Abstract
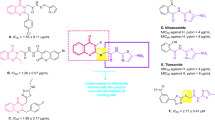
In this study, some benzimidazole-thiophene hybrid compounds containing isatin and salicyl moieties were synthesized and investigated for their urease and lipase inhibition properties. Most of the synthesized compounds were found to show effective antiurease activities, when compared with standard Thiourea (0.5027 ± 0.0293 µM). The synthesized isatin containing hybrids (IVa–IVc) showed antiurease inhibitory activities ranging from 0.411 ± 0.0337 to 0.431 ± 0.0446 µM, while IC50 of standard thiourea was 0.5027 ± 0.0293 µM. The general of salicyl containing hybrids showed low antiurease inhibitory activities, while compound (Ve) showed 0.338 ± 0.0387 µM activity. The lipase inhibition activities of the synthesized hybrid molecules showed the activities ranging from 0.287 ± 0.0341 to 0.912 ± 0.0832 µM, while the standard Orlistat showed 0.21 ± 0.012 µM activity. In the light of the information obtained from this study, the results showed that the synthesized hybrid compounds could be potential antilipase and antiurease agents.




Similar content being viewed by others
REFERENCES
Jin, D., Zhao, S., Zheng, N., Bu, D., Beckers, Y., Denman, S.E., McSweeney, C.S., and Wang, J., Front. Microbiol., 2017, vol. 8, pp. 1–10. https://doi.org/10.3389/fmicb.2017.00385
Biglar, M., Mirzazadeh, R., Asadi, M., Sepehri, S., Valizadeh, Y., Sarrafi, Y., Amanlou, M., Larijani, B., Mohammadi-Khanaposhtani, M., and Mahdavi, M., Bioorg. Chem., 2020, vol. 95, pp. 1–7. https://doi.org/10.1016/j.bioorg.2019.103529
Azizian, H., Nabati, F., Sharifi, A., Siavoshi, F., Mahdavi, M., and Amanlou, M. J., Mol. Model., 2012, vol. 18, pp. 2917–2927. https://doi.org/10.1007/s00894-011-1310-2
Sadat-Ebrahimi, S.E., Bigdelou, A., Sooreshjani, R.H., Montazer, M.N., Zomorodian, K., Irajie, C., Yahya-Meymandi, A., Biglar, M., Larijani, B., Amanlou, M., Iraji, A., and Mahdavi, M., J. Mol. Struct., 2022, vol. 1263, pp. 1–9. https://doi.org/10.1016/j.molstruc.2022.133078
Abid, O.-U.-R., Babar, T.M., Ali, F.I., Ahmed, S., Wadood, A., Rama, N.H., Uddin, R., Ul-Haq, Z., Khan, A., and Choudhary, M.I., ACS Med. Chem Lett., 2010, vol. 1, pp. 145–149. https://doi.org/10.1021/ml100068u
Pervez, H., Chohan, Z.H., Ramzan, M., Nasim, F.-U.-H., and Khan, K.M., J. Enzyme Inhib. Med. Chem., 2009, vol. 24, pp. 437–446. https://doi.org/10.1080/14756360802188420
Perveen, S., Khan, K.M., Lodhi, M.A., Choudhary, M.I., Atta-ur-Rahman, and Voelter, W., Lett. Drug Des. Discov., 2008, vol. 5, pp. 401–405. https://doi.org/10.2174/157018008785777315
Shamim, S., Khan, K.M., Salar, U., Ali, F., Lodhi, M.A., Taha, M., Khan, F.A., Ashraf, S., Ul-Haq, Z., Ali, M., Ali, M., and Perveen, S., Bioorg. Chem., 2018, vol. 76, pp. 37–52. https://doi.org/10.1016/j.bioorg.2017.10.021
Taha, M. and Wadood, A., Bioorg. Chem., 2018, vol. 78, pp. 411–417. https://doi.org/10.1016/j.bioorg.2018.04.007
de la Torre, A.F., Ali, A., Galetto, F.Z., Braga, A.L., Delgado, J.A.C., and Paixao, M.W., Mol. Divers., 2020, vol. 24, pp. 1–10. https://doi.org/10.1007/s11030-019-09923-w
Saeed, A., Imran, A., Channar, P.A., Shahid, M., Mahmood, W., and Iqbal, J., Chem. Biol. Drug Des., 2015, vol. 85, pp. 225–230. https://doi.org/10.1111/cbdd.12379
Aslam, M.A.S., Mahmood, S., Shahid, M., Saeed, A., and Iqbal, J., Eur. J. Med. Chem., 2011, vol. 46, pp. 5473–5479. https://doi.org/10.1016/j.ejmech.2011.09.009
Kafarski, P. and Talma, M., J. Adv. Res., 2018, vol. 13, pp. 101–112. https://doi.org/10.1016/j.jare.2018.01.007
Pi-Sunyer, X., Postgrad Med., 2009, vol. 121, pp. 21–33. https://doi.org/10.3810/pgm.2009.11.2074
Gregoire, F.M., Smas, C.M., and Sul, H.S., Physiol. Rev., 1998, vol. 78, pp. 783–809. https://doi.org/10.1152/physrev.1998.78.3.783
