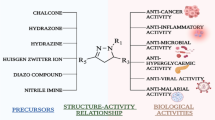
Abstract
In this study, a series of novel pirfenidone derivatives were designed and synthesized, and their activities against pulmonary fibrosis were evaluated. The structures of all the derivatives were characterized by 1H and 13C nuclear magnetic resonance and high-resolution mass spectrometry spectra. A preliminary investigation of their bioactivity demonstrated that all the target compounds exhibited different degrees of activity against pulmonary fibrosis and that the activities of most derivatives were markedly superior to that of pirfenidone.




Similar content being viewed by others
REFERENCES
Raghu, G., Flaherty K.R., Lederer D.J., Lynch D.A., Colby T.V., Myers J.L., Groshong S.D., Larsen B.T., Chung J.H., Steele M.P., Benzaquen S., Calero K., Case A.H., Criner G.J., Nathan S.D., Rai N.S., Ramaswamy M., Hagmeyer L., Davis J.R., Gauhar U.A., Pankratz D.G., Choi Y., Huang J., Walsh P.S., Neville H., Lofaro L.R., Barth N.M., Kennedy G.C., Brown K.K., Martinez F.J., Lancet Respir. Med., 2019, vol. 7, pp. 487–496. https://doi.org/10.1016/s2213-2600(19)30059-1
Yang, S.N., Liu, P.P., Jiang, Y.L., Wang, Z., Dai, H.P., and Wang, C., Front. Cell Dev. Biol., 2021, vol. 9, 639657. https://doi.org/10.3389/fcell.2021.639657
Barratt, S.L., Creamer, A., Hayton, C., and Chaudhuri, N., J. Clin. Med., 2018, vol. 7. https://doi.org/10.3390/jcm7080201
Lynch, D.A., Sverzellati, N., Travis, W.D., Brown, K.K., Colby, T.V., Galvin, J.R., Goldin, J.G., Hansell, D.M., Inoue, Y., Johkoh, T., Nicholson, A.G., Knight, S.L., Raoof, S., Richeldi, L., Ryerson, C.J., Ryu, J.H., and Wells, A.U., Lancet Respir. Med., 2018, vol. 6, pp. 138–153. https://doi.org/10.1016/s2213-2600(17)30433-2
Maher, T.M., Kreuter, M., Lederer, D.J., Brown, K.K., Wuyts, W., Verbruggen, N., Stutvoet, S., Fieuw, A., Ford, P., Abi-Saab, W., and Wijsenbeek, M., BMJ Open Respir. Res., 2019, vol. 6, art. e000422. https://doi.org/10.1136/bmjresp-2019-000422
Raghu, G., S.Y., Yeh, W.S., Maroni, B., Li, Q., Lee, Y.C., and Collard, H.R., The Lancet Respir. Med., 2014, vol. 2, pp. 566–572. https://doi.org/10.1016/s2213-2600(14)70101-8
Yang, J.T., Pan, X., Wang, L., and Yu, G.Y., Mol. Med., 2020, vol. 26, 95. https://doi.org/10.1186/s10020-020-00223-w
Raghu, G., Remy-Jardin, M., Myers, J.L., Richeldi, L., Ryerson, C.J., Lederer, D.J., Behr, J., Cottin, V., Danoff, S.K., Morell, F., Flaherty, K.R., Wells, A., Martinez, F.J., Azuma, A., Bice, T.J., Bouros, D., Brown, K.K., Collard, H.R., Duggal, A., Galvin, L., Inoue, Y., Jenkins, R.G., Johkoh, T., Kazerooni, E.A., Kitaichi, M., Knight, S.L., Mansour, G., Nicholson, A.G., Pipavath, S.N.J., Buendia-Roldan, I., Selman, M., Travis, W.D., Walsh, S., Wilson, K.C., Am. Thorac. Soc., Eur. Respir. Soc., Jpn. Respir. Soc., and Lat. Am. Thorac. Soc., Am. J. Respir. Crit. Care Med., 2018, vol. 198, pp. E44–E68. https://doi.org/10.1164/rccm.201807-1255ST
Pan, Y., Huang, W., Shen, Z., and Ma, Z., Chin. Pharm. J., 2012, vol. 47, pp. 1873–1876.
