Abstract
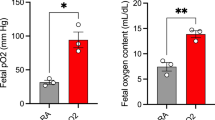
We hypothesized that chronic hypoxia disrupts mitochondrial function via oxidative stress in fetal organs. Pregnant guinea pig sows were exposed to either normoxia or hypoxia (10.5% O2, 14 days) in the presence or absence of the antioxidant, N-acetylcysteine (NAC). Near-term anesthetized fetuses were delivered via hysterotomy, and fetal livers, hearts, lungs, and forebrains harvested. We quantified the effects of chronic hypoxia on cytochrome oxidase (CCO) activity and 2 factors known to regulate CCO activity: malondialdehyde (MDA) and CCO subunit 4 (COX4). Hypoxia increased the MDA levels in fetal liver, heart, and lung with a corresponding reduction in CCO activity, prevented by prenatal NAC. The COX4 expression paralleled CCO activity in fetal liver and lung, but was unaltered in fetal hearts due to hypoxia. Hypoxia reduced the brain COX4 expression despite having no effect on CCO activity. This study identifies the mitochondrion as an important target site in tissue-specific oxidative stress for the induction of fetal hypoxic injury.
Similar content being viewed by others
References
Myatt L. Placental adaptive responses and fetal programming. J Physiol. 2006;572(pt 1):25–30.
Kingdom JC, Kaufmann P. Oxygen and placental villous development: origins of fetal hypoxia. Placenta. 1997;18(8):613–621.
Giaccia AJ, Simon MC, Johnson R. The biology of hypoxia: the role of oxygen sensing in development, normal function, and disease. Genes Dev. 2004;18(18):2183–2194.
Gilbert WM, Danielsen B. Pregnancy outcomes associated with intrauterine growth restriction. Am J Obstet Gynecol. 2003; 188(6):1596–1599; discussion 1599–1601.
Kramer MS, Séguin L, Lydon J, Goulet L. Socio-economic disparities in pregnancy outcome: why do the poor fare so poorly? Paediatr Perinat Epidemiol. 2000;14(3):194–210.
Julian CG. High altitude during pregnancy. Clin Chest Med. 2011; 32(1):21–31, vii.
Barker DJ. The fetal and infant origins of adult disease. BMJ. 1990;301(6761):1111.
Guzy RD, Hoyos B, Robin E, et al. Mitochondrial complex III is required for hypoxia-induced ROS production and cellular oxygen sensing. Cell Metab. 2005;1(6):401–408.
Fisher AB. Activation of endothelial NADPH oxidase as the source of a reactive oxygen species in lung ischemia. Chest. 1999;116(1 suppl):25S-26S.
Killilea DW, Hester R, Balczon R, Babal P, Gillespie MN. Free radical production in hypoxic pulmonary artery smooth muscle cells. Am J Physiol Lung Cell Mol Physiol. 2000;279(2): L408–L412.
Buonocore G, Perrone S, Longini M, Terzuoli L, Bracci R. Total hydroperoxide and advanced oxidation protein products in preterm hypoxic babies. Pediatr Res. 2000;47(2):221–224.
Raza H, Prabu SK, John A, Avadhani NG. Impaired mitochondrial respiratory functions and oxidative stress in streptozotocin-induced diabetic rats. Int J Mol Sci. 2011;12(5):3133–3147.
Valko M, Leibfritz D, Moncol J, Cronin MTD, Mazur M, Telser J. Free radicals and antioxidants in normal physiological functions and human disease. Int J Biochem Cell Biol. 2007;39(1):44–84.
Bedard K, Heinz KK. The NOX family of ROS-generating NADPH oxidases: physiology and pathophysiology. Physiol Rev. 2007;87(1):245–313.
Thompson L, Dong Y, Evans L. Chronic hypoxia increases inducible NOS-derived nitric oxide in fetal guinea pig hearts. Pediatr Res. 2009;65(2):188–192.
Evans LSC, Liu H, Pinkas GA, Thompson LP. Chronic hypoxia increases peroxynitrite, MMP9 expression, and collagen accumulation in fetal guinea pig hearts. Pediatr Res. 2012;71(1):25–31.
Dong Y, Yu Z, Sun Y, et al. Chronic fetal hypoxia produces selective brain injury associated with altered nitric oxide synthases. Am J Obstet Gynecol. 2011;204(3):254.e16–e28.
Aon M, Cortassa S, O’Rourke B. Redox-optimized ROS balance: a unifying hypothesis. Biochim Biophys Acta. 2010;1797(6–7): 865–877.
Demin OV, Kholodenko BN, Skulachev VP. A model of O2-generation in the complex III of the electron transport chain. Mol Cell Biochem. 1998;184(1–2):21–33.
Miwa S, Brand MD. Mitochondrial matrix reactive oxygen species production is very sensitive to mild uncoupling. Biochem Soc Trans. 2003;31(pt 6):1300–1301.
Chen Q, Vazquez EJ, Moghaddas S, Hoppel CL, Lesnefsky EJ. Production of reactive oxygen species by mitochondria: central role of complex III. J Biol Chem. 2003;278(38):36027–36031.
Chen J, Petersen DR, Schenker S, Henderson GI. Formation of malondialdehyde adducts in livers of rats exposed to ethanol: role in ethanol-mediated inhibition of cytochrome c oxidase. Alcohol Clin Exp Res. 2000;24(4):544–552.
Behn C, Araneda OF, Llanos AJ, Celedón G, González G. Hypoxia-related lipid peroxidation: evidences, implications and approaches. Respir Physiol Neurobiol. 2007;158(2–3): 143–150.
