Abstract
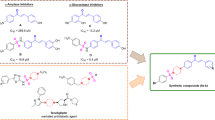
Design and synthesis of two series of trifluoromethyl-containing polysubstituted pyrimidine derivatives in order to seek more effective anti-tumor drugs, and the antiproliferative activity of these compounds against four human tumor cell lines (PC-3, MGC-803, MCF-7, HGC-27) was evaluated by MTT method. Most of the compounds have moderate anti-proliferative activities, among which compounds (XXId) and (XXIe) have good selectivity for PC-3 cells. The IC50 (concentration required to achieve 50% inhibition of the tumor cell proliferation) values of compounds (XXId) and (XXIe) for PC-3 cell were 4.42 ± 0.46 μmol L–1 and 4.85 ± 0.59 μmol L–1, respectively, and their anti-proliferative activities were significantly better than that of the positive control 5-fluorouracil (6.39 ± 0.71 μmol L–1). Molecular docking results indicated that compounds (XXId) and (XXIe) were likely to be well integrated in the hydrophobic pocket of ubiquitin-specific protease 7 (USP7), which has the value of further research and development.


Similar content being viewed by others
REFERENCES
Aberoumandi, S.M., Mohammadhosseini, M., Abasi, E., Saghati, S., Nikzamir, N., Akbarzadeh, A., Panahi, Y., and Davaran, S., Artif. Cells Nanomed. Biotechnol., 2017, vol. 45, pp. 1–11. https://doi.org/10.1080/21691401.2016.1228658
Torre, L.A., Siegel, R.L., Ward, E.M., and Jemal, A., Cancer Epidemiol. Biomarkers Prev., 2016, vol. 25, pp. 16–27. https://doi.org/10.1158/1055-9965.Epi-15-0578
Poggio, F., Bruzzone, M., Ceppi, M., Pondé, N.F., La Valle, G., Del Mastro, L., de Azambuja, E., and Lambertini, M., Ann Oncol., 2018, vol. 29, pp. 1497–1508. https://doi.org/10.1093/annonc/mdy127
Gosavi, R., Chia, C., Michael, M., Heriot, A.G., Warrier, S.K., and Kong, J.C., Int. J. Colorectal Dis., 2021, vol. 36, pp. 2063–2070. https://doi.org/10.1007/s00384-021-03945-3
Norouzi, M., Drug. Dis. Today, 2018, vol. 23, pp. 912–919. https://doi.org/10.1016/j.drudis.2018.02.007
Passaro, A., Guerini-Rocco, E., Pochesci, A., Vacirca, D., Spitaleri, G., Catania, C.M., Rappa, A., Barberis, M., and de Marinis, F., Pharmacol. Res., 2017, vol. 117, pp. 406–415. https://doi.org/10.1016/j.phrs.2017.01.003
Zeng, F., Wang, F., Zheng, Z., Chen, Z., Wah To, K.K., Zhang, H., Han, Q., and Fu, L., Acta Pharm. Sin. B, 2020, vol. 10, pp. 799–811. https://doi.org/10.1016/j.apsb.2020.01.008
Yang, J.C., Reckamp, K.L., Kim, Y.C., Novello, S., Smit, E.F., Lee, J.S., Su, W.C., Akerley, W.L., Blakely, C.M., Groen, H.J.M., Bazhenova, L., Carcereny Costa, E., Chiari, R., Hsia, T.C., Golsorkhi, T., Despain, D., Shih, D., Popat, S., and Wakelee, H., JTO Clin. Res. Rep., 2021, vol. 2, p. 100114. https://doi.org/10.1016/j.jtocrr.2020.100114
