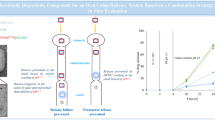
Abstract
Ceftriaxone (CTX) is a widely used injectable third-generation cephalosporin that exhibits broad-spectrum antibacterial activity. Unfortunately, the oral route of this drug suffers different encumbrances, such as instability in the upper part of the GIT and enzymatic degradation, as well as poor permeability. There is no reported tablet dosage form for this drug. In this respect, the authors investigated the possibility of developing an enteric-coated oral tablet of CTX that would be helpful for better patient compliance. The tablet consists of directly compressed core of CTX, citric acid (CA), sodium chloride (NaCl), and two biopolymers—chitosan (CH), a permeation enhancer, and silicified microcrystalline cellulose (SMCC), a wicking agent. Both biopolymers are naturally occurring polysaccharides that are biodegradable in the colon and able to incorporate acid labile drugs. CA is a pH modulator to protect CTX from protease enzymes, while NaCl is a translocation enhancer that helps drug penetration. The enteric coat of the core was shellac (SH) with plasticizer glycerol tristearate (GTS) and CA that was applied by direct compression (dry coating). The solventless heat curable coat resulted in an enteric-coated tablet that complies with the USP pharmacopeia. The optimized formula was further subjected to in vitro release and stability studies, as well as ingredient compatibility. In vivo oral bioavailability of the enteric-coated tablets in rabbits gave promising results (absolute bioavailability of about 80%). Synergistically, all ingredients together augmented oral bioavailability of CTX. This developed formula could be a perspective delivery system for those drugs intended to be absorbed from the colon such as peptides and peptide-like drugs.








Similar content being viewed by others
References
Hirzel C, Hirzberger L, Furrer H, Endimiani A. Bactericidal activity of penicillin, ceftriaxone, gentamicin and daptomycin alone and in combination against Aerococcus urinae. Int J Antimicrob Ag. 2016;48(3):271–6.
DeBrouse DR. (2009). Google Patents No.0123537 A. Retrieved from https://patents.google.com/patent/US20090123537A1/en. Accessed 9 May 2019.
Shah N, Seth A, Balaraman R, Aundhia C, Maheshwari R, Parmar G. Nanostructured lipid carriers for oral bioavailability enhancement of raloxifene: design and in vivo study. J Adv Res. 2016;7(3):423–34.
Lee S, Lee D, Chae S, Byun Y. Pharmacokinetics of a new orally available ceftriaxone formulation in physical complexation with a cationic analogue of bile acid in rats. Anti Microb Ag Chemo. 2006;50(5):1869–71.
Neelam S, Arundhati B, Puneet G. Enhancement of intestinal absorption of poorly absorbed ceftriaxone sodium by using mixed micelles of polyoxy ethylene (20) cetyl ether & oleic acid as peroral absorption enhancers. Schol Res Lib. 2010;2(3):131–42.
Beskid G, Unowsky J, Behl C, Siebelist J, Tossounian J, McGarry C, et al. Enteral, oral, and rectal absorption of ceftriaxone using glyceride enhancers. Chemotherapy. 1988;34(2):77–84.
Patel N, Lalwani D, Gollmer S, Injeti E, Sari Y, Nesamony J. Development and evaluation of a calcium alginate based oral ceftriaxone sodium formulation. Progress in Biomaterials. 2016;5(2):117–33.
Zaki N, Hafez M. Enhanced antibacterial effect of ceftriaxone sodium-loaded chitosan nanoparticles against intracellular Salmonella typhimurium. AAPS PharmSciTech. 2012;13(2):411–21.
Philip A, Philip B. Colon targeted drug delivery systems: a review on primary and novel approaches. Oman Med J. 2010;25(2):70–8.
Bernkop-Schnürch A, Dünnhaupt S. Chitosan-based drug delivery systems. Eur J Pharm Biopharm. 2012;81(3):463–9.
