Abstract
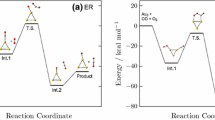
The interaction of CO and O2 and the subsequent oxidation of CO in the presence of cyclic thiolate and dithiolate complexes of Au(I), which represent the model fragments of thiolate-protected gold clusters, were studied using the density functional theory (PBE). On the basis of the calculated values, it was shown that O2 and CO were weakly bound to a cyclic thiolate complex of Au(I). In the presence of a dithiolate complex, the activation of O2 and CO and the subsequent oxidation of CO occurred with low activation energies. The results obtained demonstrate the important role of ligands in the catalytic process.




Similar content being viewed by others
REFERENCES
Bond, G.C., Louis, C., and Thompson, D.T., Catalysis by Gold, London: Imperial College, 2007.
Stratakis, M. and Garcia, H., Chem. Rev., 2012, vol. 112, no. 8, p. 4469.
Pichugina, D.A., Kuz’menko, N.E., and Shestakov, A.F., Russ. Chem. Rev., 2015, vol. 84, no. 11, p. 1114.
Li, G. and Jin, R., Acc. Chem. Res., 2014, vol. 47, no. 3, p. 816.
Zhang, G., Wang, R., and Li, G., Chin. Chem. Lett., 2018, vol. 29, no. 5, p. 687.
Akola, J., Walter, M., Whetten, R.L., Häkkinen, H., and Grönbeck, H., J. Am. Chem. Soc., 2008, vol. 130, no. 12, p. 3756.
Heaven, M.W., Dass, A., White, P.S., Holt, K.M., and Murray, R.W., J. Am. Chem. Soc., 2008, vol. 130, no. 12, p. 3754.
Qian, H., Eckenhoff, W.T., Zhu, Y., Pintauer, T., and Jin, R., J. Am. Chem. Soc., 2010, vol. 132, no. 24, p. 8280.
Crasto, D., Malola, S., Brosofsky, G., Dass, A., and Häkkinen, H., J. Am. Chem. Soc., 2014, vol. 136, no. 13, p. 5000.
Das, A., Li, T., Li, G., Nobusada, K., Zeng, C., Rosi, N.L., and Jin, R., Nanoscale, 2014, vol. 6, p. 6458.
Zeng, C., Liu, C., Chen, Y., Rosi, N.L., and Jin, R., J. Am. Chem. Soc., 2014, vol. 136, no. 34, p. 11922.
Wang, P., Xiong, L., Sun, X., Ma, Z., and Pei, Y., Nanoscale, 2018, vol. 10, no. 8, p. 3918.
Yoskamtorn, T., Yamazoe, S., Takahata, R., Nishigaki, J., Thivasasith, A., Limtrakul, J., and Tsukuda, T., ACS Catal., 2014, vol. 4, no. 10, p. 3696.
Li, G. and Jin, R., Nanotechnol. Rev., 2013, vol. 2, no. 5, p. 529.
Li, G. and Jin, R., J. Am. Chem. Soc., 2014, vol. 136, no. 32, p. 11347.
Liu, Y., Tsunoyama, H., Akita, T., Xie, S., and Tsukuda, T., ACS Catal., 2011, vol. 1, no. 1, p. 2.
Kurashige, W., Yamaguchi, M., Nobusada, K., and Negishi, Y., J. Phys. Chem. Lett., 2012, vol. 3, no. 18, p. 2649.
Harkness, K.M., Cliffel, D.E., and McLean, J.A., Analyst, 2010, vol. 135, no. 5, p. 868.
Nie, X., Qian, H., Ge, Q., Xu, H., and Jin, R., ACS Nano, 2012, vol. 6, p. 6014.
Wu, Z., Jiang, D.E., Mann, A.K., Mullins, D.R., Qiao, Z.A., Allard, L.F., Zeng, C., Jin, R., and Overbury, S.H., J. Am. Chem. Soc., 2014, vol. 136, no. 16, p. 6111.