Fukuda R, Zhang H, Whan KJ, Shimoda L, Dang CV, Semenza GL. HIF-1 regulates cytochrome oxidase subunits to optimize efficiency of respiration in hypoxic cells. Cell. 2007;129(1):111–122.
Beloosesky R, Gayle DA, Amidi F, et al. N-acetyl-cysteine suppresses amniotic fluid and placenta inflammatory cytokine responses to lipopolysaccharide in rats. Am J Obstet Gynecol. 2006;194(1):268–273.
Wang B-J, Guo Y-L, Chang H-Y, et al. N-acetylcysteine inhibits chromium hypersensitivity in coadjuvant chromium-sensitized albino guinea pigs by suppressing the effects of reactive oxygen species. Exp Dermatol. 2010;19(8):e191–e200.
Bradford MM. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem. 1976;72:248–254.
Gostimskaya I, Galkin A. Preparation of highly coupled rat heart mitochondria. J Vis Exp. 2010;(43):3–5.
Kadenbach B, Huttemann M, Arnold S, Lee I, Bender E. Mitochondrial energy metabolism is regulated via nuclearcoded subunits of cytochrome c oxidase1. Free Radic Biol Med. 2000;29(3–4):211–221.
Oh C, Dong Y, Liu H, Thompson LP. Intrauterine hypoxia upregulates proinflammatory cytokines and matrix metalloproteinases in fetal guinea pig hearts. Am J Obstet Gynecol. 2008; 199(1):78.e1–e6.
Pacelli C, Latorre D, Cocco T, et al. Tight control of mitochondrial membrane potential by cytochrome c oxidase. Mitochondrion. 2011;11(2):334–341.
Villani G, Attardi G. In vivo control of respiration by cytochrome c oxidase in wild-type and mitochondrial DNA mutation-carrying human cells. Proc Natl Acad Sci U S A. 1997;94(4):1166–1171.
Hüttemann M, Helling S, Sanderson TH, et al. Regulation of mitochondrial respiration and apoptosis through cell signaling: cytochrome c oxidase and cytochrome c in ischemia/reperfusion injury and inflammation. Biochim Biophys Acta. 2012;1817(4): 598–609.
Hájek P, Villani G, Attardi G. Rate-limiting step preceding cytochrome c release in cells primed for Fas-mediated apoptosis revealed by analysis of cellular mosaicism of respiratory changes. J Biol Chem. 2001;276(1):606–615.
Kunz WS, Kudin A, Vielhaber S, Elger CE, Attardi G, Villani G. Flux control of cytochrome c oxidase in human skeletal muscle. J Biol Chem. 2000;275(36):27741–27745.
Chandel NS, McClintock DS, Feliciano CE, et al. Reactive oxygen species generated at mitochondrial complex III stabilize hypoxia-inducible factor-1alpha during hypoxia: a mechanism of O2 sensing. J Biol Chem. 2000;275(33): 25130–25138.
Waypa GB, Schumacker PT. Hypoxic pulmonary vasoconstriction: redox events in oxygen sensing. J Appl Physiol. 2005; 98(1):404–414.
Hassanein T, Frederick T. Mitochondrial dysfunction in liver disease and organ transplantation. Mitochondrion. 2004;4(5–6): 609–620.
Schönfeld P, Schlüter T, Fischer K-D, Reiser G. Non-esterified polyunsaturated fatty acids distinctly modulate the mitochondrial and cellular ROS production in normoxia and hypoxia. J Neurochem. 2011;118(1):69–78.
Turrens JF. Mitochondrial formation of reactive oxygen species. J Physiol. 2003;552(pt 2):335–344.
Fontanesi F, Soto IC, Barrientos A. Cytochrome c oxidase biogenesis: new levels of regulation. IUBMB Life. 2008;60(9):557–568.
Fornuskova D, Stiburek L, Wenchich L, Vinsova K, Hansikova H, Zeman J. Novel insights into the assembly and function of human nuclear-encoded cytochrome c oxidase subunits 4, 5a, 6a, 7a and 7b. Biochem J. 2010;428(3):363–374.
Stiburek L, Vesela K, Hansikova H, et al. Tissue-specific cytochrome c oxidase assembly defects due to mutations in SCO2 and SURF1. Biochem J. 2005;392(pt 3):625–632.
Hervouet E, Pecina P, Demont J, et al. Inhibition of cytochrome c oxidase subunit 4 precursor processing by the hypoxia mimic cobalt chloride. Biochem Biophys Res Commun. 2006;344(4):1086–1093.
Semenza GL. Oxygen-dependent regulation of mitochondrial respiration by hypoxia-inducible factor 1. Biochem J. 2007; 405(1):1–9.
Pinti M, Gibellini L, De Biasi S, et al. Functional characterization of the promoter of the human Lon protease gene. Mitochondrion. 2011;11(1):200–206.
Haddad JJ. Antioxidant and prooxidant mechanisms in the regulation of redox(y)-sensitive transcription factors. Cell Signal. 2002;14(11):879–897.
Tatsuta T, Langer T. Quality control of mitochondria: protection against neurodegeneration and ageing. EMBO J. 2008;27(2): 306–314.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Al-Hasan, Y.M., Evans, L.C., Pinkas, G.A. et al. Chronic Hypoxia Impairs Cytochrome Oxidase Activity Via Oxidative Stress in Selected Fetal Guinea Pig Organs. Reprod. Sci. 20, 299–307 (2013). https://doi.org/10.1177/1933719112453509
Published:
Issue Date:
DOI: https://doi.org/10.1177/1933719112453509