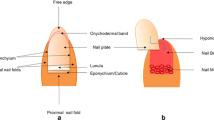
de Berker D. Nail anatomy. Clin Dermatol. 2013;31(5):509–15.
Bragulla HH, Homberger DG. Structure and functions of keratin proteins in simple, stratified, keratinized and cornified epithelia. J Anat. 2009;214(4):516–59.
Kobayashi Y, Miyamoto M, Sugibayashi K, Morimoto Y. Drug permeation through the three layers of the human nail plate. J Pharm Pharmacol. 1999;51(3):271–8.
Nogueiras-Nieto L, Gómez-Amoza JL, Delgado-Charro MB, Otero-Espinar FJ. Hydration and N-acetyl-l-cysteine alter the microstructure of human nail and bovine hoof: implications for drug delivery. J Control Release. 2011;156(3):337–44.
Dutet J, Delgado-Charro MB. Assessment of iontophoretic and passive ungual penetration by laser scanning confocal microscopy. Pharm Res. 2012;29(12):3464–74.
Stern DKMD, Diamantis SMD, Smith EMD, Wei HMDP, Gordon MMD, Muigai WBS, et al. Water content and other aspects of brittle versus normal fingernails. J Am Acad Dermatol. 2007;57(1):31–6.
de Berker DA, André J, Baran R. Nail biology and nail science. Int J Cosmet Sci. 2007;29(4):241–75.
Mertin D, Lippold BC. In-vitro permeability of the human nail and of a keratin membrane from bovine hooves: prediction of the penetration rate of antimycotics through the nail plate and their efficacy. J Pharm Pharmacol. 1997;49(9):866–72.
Hao J, Smith KA, Li SK. Time-dependent electrical properties of human nail upon hydration in vivo. J Pharm Sci. 2010;99(1):107–18.
Hao J, Li SK. Transungual iontophoretic transport of polar neutral and positively charged model permeants: effects of electrophoresis and electroosmosis. J Pharm Sci. 2008;97(2):893–905.
Nair AB, Singh K, Shinu P, Harsha S, Al-Dhubiab BE. A comprehensive study to evaluate the effect of constant low voltage iontophoresis on transungual delivery. Drug Dev Ind Pharm. 2013;39(5):807–15.
Manda P, Sammeta SM, Repka MA, Murthy SN. Iontophoresis across the proximal nail fold to target drugs to the nail matrix. J Pharm Sci. 2012;101(7):2392–7.
Kushwaha A, Shivakumar HN, Murthy SN. Iontophoresis for drug delivery into the nail apparatus: exploring hyponychium as the site of delivery. Drug Dev Ind Pharm. 2016;42(10):1678–82.
Monti D, Saccomani L, Chetoni P, Burgalassi S, Tampucci S, Mailland F. Validation of bovine hoof slices as a model for infected human toenails: in vitro ciclopirox transungual permeation. Br J Dermatol. 2011;165(1):99–105.
Mertin D, Lippold BC. In-vitro permeability of the human nail and of a keratin membrane from bovine hooves: penetration of chloramphenicol from lipophilic vehicles and a nail lacquer. J Pharm Pharmacol. 1997;49(3):241–5.
Shanbhag P, Jani U. Drug delivery through nails: present and future. New Horizons in Translational Medicine. 2017;3.
Khengar RH, Jones SA, Turner RB, Forbes B, Brown MB. Nail swelling as a pre-formulation screen for the selection and optimisation of ungual penetration enhancers. Pharm Res. 2007;24(12):2207–12.
Amichai B, Mosckovitz R, Trau H, Sholto O, Ben-Yaakov S, Royz M, et al. Iontophoretic terbinafine HCL 1.0% delivery across porcine and human nails. Mycopathologia. 2010;169(5):343–9.
Kushwaha A, Jacob M, Shiva Kumar HN, Hiremath S, Aradhya S, Repka MA, et al. Trans-ungual delivery of itraconazole hydrochloride by iontophoresis. Drug Dev Ind Pharm. 2015;41(7):1089–94.
Monti D, Egiziano E, Burgalassi S, Tampucci S, Terreni E, Tivegna S, et al. Influence of a combination of chemical enhancers and iontophoresis on in vitro transungual permeation of nystatin. AAPS PharmSciTech. 2018;19(4):1574–81.