Zhi, J., Melia, A.T., Guerciolini, R., Chung, J., Kinberg, J., Hauptman, J.B., and Patel, I.H., Clin. Pharmacol. Ther., 1994, vol. 56, pp. 82–85. https://doi.org/10.1038/clpt.1994.104
Neovius, M., Johansson, K., and Rossner, S., Obes. Rev., 2008, vol. 9, pp. 420–427. https://doi.org/10.1111/j.1467-789X.2008.00463.x
Filippatos, T.D., Derdemezis, C.S., Gazi, I.F., Nakou, E.S., Mikhailidis, D.P., and Elisaf, M.S., DRUG Saf., 2008, vol. 31, pp. 53–65. https://doi.org/10.2165/00002018-200831010-00005
Viner, R.M., Hsia, Y., Tomsic, T., and Wong, I.C.K., Obes. Rev,. 2010, vol. 11, pp. 593–602. https://doi.org/10.1111/j.1467-789X.2009.00651.x
Menteşe, E., Yılmaz, F., Emirik, M., Ülker, S., and Kahveci, B., Bioorg. Chem., 2018, vol. 76, pp. 478–486. https://doi.org/10.1016/j.bioorg.2017.12.023
Kahveci, B., Yilmaz, F., Menteşe, E., Özil, M., and Karaoǧlu, S.A. J. Heterocycl. Chem., 2014, vol. 51, pp. 982–990. https://doi.org/10.1002/jhet.1593
Ali, I., Lone, M.N. and Aboul-Enein, H.Y., Med. Chem. Comm., 2017, vol. 8, pp. 1742–1773. https://doi.org/10.1039/C7MD00067G
Ajani, O.O., Tolu-Bolaji, O.O., Olorunshola, S.J., Zhao, Y., and Aderohunmu, D. V. J. Adv. Res., 2017, vol. 8, p. 703. https://doi.org/10.1016/j.jare.2017.09.003
El Rashedy, A.A. and Aboul-Enein, Y.H., Mini-Rev. Med. Chem., 2013, vol. 13, pp. 399–407. https://doi.org/10.2174/1389557511313030008
Menteşe, E., Yilmaz, F., Baltaş, N., Bekircan, O., and Kahveci, B., J. Enzyme Inhib. Med. Chem., 2015, vol. 30, p. 435–441. https://doi.org/10.3109/14756366.2014.943203
Pandy, K. M., Krishman, P., and Paratharaj, S., J. Het. Chem., 2022, vol. 59, pp. 1665–1883. https://doi.org/10.1002/jhet.4519
Ponticello, G.S., Freedman, M.B., Habecker, C.N., Lyle, P.A., Schwam, H., Varga, S.L., Christy, M.E., Randall, W.C., and Baldwin, J.J., J. Med. Chem., 1987, vol. 30, pp. 591–597. https://doi.org/10.1021/jm00387a002
Ivasiv, V., Albertini, C., Gonçalves, A., Rossi, M., and Bolognesi, M., Curr. Top. Med. Chem., 2019, vol. 19, p. 1694–1711. https://doi.org/10.2174/1568026619666190619115735
Menteşe, E., Yilmaz, F., Baltaş, N., Bekircan, O., and Kahveci, B., J. Enzyme Inhib. Med. Chem., 2015, vol. 30, pp. 435–441. https://doi.org/10.3109/14756366.2014.943203
Menteşe, E., Emirik, M., and Sökmen, B.B., Bioorg. Chem., 2019, vol. 86, pp. 151–158. https://doi.org/10.1016/j.bioorg.2019.01.061
Bendicho, S., Trigueros, M.C., Hernández, T., and Martín, O., J. Dairy Sci., 2001, vol. 84, pp. 1590–1596. https://doi.org/10.3168/jds.S0022-0302(01)74592-4
ACKNOWLEDGMENTS
The authors thank all the colleagues in the project unit for this support.
Funding
This work was supported by Research Fund of Recep Tayyip Erdogan University under the Project Number of FBA-2020-1071.
Author information
Authors and Affiliations
Contributions
Authors FY and EM designed the experiments, synthesized the molecules and carried out spectral studies. Author BBS carried our biological activity studies.
Corresponding author
Ethics declarations
The data that support the findings of this study are available from the corresponding author upon reasonable request. This article does not contain any studies involving animals or human participants performed by any of the authors. Informed consent was not required for this article. No conflict of interest was declared by the authors.
Additional information
Publisher's Note. Pleiades Publishing remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Yilmaz, F., Menteşe, E. & Sökmen, B.B. Synthesis of Some Benzimidazole-Thiophene Hybrids as Potential Urease and Lipase Inhibitors. Russ J Bioorg Chem 49, 1293–1300 (2023). https://doi.org/10.1134/S1068162023060092
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1068162023060092