Abstract
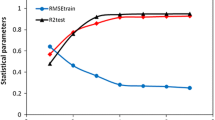
A new approach for quantitative structure-activity relationships based on MNA descriptors, fuzzy gradation method and self-consistent regression has been proposed. This approach has been realized in the computer program GUSAR (General Unrestricted Structure Activity Relationships). The method has been validated using inhibitors of cyclin-dependent kinase 1 (CDK). Prediction accuracy is comparable with that of 3D QSAR: CoMFA and CoMSIA. However, in contrast to CoMFA and CoMSIA, GUSAR approach does not require information about 3D structure of an enzyme and a ligand. Application of GUSAR method for heterogeneous training sets containing chemical compounds from different chemical classes has been shown.
Similar content being viewed by others
References
Morgan, D.O., Nature, 1995, vol. 374, pp. 131–134.
Wolfel, T., Hauer, M., Schneider, J., Serrano, M., Wolfel, C., Klehmann, H., De Plaen, E., Hankeln, T., Meyerzum-Buschenfelde, K., and Beach, D., Science, 1995, vol. 269, p. 1281.
Livingstone, D., Data Analysis for Chemists. Applications to QSAR and Chemical Product Design, Oxford University Press, 1995.
Kubinyi, H., in QSAR and Molecular Modelling: Concepts, Computation Tools and Biological Applications, Sanz, F., Giraldo, J., and Manaut, F., Eds., Barcelona: J.P. Prous Science Publishers, 1995, pp. 2–18.
Kubinyi, H., 3D QSAR in Drug Design. Theory Methods and Applications, Leiden: ESCOM, 1993.
Leach, A., Molecular Modelling. Principles and Applications, Prentice Hall, 2001.
Gasteiger, J., Handbook of Chemoinformatics. From Data to Knowledge in 4 Volumes, Weinheim: WILEYVCH Verlag GmbH & Co. KgaA, 2003.
Filimonov, D., Poroikov, V., Borodina, Yu., Gloriozova, T., J. Chem. Inf. Comput. Sci., 1999, vol. 39, no. 4, pp. 666–670.
Filimonov, D.A., Akimov, D.V., and Poroikov, V.V., Khim.-Farm. Zh., 2004, vol. 38, no. 1, pp. 21–24.
Sybyl, 6.3 ed.; SYBYL Molecular Modeling Software, Tripos Associates Ltd.: St. Louis, MO, 1992. (www.tripos.com).
Cramer, R.D., III, Patterson, D.E., and Bunce, J.D., J. Am. Chem. Soc., 1988, vol. 110, pp. 5959–5967.
Klebe, G., Abraham, U., and Mietzner, T., J. Med. Chem., 1994, vol. 37, pp. 4130–4146.
Ducrot, P., Legraverend, M., and David, S., J. Med. Chem., 2000, vol. 43, pp. 4098–4108.
Kunick, C., Lauenroth, K., Wieking, K., Xu, X., Schultz, C., Gussio, R., Zaharevitz, D., Leost, M., Meijer, L., Weber, A., Flemming, S., and Lemcke, T., J. Med. Chem., 2004, vol. 47, pp. 22–36.
Kunick, C., Zeng, Z., Gussio, R., Zaharevitz, D., Leost, M., Totzke, F., Schachtele, C., Kubbutat, M., Meijer, L., and Thomas, L., ChemBioChem., 2005, vol. 6, pp. 1–9.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © A.V. Zakharov, A.A. Lagunin, D.A. Filimonov, V.V. Poroikov, 2007, published in Biomeditsinskaya Khimiya.
Rights and permissions
About this article
Cite this article
Zakharov, A.V., Lagunin, A.A., Filimonov, D.A. et al. Quantitative structure-activity relationships of cyclin-dependent kinase 1 inhibitors. Biochem. Moscow Suppl. Ser. B 1, 17–28 (2007). https://doi.org/10.1134/S1990750807010039
Received:
Issue Date:
DOI: https://doi.org/10.1134/S1990750807010039