Abstract
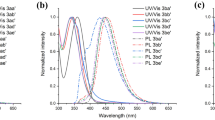
Four new alkyl-substituted 3,3′-bis(dipyrrolylmethene) dihydrobromides containing from 4 to 10 alkyl substituents were synthesized. In a highly alkylated ligand of these substances one of the hydrogen atoms of the 3,3′-methylene spacer was substituted with a phenyl group. The compounds obtained were studied by IR, 1H NMR, electron absorption, and fluorescent spectroscopy. The increased alkylation degree of pyrroles and the introduction of an aryl substituent in the 3,3′-spacer causes a significant high-frequency shift of the N-H stretching vibrations in the IR spectra, an upfield shift of the NH-proton signals in 1H NMR spectra, a decrease in the auxochromic effects of protons on the aromatic system of chromophore in the composition of salts. The red shift of maximum of the strong band in electron absorption spectra and the emission spectra of compounds in DMF, DMSO, C5H5N, C6H6, and CHCl3 was established. The salts obtained are stable in benzene and chloroform, while in electron-donor solvents the irreversible processes of solvolytic dissociation of salts to free organic base and HBr take place.
Similar content being viewed by others
References
Loudet, A. and Burgess, K., Chem Rev., 2007, vol. 107, p. 4891.
Wood, E. and Thompson, A., Chem.Rev., 2007, vol. 107, p. 1831.
Killoram, J., Allen, L., Gallagher, J.F., Gallagher, W.M., and O’Chea, D.F., Chem. Commun., 2002, p. 1862.
Berezin, M.B., Semeikin, A.S., Pashanova, N.A., Antina, E.V., Bukushina, G.B., and Lebedeva N.Sh., Zh. Obshch. Khim., 1999, vol. 69, no. 12, p. 2040.
Sisk, W.N., Ono, N., Yano, T., and Wada, M., Dyes and Pigments, 2002, vol. 55, p. 143.
Filatov, M.A., Cheprakov, A.V., and Vinogradov, S.A., Abstracts of Papers, XXXVIII Int. Conf. on Coordination Chemistry, 20–25 July 2008, Jerusalem, Israel, p. 145.
Tu, B., Ghosh, B., and Lightner, D.A., Monatschefte fur Chemie, 2004, vol. 135, p. 519.
Szacilowski, K., Macyk, W., Drzewiecka-Matuszek, A., Brindell, M., and Stochel, G., Chem. Rev., 2005, vol. 105, p. 2647.
Guseva, G.B., Dudina, N.A., Antina, E.V., V’yugin, A.I., and Semeikin, A.S., Zh. Obshch. Khim., 2008, vol. 78, no. 6, p. 987.
Mamardashvili, N.Zh. and Golubchiko, v O.A., Usp. Khim., 2001, vol. 70, no. 7, p. 656.
Zakharova, S.P., Rumyantsev, E.V., and Antina, E.V., Zh. Obshch. Khim., 2006, vol. 76, no. 7, p. 1244.
Rumyantsev, E.V., Antina, E.V., and Berezin, M.B., Zh. Fiz. Khim., 2006, vol. 80, no. 7, p. 1244.
Mula, S., Ray, A.K., Banerjee, M., Chaudhuri, T., Dasgupta, K., and Chattopadhyaya, S., J. Org. Chem., 2008, vol. 73, p. 2146.
Lavis, L.D. and Raines, R.T., ACS Chem. Biol., 2008, vol. 3, no. 3, p. 142.
Sessler, J.L., Maeda, H., Mizuno, T., Lynch, V.M., and Furuta, H., Chem. Commun., 2002, p. 862.
Cha, N.R., Moon, S.Y., and Chang, S., Tetrahedron Lett., 2003, vol. 44, p. 8265.
Ganzha, V.A., Gurinovich, G.P., Jagarov, B.M., Shul’ga, A.M., and Nizamov, A.N., Zh. Prikl. Spectroskopii, 1987, vol. 47, no. 1, p. 84.
Gordon, A.J. and Ford, R.A., The Chemist’s Companion. A Handbook of Practical Data, Techniques and References, New York: Wiley, 1972.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © E.V. Antina, G.B. Guseva, N.A. Dudina, A.I. V’yugin, A.S. Semeikin, 2009, published in Zhurnal Obshchei Khimii, 2009, Vol. 79, No. 11, pp. 1903–1912.
Rights and permissions
About this article
Cite this article
Antina, E.V., Guseva, G.B., Dudina, N.A. et al. Synthesis and spectral analysis of alkyl-substituted 3,3′-bis(dipyrrolylmethenes). Russ J Gen Chem 79, 2425–2434 (2009). https://doi.org/10.1134/S1070363209110243
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070363209110243