Abstract
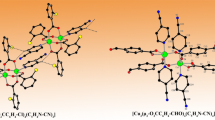
The cation-anionic coordination compounds based on the copper(II) complexes with Phen and tartratogermanate anions, [Cu(Phen)3]2[Ge2(OH)(HTart)(µ-Tart)2] · 11H2O (I) and [CuCl(Phen)2]4[{Ge2(OH)2(µ-Tart)2}Cl2] · 4Н2О (II), with different compositions and structures (CIF files CCDC nos. 1878102 (І) and 1878103 (ІІ)) are isolated for the first time from the GeO2–tartaric acid (H4Tart)–CuCl2–1,10-phenanthroline (Phen) system. The structures of compounds I and II contain the same bridging tartrate anions. A specific feature of compound I is the additional HTart3– anion, which does not perform the bridging function but is coordinated via the bis(chelate) mode, and the hydroxyl and carboxyl groups remain vacant. The dissociation of the carboxyl group results in the formation of the tartratogermanate anion with the charge –4.



Similar content being viewed by others
REFERENCES
Ambrosov, I.V., Aleshin, S.V., Alimbarova, L.M., et al., Razrabotka i registratsiya lekarstvennykh sredstv (Development and Registration of Pharmaceutical Products), 2015, no. 2, p. 144.
Seifullina, I.I. and Martsinko, E.E., Koordinatsionnye soedineniya germaniya(IV) s anionami limonnoi, vinnoi i ksilarovoi kislot (Germanium(IV) Coordination Compounds with Citrate, Tartrate, and Xylarate Anions), Odessa: ONU, 2015.
Seifullina, I.I., Ilyukhin, A.B., Martsinko, E.E., et al., Russ. J. Inorg. Chem., 2014, vol. 59, p. 298. https://doi.org/10.1134/S0036023614040172
Seifullina, I.I., Martsinko, E.E., Chebanenko, E.A., et al., Chem. J. Moldova, 2017, vol. 12, no. 2, p. 52.
Huan-Yu, L., Fen-Ying, W., Guo-Yong, W., et al., Acta Crystallogr., Sect. E: Struct. Rep. Online, 2006, vol. 62, p. m111.
Yang-Yang, H., Li-Na, X., Yan, W., et al., Polyhedron, 2013, vol. 56, p. 152.
Vreshch, O.V., Nesterova, O.V., Kokozay, V.N., et al., Z. Anorg. Allg. Chem., 2009, vol. 635, p. 2316.
Rusanova, J.A., Kozachuka, O.V., and Dyakonenko, V.V., Acta Crystallogr., Sect. E: Struct. Rep. Online, 2013, vol. 69, p. m391.
Polyanskaya, T.M., Drozdova, M.K., and Volkov, V.V., J. Struct. Chem., 2010, vol. 51, no. 6, p. 1139.
Seifullina, I.I., Martsinko, E.E., and Afanasenko, E.V., Visn. Odes. Nac. Univ. Him., 2015, vol. 20, no. 4, p. 6.
Hamdani, H.E., Amane, M.E., Atmani, Z., and Haddad, M., J. Mater. Environ. Sci., 2016, vol. 7, no. 9, p. 3100.
Menchikov, L.G. and Ignatenko, M.A., Khim.-Farm. Zh., 2012, vol. 46, no. 11, p. 3.
Klyuchnikov, N.G., Rukovodstvo po neorganicheskomu sintezu (Guidelines on Inorganic Synthesis), Moscow: Khimiya, 1965.
Sheldrick, G.M., Acta Crystallogr., Sect. A: Found. Adv., 2015, vol. 71, p. 3.
Sheldrick, G.M., Acta Crystallogr., Sect. C: Struct. Chem., 2015, vol. 71, p. 3.
Addison, A.W., Rao, N., Reedijk, J., et al., Dalton Trans., 1984, no. 7, p. 1349.
Martsinko, E.E., Seifulina, I.I., Chebanenko, E.A., et al., Vestnik Odesskogo Nats. Univ, Khimiya, 2018, vol. 23, no. 1, p. 6.
Minacheva, L.K., Seifullina, I.I., Ilyukhin, A.B., et al., Russ. J. Coord. Chem., 2013, vol. 39, no. 11, p. 751. https://doi.org/10.1134/S1070328413110043
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated by E. Yablonskaya
Rights and permissions
About this article
Cite this article
Seifullina, I.I., Martsinko, E.E., Chebanenko, E.A. et al. Complex Formation Products in the GeO2–Tartaric Acid–CuCl2–1,10-Phenanthroline System: Syntheses and Structures. Russ J Coord Chem 45, 496–504 (2019). https://doi.org/10.1134/S107032841906006X
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S107032841906006X