Sun W, Mao S, Wang Y, Junyaprasert VB, Zhang T, Na L, et al. Bioadhesion and oral absorption of enoxaparin nanocomplexes. Int J Pharm. 2010;386(1–2):275–81.
Grabovac V, Guggi D, Bernkop-Schnürch A. Comparison of the mucoadhesive properties of various polymers. Adv Drug Deliv Rev. 2005;57(11):1713–23.
Gupta S, Vyas SP. Carbopol/chitosan based pH triggered in situ gelling system for ocular delivery of timolol maleate. J Pharm Sci. 2010;78(1):959–76.
Sakloetsakun J, Bernkop-Schnürch A. In situ gelling properties of chitosan–thioglycolic acid conjugate in the presence of oxidizing agents. Biomaterials. 2009;30(1):6151–7.
Kubbinga M, Augustijns P, García MA, Heinen C, Wortelboer HM, Verwei M, et al. The effect of chitosan on the bioaccessibility and intestinal permeability of acyclovir. Euro J Pharm Biopharm. 2019;136(1):147–55.
Föger F, Bernkop-Schnürch A. In vivo evaluation of an oral delivery system for P-gp substrates based on thiolated chitosan. Biomaterials. 2006;27(23):4250–5.
Newton A, Prabakaran L. Comparative efficacy of chitosan, pectin based mesalamine colon targeted drug delivery systems on TNBS-induced IBD model rats. Anti-inflam Anti-allerg Med Chem. 2019;18(1):1–14.
Mohammed M, Syeda J, Wasan K, Wasan E. An overview of chitosan nanoparticles and its application in non-parenteral drug delivery. Pharmaceutics. 2017;9(4):53.
Almukainzi M, Araujo G, Löbenberg R. Orally disintegrating dosage forms. J Pharm Inves. 2019;49(2):229–43.
Bose S, Bogner R. Solventless pharmaceutical coating processes: a review. Pharm Dev Technol. 2007;12(2):115–31.
The United States pharmacopeia. National formulary. 40th revision. Rockville (MD)20852: United States Pharmacopeial Convention; 2017.
Sun X, Liu C, Omer A, Yang L-Y, Ouyang X-K. Dual-layered pH-sensitive alginate/chitosan/kappa-carrageenan microbeads for colon-targeted release of 5-fluorouracil. Int J Bio Mac Molec. 2019;132:487–94.
Pudjiastuti P, Hendradi E, Wafiroh S, Darmokoesoemo H, Fauzi M, Nahar L, et al. First order kinetics of salicylamide release from κ-carrageenan hard shell capsules in comparison with gelatin. IOP Conf Series: Earth and Environmental Science. 2019;217(1):1–5.
Higuchi T. Mechanism of sustained-action medication. Theoretical analysis of rate of release of solid drugs dispersed in solid matrices. J Pharm Sci. 1963;52(12):1145–9.
Korsmeyer R, Gurny R, Doelker E, Buri P, Peppas N. Mechanisms of solute release from porous hydrophilic polymers. Int J Pharm. 1983;15(1):25–35.
Campos M, de Moura AJ, dos Santos ME, Oliveira J, Hussni C, Peccinini R. Ceftriaxone pharmacokinetics by new simple and sensitive ultra-high-performance liquid chromatography method. Diag Micro Infec Disease. 2017;88(1):95–9.
Aman R, Meshali M, Ibrahim I. Novel chitosan-based solid-lipid nanoparticles to enhance the bio-residence of the miraculous phytochemical “apocynin”. Euro J Pharm Sci. 2018;124(1):304–18.
Cardot J, Davit B. In vitro–in vivo correlations: tricks and traps. AAPS J. 2012;14(3):491–9.
Wagner J, Nelson E. Percent absorbed time plots derived from blood level and/or urinary excretion data. J Pharm Sci. 1963;52(6):610–1.
Uranga J, Puertas A, Etxabide A, Dueñas M, Guerrero P, de la Caba K. Citric acid-incorporated fish gelatin/chitosan composite films. Food Hydrocoll. 2019;86:95–103.
