Abstract
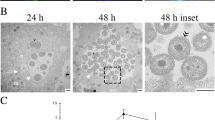
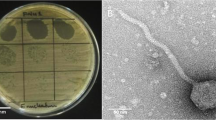
Cells isolated from a body are resistant to bacterial invasion but lose the resistance upon immortalization and transformation. In this work, interaction of immortalized 3T3 fibroblasts and their in vitro transformed analog 3T3-SV40 cells with opportunistic bacteria Serratia grimesii was studied in a conventional medium and after incubating the cells with an antioxidant N-acetylcysteine (NAC). The 3T3 cells were shown to be approximately twice less sensitive to S. grimesii infection than similar but virus-transformed 3T3- SV40 cells. Incubation of 3T3 cells with 10 and 20 mM NAC enhanced the invasion 1.6 and 2.5-fold, respectively. Under the same conditions, the invasion of 3T3-SV40 cells by the bacteria was enhanced 2.1 and 2.4- fold. These results show that 3T3 cells are more resistant to invasion by S. grimesii than 3T3-SV40 cells, and the difference is preserved after the cells are exposed to 10 and 20 mM NAC. Among the genes which expression is known to be increased by NAC, a special role plays E-cadherin shown to interact with surface proteins (invasins) of pathogenic bacteria. Incubation of 3T3 and 3T3-SV40 cells with NAC resulted in an increased expression of E-cadherin, which correlates with the increased sensitivity of these cells to invasion. Confocal fluorescence microscopy revealed, for the first time, colocalization of S. grimesii with E-cadherin of 3T3 and 3T3-SV40 cells indicating that E-cadherin can be involved in the penetration of S. grimesii into eukaryotic cells.
Similar content being viewed by others
References
Bonazzi, M., Lecuit, M., and Cossart, P., Listeria monocytogenes internalin and E-cadherin: from bench to bedside, Cold Spring Harb. Perspect. Biol., 2009, vol. 1, p. a003087. doi 10.1101/cshperspect.a003087
Bozhokina, E.S., Khaitlina, S.Yu., and Adam, T., Grimelysin, a novel metalloprotease from Serratia grimesii, is similar to ECP32, Biochem. Biophys. Res. Commun., 2008, vol. 367, pp. 888–892.
Bozhokina, E.S., Tsaplina, O.A., Efremova, T.N., Kever, L.V., Demidyuk, I.V., Kostrov, S.V., Adam, T., Komissarchik, Y.Y., and Khaitlina, S.Y., Bacterial invasion of eukaryotic cells can be mediated by actin-hydrolyzing metalloproteases grimelysin and protealysin, Cell Biol. Intern., 2011, vol. 34, pp. 111–118.
Bozhokina, E.S., Vakhromova, E.N., Gamaley, I.A., and Khaitlina, S.Yu., N-acetylcysteine increases susceptibility of HeLa cells to bacterial invasion, J. Cell. Biochem., 2013, vol. 114, pp. 1568–1574.
Bozhokina, E., Khaitlina, S., and Gamaley, I., Dihydrolipoic but not alpha-lipoic acid affects susceptibility of eukaryotic cells to bacterial invasion, Biochim. Biophys. Res. Commun., 2015, vol. 460, pp. 697–702.
Carabeo, R., Bacterial subversion of host actin dynamics at the plasma membrane, Cell Microbiol., 2011, vol. 13, pp. 1460–1469. eCollection 2017. doi 10.3389/fcimb.2017.00064
Cossart, P. and Sansonetti, P.J., Bacterial invasion: the paradigms of enteroinvasive pathogens, Science, 2004, vol. 304, pp. 242–248.
Efremova, T.N., Ender, N.A., Brudnaya, M.S., Komissarchik, Ya.Yu., and Khaitlina, S.Yu., Invasion of Escherichia coli A2 induces reorganization of actin microfilaments in Hep-2 cells, Tsitologiia, 1998, vol. 40, no. 6, pp. 524–528.
Efremova, T., Ender, Í., Brudnaja, M., and Komissarchik, Y. Khaitlina, S., Specific invasion of transformed cells by Escherichia coli A2 strain, Cell Biol. Intern., 2001, vol. 25, pp. 257–261.
Finlay, B.B. and Cossart, P., Exploitation of mammalian host cell functions by bacterial pathogens, Science, 1997, vol. 276, pp. 718–725.
Gamaley, I., Efremova, T., Kirpichnikova, K., Kever, L., Komissarchik, Y., Polozov, Y., and Khaitlina, S., N-acetylcystein-induced changes in susceptibility of transformed eukaryotic cells to bacterial invasion, Cell Biol. Intern., 2006, vol. 30, pp. 319–325.
Grimont, F. and Grimont, P.A.D., The fenus Serratia, Prokaryotes, 2006, vol. 6, pp. 219–244.
Gustafsson, A.C., Kupershmidt, I., Edlundh-Rose, E., Greco, G., Serafino, A., Krasnowska, E.K., Lundeberg, T., Bracci-Laudiero, L., Romano, M.C., Parasassi, T., and Lundeberg, J., Global gene expression analysis in time series following N-acetyl-L-cysteine induced epithelial differentiation of human normal and cancer cells in vitro, BMC Cancer, 2005, vol. 5, p. 75. doi 10.1186/1471-2407-5-75
Haglund, C.M. and Welch, M.D., Pathogens and polymers: microbe–host interactions illuminate the cytoskeleton, J. Cell Biol., 2011, vol. 195, pp. 7–17.
Hauck, C.R., Borisova, M., and Muenzner, P., Exploitation of integrin function by pathogenic microbes, Curr. Opin. Cell Biol., 2012, vol. 24, pp. 637–644.
