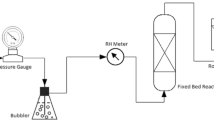
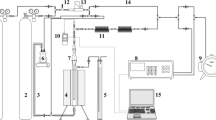
Abstract
This study concerns the effect of water-soluble polymers with different structures on the sorption properties of unregenerable lime-based sorbents of carbon dioxide. It is shown that introducing water-soluble polymers into the composition of sorbents can either prolong or shorten the periods of their protective effect. To explain these findings, the porous structure of sorbents is studied, the transport of carbon dioxide is modeled using molecular dynamics, and coefficients of the diffusion of СО2 in water–polymer solutions are calculated. Modelling results correlate with data from sorption experiments: a stronger dynamic sorption capacity is obtained for a sorbent when a water–polymer medium with a greater coefficient of СО2 diffusion is added. These results can be used to optimize systems for separating carbon dioxide from gaseous mixtures and intensify mass transfer in systems for the photo- and electrocatalytic conversion of СО2 into useful products.





Similar content being viewed by others
REFERENCES
Udara Willhelm Abeydeera, L.H., Wadu Mesthrige, J., and Samarasinghalage, T.I., Sustainability, 2019, vol. 11, no. 14, article no. 3972. https://doi.org/10.3390/su11143972
Ameyaw, B., Yao, L., Oppong, A., and Agyeman, J.K., Energy Policy, 2019, vol. 130, pp. 7–21. https://doi.org/10.1016/j.enpol.2019.03.056
Gao, J., Hou, H., Zhai, Y., Woodward, A., Vardoulakis, S., Kovats, S., Wilkinson, P., Li, L., Song, X., Xu, L., Meng, B., Liu, X., Wang, J., Zhao, J., and Liu, Q., Environ. Pollut., 2018, vol. 240, pp. 683–698. https://doi.org/10.1016/j.envpol.2018.05.011
Fasihi, M., Efimova, O., and Breyer, C., J. Cleaner Prod., 2019, vol. 224, pp. 957–980. https://doi.org/10.1016/j.jclepro.2019.03.086
Vega, F., Baena-Moreno, F.M., Gallego Fernández, L.M., Portillo, E., Navarrete, B., and Zhang, Z., Appl. Energy, 2020, vol. 260, article no. 114313. https://doi.org/10.1016/j.apenergy.2019.114313
Varghese, A.M. and Karanikolos, G.N., Int. J. Greenhouse Gas Control, 2020, vol. 96, article no. 103005. https://doi.org/10.1016/j.ijggc.2020.103005
Lee, J.-Y., Park, C.-Y., Moon, S.-Y., Choi, J.-H., Chang, B.-J., and Kim, J.-H., J. Membr. Sci., 2019, vol. 589, article no. 117214. https://doi.org/10.1016/j.memsci.2019.117214
Salih, H.A., Pokhrel, J., Reinalda, D., AlNashf, I., Khaleel, M., Vega, L.F., Karanikolos, G.N., and Abu Zahra, M., Int. J. Greenhouse Gas Control, 2021, vol. 110, article no. 103415. https://doi.org/10.1016/j.ijggc.2021.103415
Yu, W., Wang, T., Park, A.A., and Fang, M., Nanoscale, 2019, vol. 11, no. 37, pp. 17137–17156. https://doi.org/10.1039/C9NR05089B
Devendiran, D.K. and Amirtham, V.A., Renewable Sustainable Energy Rev., 2016, vol. 60, pp. 21–40. https://doi.org/10.1016/j.rser.2016.01.055
Kuehnel, M. F., Orchard, K.L., Dalle, K.E., and Reisner, E., J. Am. Chem. Soc., 2017, vol. 139, no. 21, pp. 7217–7223. https://doi.org/10.1021/jacs.7b00369
Resasco, J. and Bell, A.T., Trends Chem., 2020, vol. 2, no. 9, pp. 825–836. https://doi.org/10.1016/j.trechm.2020.06.007
Olmeda, B., Villén, L., Cruz, A., Orellana, G., and Perez-Gil, J., Biochim. Biophys. Acta, Biomembr., 2010, vol. 1798, no. 6, pp. 1281–1284. https://doi.org/10.1016/j.bbamem.2010.03.008
Derevschikov, V.S., Kazakova, E.D., Yatsenko, D.A., and Veselovskaya, J.V., Sep. Sci. Technol., 2021, vol. 56, no. 3, pp. 485–497. https://doi.org/10.1080/01496395.2020.1723029
Derevshchikov, V.S. and Kazakova, E.D., Catal. Ind., 2020, vol. 12, no. 1, pp. 1–6. https://doi.org/10.1134/S2070050420010043
Gladyshev, N.F., Gladysheva, T.V., Putin, B.V., and Putin, S.B., Izvestkovye poglotiteli novogo pokoleniya (Lime-Based Sorbents of New Generation), Moscow: Spektr, 2012.
