Abstract
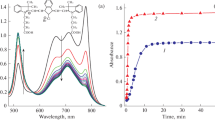
Using the example of meso-alkylsubstituted thiacarbocyanine dyes, it has been demonstrated that an ethyl substituent, as a rule, promotes the formation of long-wave J-aggreagtes in aqueous solutions and a methyl substituent favors the formation of short-wave H*-aggregates. The formation of dye aggregates proceeds through dimers by the block mechanism. The formation of three bands in the absorption region of dimers, which plausibly relate to three different forms of dimers and can be attributed to dimers consisting of the cis-, cis-/trans-, and trans-conformations of the dyes, has been recorded for the first time in meso-CH3-substituted thiacarbocyanines. The formation of three different types of H*-aggregates from the three appropriate dimeric forms has also been shown for the first time. It has been established that introducing a multicharged Eu+3 cation into aqueous solutions of dyes shifts the equilibrium, nD-aggregate, towards aggregates; meso-CH3-substituted dyes in this case shift towards H*-aggregates and meso-C2H5-substituted dyes shift towards J-aggregates. Hence it has been shown that an alkyl group in the meso-position to the polymethine chain of thiacarbocyanines plays the role of a regulating agent in the aggregation process due to stereochemical effects and, by this means, predetermines the morphology and spectral properties of the formed aggregate.
Similar content being viewed by others
References
V. I. Avdeeva and B. I. Shapiro, Zh. Nauch. Prikl. Fotogr. 44(2), 20 (1999).
V. I. Avdeeva and B. I. Shapiro, Zh. Nauch. Prikl. Fotogr. 45(6), 27 (2000).
B. I. Shapiro, Russ. Chem. Rev. 75, 433 (2006).
B. I. Shapiro, Nanotechnol. Russia 3, 139 (2008).
T. H. James, The Theory of the Photographic Process (Macmillan, New York, 1977; Khimiya, Leningrad, 1980) [in Russian].
A. H. Herz, Adv. Colloid. Interface Sci. 8, 237 (1977).
J-Aggregates, Ed. by T. Kobayashi (World Scientific, Singapore, New Jersey, London, Hong Kong, 1996).
B. I. Shapiro, Theoretical Fundamentals of Photographic Process (Editorial URSS, Moscow, 2000) [in Russian].
A. K. Chibisov, H. Gorner, and T. D. Slavnova, Chem. Phys. Lett. 390, 240 (2004)
T. D. Slavnova, A. K. Chibisov, and H. Gorner, J. Phys. Chem. A 109, 4758 (2005).
B. I. Shapiro, E. A. Belonozhkina, and V. A. Kuz’min, Nanotechnol. Russia 4, 38 (2009).
A. D. Nekrasov and B. I. Shapiro, High Energy Chem. 45, 133 (2011).
B. I. Shapiro, A. N. Isaeva, and V. A. Tverskoi, Nanotechnol. Russia 5, 427 (2010).
A. M. Kolesnikov and F. A. Mikhailenko, Usp. Khim. 56, 466 (1987).
A. D. Nekrasov, B. I. Shapiro, A. I. Tolmachev, Yu. L. Slominskii, and V. A. Kuz’min, High Energy Chem. 45, 525 (2011).
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © B.I. Shapiro, L.S. Sokolova, V.A. Kuz’min, A.I. Tolmachev, Yu.L. Slominskii, Yu.L. Briks, 2012, published in Rossiiskie Nanotekhnologii, 2012, Vol. 7, Nos. 5–6.
Rights and permissions
About this article
Cite this article
Shapiro, B.I., Sokolova, L.S., Kuz’min, V.A. et al. Effect of meso-alkyl substituents in the polymethine chain of thiacarbocyanines on the morphology of dye aggregates. Nanotechnol Russia 7, 205–212 (2012). https://doi.org/10.1134/S1995078012030159
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1995078012030159