Finlay, M.R., Anderton, M., Ashton, S., Ballard, P., Bethel, P.A., Box, M.R., Bradbury, R.H., Brown, S.J., Butterworth, S., Campbell, A., Chorley, C., Colclough, N., Cross, D.A., Currie, G.S., Grist, M., Hassall, L., Hill, G.B., James, D., James, M., Kemmitt, P., Klinowska, T., Lamont, G., Lamont, S.G., Martin, N., McFarland, H.L., Mellor, M.J., Orme, J.P., Perkins, D., Perkins, P., Richmond, G., Smith, P., Ward, R.A., Waring, M.J., Whittaker, D., Wells, S., and Wrigley, G.L., J. Med. Chem., 2014, vol. 57, pp. 8249–8267. https://doi.org/10.1021/jm500973a
Cross, D.A., Ashton, S.E., Ghiorghiu, S., Eberlein, C., Nebhan, C.A., Spitzler, P.J., Orme, J.P., Finlay, M.R., Ward, R.A., Mellor, M.J., Hughes, G., Rahi, A., Jacobs, V.N., Red Brewer, M., Ichihara, E., Sun, J., Jin, H., Ballard, P., Al-Kadhimi, K., Rowlinson, R., Klinowska, T., Richmond, G.H., Cantarini, M., Kim, D.W., Ranson, M.R., and Pao, W., Cancer Dis., 2014, vol. 4, pp. 1046–1061. https://doi.org/10.1158/2159-8290.Cd-14-0337
Jänne, P.A., Yang, J.C., Kim, D.W., Planchard, D., Ohe, Y., Ramalingam, S.S., Ahn, M.J., Kim, S.W., Su, W.C., Horn, L., Haggstrom, D., Felip, E., Kim, J.H., Frewer, P., Cantarini, M., Brown, K.H., Dickinson, P.A., Ghiorghiu, S., and Ranson, M., New Engl. J. Med., 2015, vol. 372, pp. 1689–1699. https://doi.org/10.1056/NEJMoa1411817
Cao, Y., Yang, Y., Ampomah-Wireko, M., Obaid Arhema Frejat, F., Zhai, H., Zhang, S., Wang, H., Yang, P., Yuan, Q., Wu, G., and Wu, C., Bioorg. Med. Chem. Lett., 2022, vol. 64, p. 128654. https://doi.org/10.1016/j.bmcl.2022.128654
Mareddy, J., Suresh, N., Kumar, C.G., Kapavarapu, R., Jayasree, A., and Pal, S., Bioorg. Med. Chem. Lett., 2017, vol. 27, pp. 518–523. https://doi.org/10.1016/j.bmcl.2016.12.030
Jia, Y., Si, L., Lin, R., Jin, H., Jian, W., Yu, Q., and Yang, S., Eur. J. Med. Chem., 2019, vol. 172, pp. 62–70. https://doi.org/10.1016/j.ejmech.2019.03.033
Meanwell, N.A., J. Med. Chem., 2018, vol. 61, pp. 5822–5880. https://doi.org/10.1021/acs.jmedchem.7b01788
Clegg, N.J., Wongvipat, J., Joseph, J.D., Tran, C., Ouk, S., Dilhas, A., Chen, Y., Grillot, K., Bischoff, E.D., Cai, L., Aparicio, A., Dorow, S., Arora, V., Shao, G., Qian, J., Zhao, H., Yang, G., Cao, C., Sensintaffar, J., Wasielewska, T., Herbert, M.R., Bonnefous, C., Darimont, B., Scher, H.I., Smith-Jones, P., Klang, M., Smith, N.D., De Stanchina, E., Wu, N., Ouerfelli, O., Rix, P.J., Heyman, R.A., Jung, M.E., Sawyers, C.L., and Hager, J.H., Cancer Res., 2012, vol. 72, pp. 1494–1503. https://doi.org/10.1158/0008-5472.CAN-11-3948
Stein, E.M., DiNardo, C.D., Fathi, A.T., Pollyea, D.A., Stone, R.M., Altman, J.K., Roboz, G.J., Patel, M.R., Collins, R., Flinn, I.W., Sekeres, M.A., Stein, A.S., Kantarjian, H.M., Levine, R.L., Vyas, P., MacBeth, K.J., Tosolini, A., VanOostendorp, J., Xu, Q., Gupta, I., Lila, T., Risueno, A., Yen, K.E., Wu, B., Attar, E.C., Tallman, M.S., and de Botton, S., Blood, 2019, vol. 133, pp. 676–687. https://doi.org/10.1182/blood-2018-08-869008