Gladysheva, T.V., Gladyshev, N.F., Dvoretskii, S.I., and Suvorova, Yu.A., Izvestkovye khemosorbenty: poluchenie, svoistva, primenenie (Lime-Based Chemosorbents: Synthesis, Properties, Application), Moscow: Spektr, 2015.
Yurkevich, A.A., Ivakhnyuk, G.K., and Fedorov, N.F., Tekhnologicheskie osnovy proizvodstva khimicheskikh komponentov sistem zhizneobespecheniya (Technological Principles for the Production of Chemical Components for Life Support Systems), St. Petersburg: Mendeleev, 2014.
Ivakhnyuk, G.K., Kozhina, T.G., Samonin, V.V., Fedorov, N.F., and Slesareva, M.O., Zh. Prikl. Khim., 1991, vol. 64, no. 3, pp. 578–582.
Samonin, V.V., Kruglikova, I.V., and Fedorov, N.F., Zh. Prikl. Khim., 1994, vol. 67, no. 2, pp. 300–302.
Koreshonkova, M.O., Ivakhnyuk, G.K., Krylov, V.K., and Malinin, V.R., Zh. Prikl. Khim., 1997, vol. 70, no. 10, pp. 1743–1744.
Murray, J.M., Renfrew, C.W., Bedi, A., McCrystal, C.B., Jones, D.S., and Fee, J.P., Anesthesiology, 1999, vol. 91, no. 5, pp. 1342–1348. https://doi.org/10.1097/00000542-199911000-00026
Derevshchikov, V.S., Kazakova, E.D., Veselovskaya, J.V., Yatsenko, D.A., and Kozlov, D.V., Russ. J. Phys. Chem. A, 2021, vol. 95, no. 7, pp. 1455–1460. https://doi.org/10.1134/S0036024421070098
Lowell, S., Shields, J.E., Thomas, M.A., and Thommes, M., Characterization of Porous Solids and Powders: Surface Area, Pore Size and Density, New York: Springer, 2004. https://doi.org/10.1007/978-1-4020-2303-3
Oostenbrink, C., Villa, A., Mark, A.E., and Van Gunsteren, W.F., J. Comput. Chem., 2004, vol. 25, no. 13, pp. 1656–1676. https://doi.org/10.1002/jcc.20090
Essmann, U., Perera, L., Berkowitz, M.L., Darden, T., Lee, H., and Pedersen, L.G., J. Chem. Phys., 1995, vol. 103, no. 19, pp. 8577–8593. https://doi.org/10.1063/1.470117
Parrinello, M. and Rahman, A., J. Appl. Phys., 1981, vol. 52, no. 12, pp. 7182–7190. https://doi.org/10.1063/1.328693
Hoover, W.G., Phys. Rev. A, 1985, vol. 31, no. 3, pp. 1695–1697. https://doi.org/10.1103/PhysRevA.31.1695
ALOthman, Z.A., Materials, 2012, vol. 5, no. 12, pp. 2874–2902. https://doi.org/10.3390/ma5122874
Rouquerol, J., Avnir, D., Fairbridge, C.W., Everett, D.H., Haynes, J.H., Penicone, N., Ramsay, J.D.F., Sing, K.S.W., and Unger, K.K., Pure Appl. Chem., 1994, vol. 66, no. 8, pp. 1739–1758. https://doi.org/10.1351/pac199466081739
Lange, K.R., Surfactants: A Practical Handbook, Munich: Hanser Publishers/Cincinnati, OH: Hanser Gardner, 1999.
Danckwerts, P.V., Gas-Liquid Reactions, New York: McGraw-Hill, 1970.
Funding
This work was supported by the RF Ministry of Science and Higher Education as part of a state task for the Boreskov Institute of Catalysis, project no. AAAA-A21-121011390054-1 (0239-2021-0010). Our molecular dynamics modelling was supported in part by the RF Ministry of Science and Higher Education as part of a state task for the Voevodsky Institute of Chemical Kinetics and Combustion, project no. 0304-2017-0009.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflicts of interest.
Additional information
Translated by E. Glushachenkova
Publisher’s Note.
Pleiades Publishing remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Derevshchikov, V.S., Selyutina, O.Y. Effect of Water-Soluble Polymers on the Dynamics of Carbon Dioxide Sorption by Lime-Based Sorbents. Catal. Ind. 15, 325–332 (2023). https://doi.org/10.1134/S2070050423040062
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S2070050423040062