Abstract
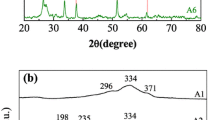
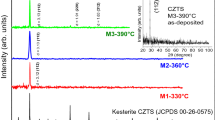
This paper concentrates on the electrodeposition of CZTS kesterite thin films on ITO coated glass. The main objective of the article is to compare the electrodeposition from an ionic liquid electrolyte, which is choline–urea, and from an aqueous electrolyte, that is the citrate buffer solution. The electrochemical behaviour of the citrate buffer shown a remarkable evolution of the hydrogen, which was less in the choline chloride–urea electrolyte. The XRD pattern of the films provided from the two electrolytes confirms the presence of the kesterite form of Cu2ZnSnS4. The surface morphology of the samples was examined using scanning electron microscopy and showed that electrodeposition from the citrate buffer electrolyte leads to have CZTS thin films with less porous morphology.




Similar content being viewed by others
REFERENCES
Chan, C.P., Lam, H., and Surya, C., Preparation of Cu2ZnSnS4 films by electrodeposition using ionic liquids, Solar Energy Mater. Solar Cells, 2010, vol. 94, no. 2, pp. 207–211.
Araki, H., Yuki Kubo, Kazuo Jimbo, Win Shwe Maw, Hironori Katagiri, Makoto Yamazaki, Koichiro Oishi, and Akiko Takeuchi, Preparation of Cu2ZnSnS4 thin films by sulfurizing electroplated precursors, Solar Energy Mater. Solar Cells, 2009, vol. 93, no. 6–7, pp. 996–999.
Shin, S.W., Hun, J.H., Park, C.Y., Mojolkar, A.V., Lee, J.Y., and Kim, J.H., Quaternary Cu2ZnSnS4 nanocrystals: facile and low cost synthesis by microwave-assisted solution method, J. Alloys Compd., 2012, vol. 516, pp. 96–101.
Ge, J., Wu, Y., Zhang, C., Zuo, S., Jiang, J., Ma, J., Yang, P., and Chu, J., Comparative study of the influence of two distinct sulfurization ramping rates on the properties of Cu2ZnSnS4 thin films, Appl. Surface Sci., 2012, vol. 258, no. 19, pp. 7250–7254.
Jae-Seung, S., Sang-Yul, L., Jae-Choon, L., Hyo-Duk, N., and Kyoo-Ho, K., Electrical and optical properties of Cu2ZnSnS4 thin films prepared by rf magnetron sputtering process, Solar Energy Mater. Solar Cells, 2003, vol. 75, pp. 155–162.
Shin, S.W., Pawar, S.M., Park, C.Y., Yun, J.H., Moon, J.H., Kim, J.H., and Lee, J.Y., Studies on Cu2ZnSnS4 (CZTS) absorber layer using different stacking orders in precursor thin films, Solar Energy Mater. Solar Cells, 2011, vol. 95, no. 12, pp. 3202–3206.
Dergacheva, M.B., Urazov, K.A., and Nurtazina, A.E., Electrodeposition of thin Cu2ZnSnS4 films, Russ. J. Electrochem., 2017, vol. 53, pp. 324–332. https://doi.org/10.1134/S102319351703003X
Oishi, K., Saito, G., Ebina, K., Nagahashi, M., Jimbo, K., Maw, W.S., Katagiri, H., Yamazaki, M., Araki, H., and Takeuchi, A., Growth of Cu2ZnSnS4 thin films on Si (100) substrates by multisource evaporation, Thin Solid Films, 2008, vol. 517, no. 4, pp. 1449–1452.
Farinella, M., Inguanta, R., Spanò, T., Livreri, P., Piazza, S., and Sunseri, C., Electrochemical deposition of CZTS thin films on flexible substrate, Energy Proc., 2014, vol. 44, pp. 105–110.
Farinella, M., Livreri, P., Piazza, S., Sunseri, C., and Inguanta, R., One-step electrodeposition of CZTS for solar cell absorber layer, in Electroplating of Nanostructures, Aliofkhazraei, M., Ed., InTech, 2015.
Chen, H., Ye, X., He, X., Ding, J., Zhang, Y., Han, J., Liu, J., Liao, C., Mei, J., and Lau, W., Electrodeposited CZTS solar cells from Reline electrolyte, Green Chem., 2014, vol. 16, no. 8, p. 3841.
Gurav, K.V., Shin, S.W., Patil, U.M., Suryawanshi, M.P., Pawar, S.M., Gang, M.G., Vanalakar, S.A., Yun, J.H., and Kim, J.H., Improvement in the properties of CZTSSe thin films by selenizing single-step electrodeposited CZTS thin films, J. Alloys Compd., 2015, vol. 631, pp. 178–182.
Pawar, S.M., Pawar, B.S., Moholkar, A.V., Choi, D.S., Yun, J.H., Moon, J.H., Kolekar, S.S., and Kim, J.H., Single step electrosynthesis of Cu2ZnSnS4 (CZTS) thin films for solar cell application, Electrochim. Acta, 2010, vol. 55, no. 12, pp. 4057–4061.
Azmi, S., Pezzato, L., Sturaro, M., Khoumri, E.M., Martucci, A., and Dabalà, M., A green and low-cost synthetic approach based on deep eutectic choline-urea solvent toward synthesis of CZTS thin films, Ionics, vol. 24, 2018, pp. 1–7. https://doi.org/10.1007/s11581-018-2719-8
Ateya, B.G., AlKharafi, F.M., and Al-Azab, A.S., Electrodeposition of sulfur from sulfide contaminated brines, Electrochem. Solid-State Lett., 2003, vol. 6, no. 9, p. C137.
Azmi, S., Nohair, M., Marrakchi, M., Khoumri, E.M., and Dabala, M., Effect of the complexing agents on the properties of electrodeposited CZTS thin films, Proc. 7th Int. Conf. on Renewable Energy Research and Applications (ICRERA), San Diego, CA, 2018, pp. 1346–1351. https://doi.org/10.1109/ICRERA.2018.8566894
Guo, M., Zhu, X., and Li, H., Comparative study of Cu2ZnSnS4 thin film solar cells fabricated by direct current and pulse reverse co-electrodeposition, J. Alloys Compd., 2016, vol. 657, pp. 336–340.
Tlemçani, T.S., Benamar, E.B., Cherkaoui, F., Hajji, F., Edfouf, Z., Taibi, M., Labrim, H., Belhorma, B., Aazou, S., Schmerber, G., Bouras, K., Sekkat, Z., Dinia, A., Ulyashin, A., Slaoui, A., and Abd-lefdil, M., Deposition time effect on the physical properties of Cu2ZnSnS4 (CZTS) thin films obtained by electrodeposition route onto Mo-coated glass substrates, Energy Proc., 2015, vol. 84, pp. 127–133.
Author information
Authors and Affiliations
Corresponding author
Additional information
The article is published in the original.
Rights and permissions
About this article
Cite this article
Azmi, S., Khoumri, E.M., Pezzato, L. et al. Influence of Solvent on the Proprieties of Cu2ZnSnS4 Thin Film Fabricated by One-Step Electrodeposition. Russ J Electrochem 55, 796–801 (2019). https://doi.org/10.1134/S1023193519070036
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1023193519070036