Dutet J, Delgado-Charro MB. In vivo transungual iontophoresis: effect of DC current application on ionic transport and on transonychial water loss. J Control Release. 2009;140(2):117–25.
Nair AB, Vaka SR, Sammeta SM, Kim HD, Friden PM, Chakraborty B, et al. Trans-ungual iontophoretic delivery of terbinafine. J Pharm Sci. 2009;98(5):1788–96.
Nair AB, Kim HD, Davis SP, Etheredge R, Barsness M, Friden PM, et al. An ex vivo toe model used to assess applicators for the iontophoretic ungual delivery of terbinafine. Pharm Res. 2009;26(9):2194–201.
Baden HP, Goldsmith LA, Fleming B. A comparative study of the physicochemical properties of human keratinized tissues. Biochim Biophys Acta. 1973;322(2):269–78.
Lusiana, Reichl S, Müller-Goymann CC. Keratin film made of human hair as a nail plate model for studying drug permeation. Eur J Pharm Biopharm 2011;78(3):432–440.
McAuley WJ, Jones SA, Traynor MJ, Guesné S, Murdan S, Brown MB. An investigation of how fungal infection influences drug penetration through onychomycosis patient’s nail plates. Eur J Pharm Biopharm. 2016;102:178–84.
Baraldi A, Jones SA, Guesné S, Traynor MJ, McAuley WJ, Brown MB, et al. Human nail plate modifications induced by onychomycosis: implications for topical therapy. Pharm Res. 2014;32(5):1626–33.
Mohorcic M, Torkar A, Friedrich J, Kristl J, Murdan S. An investigation into keratinolytic enzymes to enhance ungual drug delivery. Int J Pharm. 2007;332(1–2):196–201.
Gupta R, Ramnani P. Microbial keratinases and their prospective applications: an overview. Appl Microbiol Biotechnol. 2006;70(1):21–33.
Lusiana RS, Müller-Goymann CC. Infected nail plate model made of human hair keratin for evaluating the efficacy of different topical antifungal formulations against Trichophyton rubrum in vitro. Eur J Pharm Biopharm. 2013;84(3):599–605.
Benzeval I, Bowen CR, Guy RH, Delgado-Charro MB. Effects of iontophoresis, hydration, and permeation enhancers on human nail plate: infrared and impedance spectroscopy assessment. Pharm Res. 2013;30(6):1652–62.
Murdan S, Hinsu D, Guimier M. A few aspects of transonychial water loss (TOWL): inter-individual, and intra-individual inter-finger, inter-hand and inter-day variabilities, and the influence of nail plate hydration, filing and varnish. Eur J Pharm Biopharm. 2008;70(2):684–9.
Hamilton JB, Terada H, Mestler GE. Studies of growth throughout the lifespan in Japanese: growth and size of nails and their relationship to age, sex, heredity, and other factors. J Gerontol. 1955;10(4):401–15.
Batory M, Namieciński P, Rotsztejn H. Evaluation of structural damage and pH of nail plates of hands after applying different methods of decorating. Int J Dermatol. 2019;58(3):311–8.
Murdan S, Milcovich G, Goriparthi GS. An assessment of the human nail plate pH. Skin Pharmacol Physiol. 2011;24(4):175–81.
Hashimoto K. Ultrastructure of the human toenail. I. Proximal nail matrix. J Invest Dermatol. 1971;56(3):235–46.
Finlay AY, Moseley H, Duggan TC. Ultrasound transmission time: an in vivo guide to nail thickness. Br J Dermatol. 1987;117(6):765–70.
Baran R, de Berker DAR, Holzberg M, Thomas L. Baran and Dawber’s diseases of the nails and their management. Somerset: John Wiley & Sons, Incorporated; 2012.
Dutet J, Delgado-Charro MB. Electroosmotic transport of mannitol across human nail during constant current iontophoresis. J Pharm Pharmacol. 2010;62(6):721–9.
Bradley Phipps J, Gyory JR. Transdermal ion migration. Adv Drug Deliv Rev. 1992;9(2):137–76.