Nie, X., Zeng, C., Ma, X., Qian, H., Ge, Q., Xu, H., and Jin, R., Nanoscale, 2013, vol. 5, p. 5912.
Li, Y., Chen, Y., House, S.D., Zhao, S., Wahab, Z., Yang, J.C., and Jin, R., ACS Appl. Mater. Interfaces, 2018, vol. 10, no. 35, p. 29425.
Bonasia, P.J., Gindelberger, D.E., and Arnold, J., Inorg. Chem., 1993, vol. 32, no. 23, p. 5126.
Perdew, J.P., Burke, K., and Ernzerhof, M., Phys. Rev. Lett., 1996, vol. 77, no. 18, p. 3865.
Stevens, W.J., Krauss, M., Basch, H., and Jasien, P.G., Can. J. Chem., 1992, vol. 70, no. 2, p. 612.
Nikitina, N.A., Pichugina, D.A., and Kuz’menko, N.E., Russ. J. Phys. Chem. A, 2017, vol. 91, no. 8, p. 1507.
Pichugina, D.A., Shestakov, A.F., and Kuz’menko, N.E., Kinet. Catal., 2007, vol. 48, no. 2, p. 305.
Askerka, M., Pichugina, D., Kuz’menko, N., and Shestakov, A., J. Phys. Chem. A, 2012, vol. 116, no. 29, p. 7686.
Pichugina, D.A., Nikolaev, S.A., Mukhamedzyanova, D.F., and Kuz’menko, N.E., Russ. J. Phys. Chem. A, 2014, vol. 88, no. 6, p. 959.
Polynskaya, Y.G., Pichugina, D.A., and Kuz’menko, N.E., Comput. Theor. Chem., 2015, vol. 1055, p. 61.
Schlegel, H.B., J. Comput. Chem., 1982, vol. 3, no. 2, p. 214.
Gonzalez, C., and Schlegel, H.B., J. Chem. Phys., 1989, vol. 90, no. 4, p. 2154.
Laikov, D.N. and Ustynyuk, Yu.A., Russ. Chem. Bull., 2005, no. 3, p. 820.
Sadovnichy, V., Tikhonravov, A., Voevodin, Vl., and Opanasenko, V., Contemporary High Performance Computing: From Petascale toward Exascale, Boca Raton: CRC Press, 2013, p. 283.
Kawasaki, H., Kumar, S., Li, G., Zeng, C., Kauffman, D.R., Yoshimoto, J., Iwasaki, Y., and Jin, R., Chem. Mater., 2014, vol. 26, no. 9, p. 2777.
Li, Z., Liu, C., Abroshan, H., Kauffman, D.R., and Li, G., ACS Catal., 2017, vol. 7, no. 5, p. 3368.
Li, L., Gao, Y., Li, H., Zhao, Y., Pei, Y., Chen, Z., and Zeng, X.C., J. Am. Chem. Soc., 2013, vol. 135, no. 51, p. 19336.
Zeng, W., Tang, J., Wanga, P., and Pei, Y., RSC Adv., 2016, vol. 6, p. 55867.
Haruta, M., Gold Bull., 2004, vol. 37, p. 27.
Bond, G.C. and Thompson, D.T., Gold Bull., 2000, vol. 33, p. 41.
Knoppew, S. and Bürgi, T., Phys. Chem. Chem. Phys., 2013, vol. 15, p. 15 816.
ACKNOWLEDGMENTS
This work was performed using the equipment of the Center for Collective Use of High-Performance Computing Resources at the Moscow State University.
Funding
This work was supported by the Russian Foundation for Basic Research (grant no. 17-03-00962).
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated by Valentin Makhlyarchuk
Rights and permissions
About this article
Cite this article
Nikitina, N.A., Pichugina, D.A. & Kuz’menko, N.E. Simulation of CO Oxidation in the Presence of Cyclic Gold Thiolate Complexes: The Effect of a Ligand. Kinet Catal 60, 606–611 (2019). https://doi.org/10.1134/S0023158419050033
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0023158419050033