Abstract
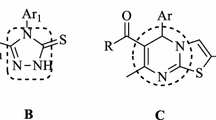
A series of aralkanoic acids was converted into aralkanoic acid hydrazides through their esters formation. The aralkanoic acid hydrazides upon treatment with carbon disulfide and methanolic potassium hydroxide yielded potassium dithiocarbazinate salts, which on refluxing with aqueous hydrazine hydrate yielded 5-aralkyl-4-amino-3-mercapto-1,2,4-triazoles. The target compounds, 3-aralkyl-6-(substitutedquinolinyl) [1,2,4]triazolo[3,4-b][1,3,4]thiadiazoles, were synthesized by condensing various quinolinyl substituted carboxylic acids with 5-aralkyl-4-amino-3-mercapto-1,2,4-triazoles in phosphorus oxychloride. The structures of the newly synthesized triazolothiadiazoles were characterized by IR, 1H NMR, 13C NMR, and elemental analysis studies. The structure of one of the 5-aralkyl-4-amino-3-mercapto-1,2,4-triazoles was unambiguously deduced by single crystal X-ray diffraction analysis. All the synthesized compounds were screened for their acetylcholinesterase inhibition activities. Four of the triazolothiadiazoles exhibited excellent acetylcholinesterase inhibition activities as compared to the reference inhibitor.
Similar content being viewed by others
References
Quinn, D.M., Chem. Rev., 1987, vol. 87, pp. 955–979.
Colletier, J.P., Fournier, D., Greenblatt, H.M., Stogan, J., Sussman, J.L., Zaccai, G., Silman, I., and Weik, M., EMBO J., 2006, vol. 25, pp. 2746–2756.
Mallender, W.D., Szeglets, T., and Rosenberry, T.L., J. Biol. Chem., 1999, vol. 274, pp. 8491–8499.
Szeglets, T., Mallender, W.D., Thomas, P.J., and Rosenberry, T.L., Biochemistry, 1999, vol. 38, pp. 122–133.
Abramov, A.Y., Canvari, L., and Duchen, M.R., J. Neurosci., 2003, vol. 23, pp. 5088–5095.
Butterfield, D.A., Chem. Res. Toxicol., 1997, vol. 10, pp. 495–506.
Greenblatt, H.M., Dvir, H., Silman, I., and Sussman, J.L., J. Mol. Neurosci., 2003, vol. 20, pp. 369–383.
Ellis, J.M., J. Am. Osteopth. Assoc., 2005, vol. 105, pp. 145–158.
Farlow, M., Gracon, S.I., Hershey, L.A., Lewis, K.W., Sadowsky, C.H., and Ureno, J.D., J. Am. Med. Assoc., 1992, vol. 268, pp. 2523–2529.
Lahiri, D.K., Farlow, M.R., Grieg, N.H., and Sambamurti, K., Drug. Dev. Res., 2002, vol. 56, pp. 267–281.
Rogers, S.L., Farlow, M.R., Doody, R.S., Mohs, R., and Friedhoff, L.T., Neurology, 1998, vol. 50, pp. 136–145.
Eugene, L., Piatnitski, C., Hassan, M.E., and John, B., Tetrahedron. Lett., 2008, vol. 49, pp. 6709–6711.
Ellman, G.L., Courtney, K.D., Andres, V., and Featherstone, R.M., Biochem. Pharmacol., 1961, vol. 7, pp. 88–90.
Author information
Authors and Affiliations
Corresponding author
Additional information
The article is published in the original.
Rights and permissions
About this article
Cite this article
Rafiq, M., Saleem, M., Hanif, M. et al. Acetylcholinesterase inhibition activity of some quinolinyl substituted triazolothiadiazole derivatives. Russ J Bioorg Chem 41, 170–177 (2015). https://doi.org/10.1134/S1068162015020089
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1068162015020089