Ye, Z.M. and Hu Y.B., Int. J. Mol. Med., 2021, vol. 48. https://doi.org/10.3892/ijmm.2021.4965
Richeldi, L., Collard, H.R., and Jones, M.G., Lancet, 2017, vol. 389, pp. 1941–1952. https://doi.org/10.1016/s0140-6736(17)30866-8
Lederer, D.J. and Martinez, F.J., N. Engl. J. Med., 2018, vol. 378, pp. 1811–1823. https://doi.org/10.1056/NEJMra1705751
Shaney, L.B., Andrew, C., Conal, H., and Chaudhuri, N., J. Clin. Med., 2018, vol. 7, p. 21. https://doi.org/10.3390/jcm7080201
Salazar-Montes, A., Ruiz-Corro, L., Lopez-Reyes, A., Castrejon-Gomez, E., and Armendariz-Borunda, J., Eur. J. Pharmacol., 2008, vol. 595, pp. 69–77. https://doi.org/10.1016/j.ejphar.2008.06.110
Flaherty, K.R., Fell, C.D., Huggins, J.T., Nunes, H., Sussman, R., Valenzuela, C., Petzinger, U., Stauffer, J.L., Gilberg, F., Bengus, M., and Wijsenbeek, M., Eur. Respir. J., 2018, vol. 52, 1800230. https://doi.org/10.1183/13993003.00230-2018
Rasooli, R., Pourgholamhosein, F., Kamali, Y., Nabipour, F., and Mandegary, A., Inflammation, 2018, vol. 41, pp. 134–142. https://doi.org/10.1007/s10753-017-0671-9
Hanta, I., Cilli, A., and Sevinc, C., Adv. Ther., 2019, vol. 36, pp. 1126–1131. https://doi.org/10.1007/s12325-019-00928-3
Majewski, S., Bialas, A.J., Buchczyk, M., Gomolka, P., Gorska, K., Jagielska-Len, H., Jarzemska, A., Jassem, E., Jastrzebski, D., Kania, A., Koprowski, M., Krenke, R., Kus, J., Lewandowska, K., Martusewicz-Boros, M.M., Roszkowski-Sliz, K., Siemiriska, A., Sladek, K., Sobiecka, M., Szewczyk, K., Tomczak, M., Tomkowski, W., Wiatr, E., Ziora, D., Zolnowska, B., and Piotrowski, W.J., BMC Pulm. Med., 2020, vol. 20, 122. https://doi.org/10.1186/s12890-020-1162-6
Bernard, K. and Thannickal, V.J., Antioxid. Redox Signal., 2020, vol. 33, pp. 455–479. https://doi.org/10.1089/ars.2020.8032
Zhu, W., Shen, J., Li, Q., Pei, Q., Chen, J., Chen, Z., Liu, Z., and Hu, G., Arch. Pharm. (Weinheim), 2013, vol. 346, pp. 654–666. https://doi.org/10.1002/ardp.201300138
Wu, L., Liu, B., Li, Q., Chen, J., Tao, L., and Hu, G., Molecules, 2012, vol. 17, pp. 1373–1387. https://doi.org/10.3390/molecules17021373
Chen, J., Lu, M.M., and Liu, B., Bioorg. Med. Chem. Lett, 2012, vol. 22, pp. 2300–2302. https://doi.org/10.1016/j.bmcl.2012.01.073
Lou, Q., Meng, X., Lao, Z., Xuan, L., Bai, J., Hou, Q., Hu, G., Luo, R., Tao, L., and Li, Z., Molecules, 2012, vol. 17, pp. 884–896. https://doi.org/10.3390/molecules17010884
Chen, J., Peng, Z., Lu, M., Xiong, X., Chen, Z., Li, Q., Cheng, Z., Jiang, D., Tao, L., and Hu, G., Bioorg. Med. Chem. Lett., 2018, vol. 28, pp. 222–229. https://doi.org/10.1016/j.bmcl.2017.07.001
Chen, Z., Liu, J., Jin, C., Tan, Q., and Ye, M., Tetrahedron Lett., 2019, vol. 60, pp. 1265–1267. https://doi.org/10.1016/j.tetlet.2019.04.001
Chun, S.L. and Darryl, D.D., Tetrahedron Lett., 2004, vol. 45, pp. 4257–4260. https://doi.org/10.1016/j.tetlet.2004.04.019
Sugahara, M. and Ukita, T., Chem. Pharm. Bull., 1997, vol. 45, pp. 719–721. https://doi.org/10.1248/cpb.45.719
Kaur Navjeet, Synth. Commun., 2015, vol. 45, pp. 1269–1300. https://doi.org/10.1080/00397911.2013.827725
Lin, S.N. and Li, H.Y., Tetrahedron Lett., 2005, vol. 46, pp. 4315–4319. https://doi.org/10.1016/j.tetlet.2005.04.101
Hiromichi, F., Kenichi, M., and Yusuke, O., Tetrahedron Lett., 2005, vol. 46, pp. 2197–2199. https://doi.org/10.1016/j.tetlet.2005.02.025
Funding
This study was supported by the Open Fund of Key Laboratory of Biotechnology and Bioresources Utilization (Dalian Minzu University), Ministry of Education (no. KF2021005), China.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
COMPLIANCE WITH ETHICAL STANDARDS
This article does not contain any studies involving human participants performed by any of the authors and does not contain any studies involving animals performed by any of the author.
Conflict of Interests
The authors declare that they have no conflicts of interest.
Additional information
Corresponding autor: e-mail: yanghj@swun.edu.cn.
Supplementary Information
Rights and permissions
About this article
Cite this article
Gengga Sangmao, Li, Y., Wang, W. et al. Design, Synthesis, and Biological Evaluation of a Novel Series of Pirfenidone Derivatives. Russ J Bioorg Chem 48, 430–437 (2022). https://doi.org/10.1134/S1068162022020091
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1068162022020091