Hoffmann, C., Ohlsen, K., and Hauck, C.R., Integrinmediated uptake of fibronectin-binding bacteria, Eur. J. Cell Biol., 2011, vol. 90, pp. 891–896.
Khaitlina, S.Yu., Collins, J.H., Kuznetsova, I.M, Pershina, V.P., Synakevich, I.G., Turoverov, K.K., and Usmanova, A.M., Physicochemical properties of actin cleaved with bacterial protease from E. coli A2 strain, FEBS Lett., 1991, vol. 279, pp. 49–51.
Mattock, E. and Blocker, A.J., How do the virulence factors of Shigella work together to cause disease?, Front. Cell Infect. Microbiol., 2017, vol. 64. eCollection 2017. doi 10.1186/1471-2407-5-75
Parasassi, T., Brunelli, R., Bracci-Laudiero, L., Greco, G., Gustafsson, A.C., Krasnowska, E.K., Lundeberg, J., Lundeberg, T., Pittaluga, E., Romano, M.C., and Serafino, A., Differentiation of normal and cancer cells induced by sulfhydryl reduction: biochemical and molecular mechanisms, Cell Death Differ., 2005, vol. 12, pp. 1285–1296.
Parasassi, T., Brunelli, R., Costa, G., De Spirito, M., Krasnowska, E., Lundeberg, T., Pittaluga, E., and Ursini, F., Thiol redox transitions in cell signaling: a lesson from Nacetylcysteine, Scientific World J., 2010, vol. 10, pp. 1192–1202. doi 10.1100/tsw.2010.104
Pizarro-Cerda, J. and Cossart, P., Bacterial adhesion and entry into host cells, Cell, 2006, vol. 124, pp. 715–727.
Pizarro-Cerda, J., Kuhbacher, A., and Cossart, P., Entry of Listeria monocytogenes in mammalian epithelial cells: an updated view, Cold Spring Harb. Perspect. Med., 2012, vol. 2, p. a010009.
Prouty, A.M. and Gunn, J.S., Salmonella enterica serovar typhimurium invasion is repressed in the presence of bile, Infect. Immun., 2000, vol. 68, pp. 6763–6769.
Rubin, H., Multistage carcinogenesis in cell culture, Dev. Biol., 2001, vol. 106, pp. 61–66.
Rubin, H., Chow, M., and Yao, A., Cellular aging, destabilization, and cancer, Proc. Natl. Acad. Sci. U. S. A., 1996, vol. 93, pp. 1825–1830.
Sansonetti, P., Phagocytosis of bacterial pathogens: implications in the host response, Semin. Immunol., 2001, vol. 6, pp. 381–390.
Schwarz-Linek, U., Höök, M., Potts, J.R., The molecular basis of fibronectin-mediated bacterial adherence to host cells, Mol. Microbiol., 2004, vol. 52, pp. 631–641. doi 10.1111/j.1365-2958.2004.04027.x
Singh, B., Fleury, C., Jalalvand, F., and Riesbeck, K., Human pathogens utilize host extracellular matrix proteins laminin and collagen for adhesion and invasion of the host, FEMS Microbiol. Rev., 2012, vol. 36, pp. 1122–1180.
Todaro, G.J., Green, H., and Goldberg, B.D., Transformation of Properties of an established cell line by SV40 and polyoma virus, Proc. Natl. Acad. Sci. U. S. A., 1964, vol. 51, pp. 66–73.
Tran Van Nhieu, G., Enninga, J., Sansonetti, P., and Grompone, G., Tyrosine kinase signaling and type III effectors orchestrating Shigella invasion, Curr. Opin. Microbiol., 2005, vol. 8, pp. 16–22.
Valencia-Gallardo, C.M., Carayol, N., and Tran Van Nhieu, G., Cytoskeletal mechanics during Shigella invasion and dissemination in epithelial cells, Cell Microbiol., 2015, vol. 17, pp. 174–182.
Van Wijk, X.M., Dohrmann, S., Hallstrom, B.M., Li, S., Voldborg, B.G., Meng, B.X., McKee, K.K., van Kuppevelt, T.H., Yurchenco, P.D., Palsson, B.O., Lewis, N.E., Nizet, V., and Eskoa, J.D., Whole-genome sequencing of invasion-resistant cells identifies laminin-2 as a host factor for bacterial invasion, mBio, 2017, vol. 1, p. e02128–16.
Velge, P., Bottreau, E., Kaeffer, B., and Pardon, P., Cell immortalization enhances Listeria monocytogenes invasion, Med. Microbiol. Immunol., 1994, vol. 183, pp. 145–158.
Velge, P., Kaeffer, B., Bottreau, E., and Van Langendonck, N., The loss of contact inhibition and anchorage-dependent growth are key steps in the acquisition of Listeria monocytogenes susceptibility phenotype by non-phagocytic cells, Biol. Cell., 1995, vol. 85, pp. 55–66.
Velge, P., Bottreau, E., Van-Langendonck, N., and Kaeffer, B., Cell proliferation enhances entry of Listeria monocytogenes into intestinal epithelial cells by two proliferationdependent entry pathways, J. Med. Microbiol., 1997, vol. 46, pp. 681–692.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ivlev, A.P., Efremova, T.N., Khaitlina, S.Y. et al. Difference in Susceptibility of 3T3 and 3T3-SV40 Cells to Invasion by Opportunistic Pathogens Serratia grimesii. Cell Tiss. Biol. 12, 33–40 (2018). https://doi.org/10.1134/S1990519X1801008X
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1990519X1801008X