Chauhan, D., Tian, Z., Nicholson, B., Kumar, K.G., Zhou, B., Carrasco, R., McDermott, J.L., Leach, C.A., Fulcinniti, M., Kodrasov, M.P., Weinstock, J., Kingsbury, W.D., Hideshima, T., Shah, P.K., Minvielle, S., Altun, M., Kessler, B.M., Orlowski, R., Richardson, P., Munshi, N., and Anderson, K.C., Cancer Cell, 2012, vol. 22, pp. 345–358. https://doi.org/10.1016/j.ccr.2012.08.007
An, T., Gong, Y., Li, X., Kong, L., Ma, P., Gong, L., Zhu, H., Yu, C., Liu, J., Zhou, H., Mao, B., and Li, Y., Biochem. Pharmacol., 2017, vol. 131, pp. 29–39. https://doi.org/10.1016/j.bcp.2017.02.011
Wang, M., Zhang, Y., Wang, T., Zhang, J., Zhou, Z., Sun, Y., Wang, S., Shi, Y., Luan, X., Zhang, Y., Wang, Y., Wang, Y., Zou, Z., Kang, L., and Liu, H., Cell Physiol. Biochem., 2017, vol. 43, pp. 1755–1766. https://doi.org/10.1159/000484062
Tasneem, S., Sheikh, K.A., Naematullah, M., Mumtaz Alam, M., Khan, F., Garg, M., Amir, M., Akhter, M., Amin, S., Haque, A., and Shaquiquzzaman, M., Bioorg. Chem., 2022, vol. 126, p. 105885. https://doi.org/10.1016/j.bioorg.2022.105885
Maes, T., Mascaró, C., Rotllant, D., Lufino, M.M.P., Estiarte, A., Guibourt, N., Cavalcanti, F., Griñan-Ferré, C., Pallàs, M., Nadal, R., Armario, A., Ferrer, I., Ortega, A., Valls, N., Fyfe, M., Martinell, M., Castro Palomino, J.C., and Buesa Arjol, C., PLoS One, 2020, vol. 15, p. e0233468. https://doi.org/10.1371/journal.pone.0233468
Abdel-Mohsen, H.T., Conrad, J., Harms, K., Nohr, D., and Beifuss, U., RSC Adv., 2017, vol. 7, pp. 17427–17441. https://doi.org/10.1039/C6RA28102H
Ma, L.Y., Zheng, Y.C., Wang, S.Q., Wang, B., Wang, Z.R., Pang, L.P., Zhang, M., Wang, J.W., Ding, L., Li, J., Wang, C., Hu, B., Liu, Y., Zhang, X.D., Wang, J.J., Wang, Z.J., Zhao, W., and Liu, H.M., J. Med. Chem., 2015, vol. 58, pp. 1705–1716. https://doi.org/10.1021/acs.jmedchem.5b00037
Ciocoiu, C.C., Nikolic, N., Nguyen, H.H., Thoresen, G.H., Aasen, A.J., and Hansen, T.V., Eur. J. Med. Chem., 2010, vol. 45, pp. 3047–3055. https://doi.org/10.1016/j.ejmech.2010.03.035
Singh, M.S., Chowdhury, S., and Koley, S., Tetrahedron, 2016, vol. 72, pp. 5257–5283. https://doi.org/10.1016/j.tet.2016.07.044
Funding
This work was supported by the National Natural Sciences Foundation of China (no. 82020108030).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
The authors declare that they have no conflicts of interest.
The work has no studies involving humans or animals as subjects of the study.
Supplementary Information
Rights and permissions
About this article
Cite this article
Lingling, C., Hao, W., Fuqiang, Y. et al. Design, Synthesis and Antitumor Activity Evaluation of Trifluoromethyl-Containing Polysubstituted Pyrimidine Derivatives. Russ J Bioorg Chem 49, 86–100 (2023). https://doi.org/10.1134/S1068162023010168
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1068162023010168