Abstract
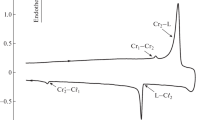
The centimeter-sized naphthalene, anthracene, and tetracene crystals have been grown from the vapor phase. An isothermal thermogravimetric method for determining the sublimation enthalpy during crystal growth under conditions of classical physical vapor transport is proposed. The sublimation enthalpy has been calculated using the obtained approximate equation for the temperature dependence of the intensity of the flux of molecules sublimating from the solid surface in the quasi-steady-state mode. The sublimation enthalpies of the linear acenes under study have been determined in narrow temperature ranges to be 71 ± 2 kJ mol–1 (328–353 K), 96 ± 3 kJ mol–1 (423–458 K), and 124 ± 11 kJ mol–1 (513–573 K) for naphthalene, anthracene, and tetracene, respectively. The found values are in good agreement with the experimental data in the literature.



Similar content being viewed by others
REFERENCES
R. A. Laudise, C. Kloc, P. G. Simpkins, and T. Siegrist, J. Cryst. Growth 187, 449 (1998). https://doi.org/10.1016/S0022-0248(98)00034-7
V. A. Postnikov, N. I. Sorokina, M. S. Lyasnikova, et al., Crystals 10, 363 (2020). https://doi.org/10.3390/cryst10050363
V. A. Postnikov, A. A. Kulishov, O. V. Borshchev, et al., Poverkhn.: Rentgenovskie, Sinkhrotronnye Neitr. Issled., No. 1, 28 (2021). https://doi.org/10.31857/s1028096021010131
V. A. Postnikov, A. A. Kulishov, M. S. Lyasnikova, et al., Zh. Fiz. Khim. 95, 1101 (2021). https://doi.org/10.31857/S0044453721070220
J. P. Murray, K. J. Cavell, and J. O. Hill, Thermochim. Acta 36, 97 (1980). https://doi.org/10.1016/0040-6031(80)80114-6
L. A. Torres-Gómez, G. Barreiro-Rodríguez, and A. Galarza-Mondragón, Thermochim. Acta 124, 229 (1988). https://doi.org/10.1016/0040-6031(88)87025-4
M. Knudsen, Ann. Phys. 333, 999 (1909). https://doi.org/10.1002/andp.19093330505
M. A. V. Ribeiro da Silva, M. J. S. Monte, and L. M. N. B. F. Santos, J. Chem. Thermodyn. 38, 778 (2006). https://doi.org/10.1016/j.jct.2005.08.013
D. Ambrose, I. J. Lawrenson, and C. H. S. Sprake, J. Chem. Thermodyn. 7, 1173 (1975). https://doi.org/10.1016/0021-9614(75)90038-5
C. G. de Kruif, T. Kuipers, J. C. van Miltenburget, et al., J. Chem. Thermodyn. 13, 1081 (1981). https://doi.org/10.1016/0021-9614(81)90006-9
O. T. Glukhova, N. M. Arkhangelova, A. B. Teplitsky, et al., Thermochim. Acta 95, 133 (1985). https://doi.org/10.1016/0040-6031(85)80041-1
B. T. Grayson and L. A. Fosbraey, Pestic. Sci. 13, 269 (1982). https://doi.org/10.1002/ps.2780130308
K. Nass, D. Lenoir, and A. Kettrup, Angew. Chem. Int. Ed. Engl. 34, 1735 (1995). https://doi.org/10.1002/anie.199517351
O. Shalev and M. Shtein, Org. Electron. 14, 94 (2013). https://doi.org/10.1016/j.orgel.2012.09.033
R. V. Ralys, G. S. Yablonsky, and A. A. Slobodov, Sci. Tech. J. Inf. Technol. Mech. Opt. 15, 1072 (2015). https://doi.org/10.17586/2226-1494-2015-15-6-1072-1080
W. Zielenkiewicz, G. L. Perlovich, and M. Wszelaka-Rylik, J. Thermal Anal. Calorim. 57, 225 (1999). https://doi.org/10.1023/A:1010179814511
P. V. Lebedev-Stepanov, Introduction into Self-Organization and Self-Assembly of Nanoparticle Ensembles (NRNU MEPhI, Moscow, 2015) [in Russian].