Mudry B, Guy RH, Delgado-Charro MB. Electromigration of ions across the skin: determination and prediction of transport numbers. J Pharm Sci. 2006;95(3):561–9.
Sims SM, Higuchi WI, Srinivasan V. Interaction of electric field and electro-osmotic effects in determining iontophoretic enhancement of anions and cations. Int J Pharm. 1991;77(2):107–18.
Murthy SN, Waddell DC, Shivakumar HN, Balaji A, Bowers CP. Iontophoretic permselective property of human nail. J Dermatol Sci. 2007;46(2):150–2.
Mennini N, Bragagni M, Maestrelli F, Mura P. Physico-chemical characterization in solution and in the solid state of clonazepam complexes with native and chemically-modified cyclodextrins. J Pharm Biomed Anal. 2014;89:142–9.
Hao J, Smith KA, Li SK. Chemical method to enhance transungual transport and iontophoresis efficiency. Int J Pharm. 2008;357(1–2):61–9.
Prausnitz MR. A practical assessment of transdermal drug delivery by skin electroporation. Adv Drug Deliv Rev. 1999;35(1):61–76.
Kalinowski DP, Edsberg LE, Hewson RA, Johnson RH, Brogan MS. Low-voltage direct current as a fungicidal agent for treating onychomycosis. J Am Podiatr Med Assoc. 2004;94(6):565–72.
Nair AB, Sammeta SM, Kim HD, Chakraborty B, Friden PM, Murthy SN. Alteration of the diffusional barrier property of the nail leads to greater terbinafine drug loading and permeation. Int J Pharm. 2009;375(1–2):22–7.
Baswan SM, Li SK, LaCount TD, Kasting GB. Size and charge dependence of ion transport in human nail plate. J Pharm Sci. 2016;105(3):1201–8.
Uchida K, Yamaguchi H. Studies on the affinity of terbinafine with keratin. Jpn J Med Mycol. 1993;34(2):207–12.
Darkes MJM, Scott LJ, Goa KL. Terbinafine: a review of its use in onychomycosis in adults. Am J Clin Dermatol. 2003;4(1):39–65.
Balfour JA, Faulds D. Terbinafine. A review of its pharmacodynamic and pharmacokinetic properties, and therapeutic potential in superficial mycoses. Drugs. 1992;43(2):259–84.
Ghannoum MA, Hajjeh RA, Scher R, Konnikov N, Gupta AK, Summerbell R, et al. A large-scale north American study of fungal isolates from nails: the frequency of onychomycosis, fungal distribution, and antifungal susceptibility patterns. J Am Acad Dermatol. 2000;43(4):641–8.
Neubert RH, Gensbügel C, Jäckel A, Wartewig S. Different physicochemical properties of antimycotic agents are relevant for penetration into and through human nails. Pharmazie. 2006;61(7):604–7.
Hui X, Baker SJ, Wester RC, Barbadillo S, Cashmore AK, Sanders V, et al. In vitro penetration of a novel oxaborole antifungal (AN2690) into the human nail plate. J Pharm Sci. 2007;96(10):2622–31.
Monti D, Mazzantini D, Tampucci S, Vecchione A, Celandroni F, Burgalassi S, et al. Ciclopirox and efinaconazole transungual permeation, antifungal activity, and proficiency to induce resistance in Trichophyton rubrum. Antimicrob Agents Chemother. 2019;63(10).
Corrêa JCR, Salgado HRN. Review of fluconazole properties and analytical methods for its determination. Crit Rev Anal Chem. 2011;41(2):124–32.
Jaruratanasirikul S, Sriwiriyajan S. Effect of omeprazole on the pharmacokinetics of itraconazole. Eur J Clin Pharmacol. 1998;54(2):159–61.
Uzqueda M, Martín C, Zornoza A, Sánchez M, Martínez-Ohárriz MC, Vélaz I. Characterization of complexes between naftifine and cyclodextrins in solution and in the solid state. Pharm Res. 2006;23(5):980–8.
Narasimha Murthy S, Wiskirchen DE, Bowers CP. Iontophoretic drug delivery across human nail. J Pharm Sci. 2007;96(2):305–11.