Snejdrova E, Dittrich M. Pharmaceutical applications of plasticized polymers. Rec Adv Plast. 2012;4:70–90.
Osman Z. Investigation of different shellac grades and improvement of release from air suspension coated pellets. PhD [dissertation]. Mainz: Universitätsbibliothek Mainz; 2012. Available from: https://d-nb.info/1029698821/34. Accessed 9 May 2019.
Farag Y, Leopold C. Physicochemical properties of various shellac types. Dissolut Technol. 2009;16:33–9.
Kapoor D, Gupta P. A malleable technique for future coating process solventless coating. J Critic Rev. 2016;3(2):55–9.
Saigal N, Baboota S, Ahuja A, Ali J. Microcrystalline cellulose as a versatile excipient in drug research. J Young Pharmacists. 2009;1(1):6–12.
Marczyński Z, Zgoda M, Jambor J. Application of silicified microcrystalline cellulose (Prosolv) as a polymer carrier of Epilobium parviflorum Schreb. extract in oral solid drug form. Polim Med. 2007;37(2):21–32.
Owens H, Dash A. Ceftriaxone sodium: comprehensive profile. In: Harry B. Profiles of drug substances, excipients and related methodology. USA. 2003;30:21–57.
Awadeen R, Boughdady M, Meshali M. New in-situ gelling biopolymer-based matrix for bioavailability enhancement of glimepiride; in-vitro/in-vivo X-ray imaging and pharmacodynamic evaluations. Pharm Dev Technol. 2018;2(5):539–49.
Shantha K, Harding D. Synthesis and characterisation of chemically modified chitosan microspheres. Carbohydr Polym. 2002;48(3):247–53.
Guinesi L, Cavalheiro E. The use of DSC curves to determine the acetylation degree of chitin/chitosan samples. Thermochim Acta. 2006;444(2):128–33.
Theismann E-M, Keppler JK, Knipp J-R, Fangmann D, Appel E, Gorb SN, et al. Adjustment of triple shellac coating for precise release of bioactive substances with different physico-chemical properties in the ileocolonic region. Int J Pharm. 2019;564(1):472–84.
Limmatvapirat S, Limmatvapirat C, Puttipipatkhachorn S, Nunthanid J, Luangtana-anan M, Sriamornsak P. Modulation of drug release kinetics of shellac-based matrix tablets by in-situ polymerization through annealing process. Eur J Pharm Biopharm. 2008;69(3):1004–13.
Borg T, Mohamed E, El Naggar E, El-Sheakh A, Hamed M. Colon targeting of naringin for cytoprotection against ulcerative colitis: in vitro-in vivo study. IOSR J Pharm Bio Sci. 2017;12(2):23–9.
Asghar L, Chure C, Chandran S. Colon specific delivery of indomethacin: effect of incorporating pH sensitive polymers in xanthan gum matrix bases. AAPS PharmSciTech. 2009;10(2):418–29.
Abdelbary A, El-Gazayerly O, El-Gendy N, Ali A. Floating tablet of trimetazidine dihydrochloride: an approach for extended release with zero-order kinetics. AAPS PharmSciTech. 2010;11(3):1058–67.
Sexton D, Faucette R. (2019). Google Patents US0022641A1. Retrieved from https://patents.google.com/patent/US20190022641A1/en?. Accessed 9 May 2019.
Bonferoni MC, Sandri G, Rossi S, Ferrari F, Gibin S, Caramella C. Chitosan citrate as multifunctional polymer for vaginal delivery: evaluation of penetration enhancement and peptidase inhibition properties. Euro J Pharm Sci. 2008;33(2):166–76.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Maghrabia, A.E., Boughdady, M.F. & Meshali, M.M. New Perspective Enteric-Coated Tablet Dosage Form for Oral Administration of Ceftriaxone: In Vitro and In Vivo Assessments. AAPS PharmSciTech 20, 306 (2019). https://doi.org/10.1208/s12249-019-1512-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1208/s12249-019-1512-y