I. S. Grigor’ev and E. Z. Meilikhov, Physical Values: A Handbook (Energoatomizdat, Moscow, 1991) [in Russian].
L. D. Landau and E. M. Lifshitz, Course of Theoretical Physics, Vol. 5: Statistical Physics, Part 1 (Fizmatlit, Moscow, 2002; Pergamon, Oxford, 1984).
D. W. J. Cruickshank, Acta Crystallogr. 10, 504 (1957). https://doi.org/10.1107/s0365110x57001826
C. P. Brock and J. D. Dunitz, Acta Crystallogr. B 46, 795 (1990). https://doi.org/10.1107/S0108768190008382
J. M. Robertson, V. C. Sinclair, and J. Trotter, Acta Crystallogr. 14, 697 (1961). https://doi.org/10.1107/s0365110x61002151
R. D. Chirico, S. E. Knipmeyer, and W. V. Steele, J. Chem. Thermodyn. 34, 1873 (2002). https://doi.org/10.1016/S0021-9614(02)00262-8
P. Goursot, H. L. Girdhar, and E. F. Westrum, J. Phys. Chem. 74, 2538 (1970). https://doi.org/10.1021/j100706a022
M. Fulem, V. Laštovka, M. Straka, et al., J. Chem. Eng. Data 53, 2175 (2008). https://doi.org/10.1021/je800382b
B. Stevens, J. Chem. Soc. 2973 (1953). https://doi.org/10.1039/jr9530002973
L. Malaspina, R. Gigli, and G. Bardi, J. Chem. Phys. 59, 387 (1973). https://doi.org/10.1063/1.1679817
R. Bender, V. Bieling, and G. Maurer, J. Chem. Thermodyn. 15, 585 (1983). https://doi.org/10.1016/0021-9614(83)90058-7
F. Emmenegger and M. Piccand, J. Therm. Anal. Calorim. 57, 235 (1999). https://doi.org/10.1023/A:1010100531350
M. A. Siddiqi, R. A. Siddiqui, and B. Atakan, J. Chem. Eng. Data 54, 2795 (2009). https://doi.org/10.1021/je9001653
V. Oja, X. Chen, M. R. Hajaligol, and W. G. Chan, J. Chem. Eng. Data 54, 730 (2009). https://doi.org/10.1021/je800395m
P. E. Fielding and A. G. Mackay, Aust. J. Chem. 17, 1288 (1964). https://doi.org/10.1071/CH9641288
G. C. Morris, J. Mol. Spectrosc. 18, 42 (1965). https://doi.org/10.1016/0022-2852(65)90059-7
N. Wakayama and H. Inokuchi, Bull. Chem. Soc. Jpn. 40, 2267 (1967). https://doi.org/10.1246/bcsj.40.2267
V. Oja and E. M. Suuberg, J. Chem. Eng. Data 43, 486 (1998). https://doi.org/10.1021/je970222l
ACKNOWLEDGMENTS
The experiments were carried out using equipment of the Center for Collective Use “Structural Diagnostics of Materials.”
Funding
This study was supported by the Ministry of Science and Higher Education of the Russian Federation within the State assignment for the Federal Scientific Research Centre “Crystallography and Photonics” of the Russian Academy of Sciences.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflicts of interest.
Additional information
Translated by A. Sin’kov
Rights and permissions
About this article
Cite this article
Postnikov, V.A., Kulishov, A.A., Yurasik, G.A. et al. Growth of Linear Acene Crystals and Determination of Their Sublimation Enthalpy under Conditions of Physical Vapor Transport. Crystallogr. Rep. 67, 608–615 (2022). https://doi.org/10.1134/S1063774522040137
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1063774522040137