Hao J, Li SK. Mechanistic study of electroosmotic transport across hydrated nail plates: effects of pH and ionic strength. J Pharm Sci. 2008;97(12):5186–97.
Smith KA, Hao J, Li SK. Influence of pH on transungual passive and iontophoretic transport. J Pharm Sci. 2010;99(4):1955–67.
Dutet J, Delgado-Charro MB. Transungual iontophoresis of lithium and sodium: effect of pH and co-ion competition on cationic transport numbers. J Control Release. 2010;144(2):168–74.
Hao J, Smith KA, Li SK. Iontophoretically enhanced ciclopirox delivery into and across human nail plate. J Pharm Sci. 2009;98(10):3608–16.
Kalaria DR, Patel P, Patravale V, Kalia YN. Comparison of the cutaneous iontophoretic delivery of rasagiline and selegiline across porcine and human skin in vitro. Int J Pharm. 2012;438(1–2):202–8.
Smith KA, Hao J, Li SK. Effects of ionic strength on passive and iontophoretic transport of cationic permeant across human nail. Pharm Res. 2009;26(6):1446–55.
Kobayashi Y, Komatsu T, Sumi M, Numajiri S, Miyamoto M, Kobayashi D, et al. In vitro permeation of several drugs through the human nail plate: relationship between physicochemical properties and nail permeability of drugs. Eur J Pharm Sci. 2004;21(4):471–7.
Smith KA, Hao J, Kevin LS. Effects of organic solvents on the barrier properties of human nail. J Pharm Sci. 2011;100(10):4244–57.
Wessel S, Gniadecka M, Jemec GB, Wulf HC. Hydration of human nails investigated by NIR-FT-Raman spectroscopy. Biochim Biophys Acta. 1999;1433(1–2):210–6.
Marzec E, Olszewski J. Influence of water and temperature on the electrical conductivity of the human nail. J Therm Anal Calorim. 2019;138(3):2185–91.
Lauharanta J. Comparative efficacy and safety of amorolfine nail lacquer 2% versus 5% once weekly. Clin Exp Dermatol. 1992;17(Suppl 1):41–3.
Pittrof F, Gerhards J, Erni W, Klecak G. Loceryl nail lacquer--realization of a new galenical approach to onychomycosis therapy. Clin Exp Dermatol. 1992;17(Suppl 1):26–8.
Chouhan P, Saini TR. Hydration of nail plate: a novel screening model for transungual drug permeation enhancers. Int J Pharm. 2012;436(1–2):179–82.
Nair AB, Chakraborty B, Murthy SN. Effect of polyethylene glycols on the trans-ungual delivery of terbinafine. Curr Drug Deliv. 2010;7(5):407–14.
Oon HH, Tan HH. Iontophoretic terbinafine delivery in onychomycosis: questionable nail growth. Br J Dermatol. 2010;162(3):699–700.
Amichai B, Nitzan B, Mosckovitz R, Shemer A. Iontophoretic delivery of terbinafine in onychomycosis: a preliminary study. Br J Dermatol. 2010;162(1):46–50.
Samhan A. Terbinafine hydrochloride 1% Iontophoresis for the treatment of toenail onychomycosis: a randomized placebo controlled study. Journal of Novel Physiotherapies. 2015;05.
Le QV, Howard A. Dexamethasone iontophoresis for the treatment of nail psoriasis. Australas J Dermatol. 2013;54(2):115–9.
Saki N, Hosseinpoor S, Heiran A, Mohammadi A, Zeraatpishe M. Comparing the efficacy of triamcinolone acetonide iontophoresis versus topical calcipotriol/betamethasone dipropionate in treating nail psoriasis: a bilateral controlled clinical trial. Dermatol Res Pract. 2018;2018:2637691.
Author information
Authors and Affiliations
Corresponding author
Additional information
Guest Editors: Diane Burgess, Marilyn Morris and Meena Subramanyam
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Chen, K., Puri, V. & Michniak-Kohn, B. Iontophoresis to Overcome the Challenge of Nail Permeation: Considerations and Optimizations for Successful Ungual Drug Delivery. AAPS J 23, 25 (2021). https://doi.org/10.1208/s12248-020-00552-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1208/s12248-020-00552-y