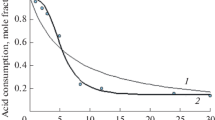
Abstract
It was supposed that, in the reaction of oxyethylation of alcohols, the kinetically independent unit is not a monomeric alcohol molecule, but a linear chain alcohol associate consisting of an average of n alcohol molecules, and that the reaction rate is of the first order not in the total concentration of all alcohol species, but in the concentration of these associates. The kinetic aspects of this phenomenon were considered. It was assumed that reactions of the first order in the concentration of an associated component, which are widespread in liquid-phase chemical kinetics, occur as a result of the release of the reaction product from the associate.






Similar content being viewed by others
REFERENCES
Stul’, B.Ya. and Chesnokov, B.B., Kinet. Katal., 2002, vol. 43, no. 5, p. 741.
Stul’, B.Ya., Russ. J. Phys. Chem. A, 2016, vol. 90, p.754.
Stul’, B.Ya., Russ. J. Phys. Chem. A, 2020, vol. 94, p. 532.
Stul’, B.Ya., Russ. J. Phys. Chem. A, 2021, vol. 95, p. 2025.
Stul’, B.Ya., Russ. J. Phys. Chem A, 2022, vol. 96, p. 1445.
Shvets, V.F. and Tsivinskii, D.N., Kinet. Katal., 1981, vol. 22, no. 5, p. 1192.
Shakhparonov, M.I., Mekhanizmy bystrykh protsessov v zhidkostyakh (Mechanisms of Fast Processes in Liquids), Moscow: Vysshaya Shkola, 1980.
Lu, W. and Yang, S., J. Phys. Chem. A, 1998, vol. 102, no. 5, p. P. 825.
Smirnova, N.A., Molekulyarnye teorii rastvorov (Molecular Theories of Solutions), Leningrad: Khimiya, 1987.
Shvets, V.F., Tsivinskii, D.N., and Makarov, M.G., Khi-m. Prom-st., 1978, no. 10, p. 737.
Organic Solvents. Physical Properties and Methods of Purification, Riddick, J.A. and Toops, E.E., Eds., New York: Interscience, 1955, 2nd ed.
Stul’, B.Ya., Asriev, S.D., and Chesnokov, B.B., Kinet. Katal., 1995, vol. 36, no. 3, p. 413.
Stul’, B.Ya., Archives of Chemistry and Chemical Engineering, 2022, vol. 4, no. 1.
Brazhkin, V.V., Phys.-Usp., 2009, vol. 52, no. 4, p. 369.
Funding
This work was supported by ongoing institutional funding. No additional grants to carry out or direct this particular research were obtained.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
As author of this work, I declare that I have no conflicts of interest.
Additional information
Translated by V. Glyanchenko
Abbreviations and notation: EO, ethylene oxide; GLC, gas-liquid chromatography; n, average number of alcohol molecules in a linear chain alcohol associate; W, reaction rate; k0,obs, k1,obs, and k2,obs, observed rate constants of the first, second, and third steps of the reaction, respectively, related to the total concentration of components; k0,n and k1,n, rate constants of the first and second steps of the reaction, respectively, related to the concentration of associates; \(C_{{\text{o}}}^{0}\) and Co, initial and current concentrations of EO, respectively; \(C_{{\text{c}}}^{0}\) and Сс, initial and current concentrations of alcohol, respectively; \(C_{{\text{1}}}^{0},\) C1, and \(C_{{\text{2}}}^{0},\) C2, initial and current concentrations of the first and second reaction products, respectively; Cass, the concentration of associates; Сcat, catalyst concentration; and τ, residence time of the reaction mixture in the reactor.
Rights and permissions
About this article
Cite this article
Stul’, B.Y. Kinetics of the Oxyethylation of Alcohols with Consideration of the Effect of Association: Kinetic Aspects of Association. Kinet Catal 64, 741–749 (2023). https://doi.org/10.1134/S0023158423060162
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0023158423060162