Abstract
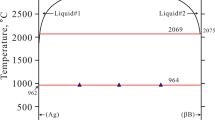
CALPHAD modelling of the Ag–Pd–Sn ternary system has been performed. The disordered phases, the melt and the fcc phase were described using the substitutional solution model. Sublattice models were used to describe intermetallic compounds and the ternary phase. The two-sublattice model (Ag,Pd)4(Ag, Sn) used for the ternary phase made it possible to reproduce the inclination of its homogeneity range. The results of the thermodynamic calculation of the Ag–Pd–Sn system are in good agreement with the experimental data on phase equilibria and enthalpies of formation of the liquid. The agreement with the data on the partial Gibbs energy of tin in the liquid is somewhat worse.




Similar content being viewed by others
REFERENCES
H.-J. Shin, Y. H. Kwon, and H.-J. Seol, J. Mech. Behav. Biomed. Mater. 107, 103728 (2020). https://doi.org/10.1016/j.jmbbm.2020.103728
R. Zhang, M. Peng, L. Ling, et al., Chem. Eng. Sci. 199, 64 (2019). https://doi.org/10.1016/j.ces.2019.01.018
R. Zerdoumi and M. Armbrüster, ACS Appl. Energy Mater. 4, 11279 (2021). https://doi.org/10.1021/acsaem.1c02119
C. Y. Lee, S. P. Yang, C. H. Yang, et al., Surf. Coat. Technol. 395, 125879 (2020). https://doi.org/10.1016/j.surfcoat.2020.125879
B. Sundman, H. L. Lukas, and S. G. Fries, Computational Thermodynamics: The Calphad Method (Cambridge Univ. Press, New York, 2007), p. 313.
A. S. Pavlenko, E. A. Ptashkina, E. G. Kabanova, et al., CALPHAD 81, 102533 (2023). https://doi.org/10.1016/j.calphad.2023.102533
G. H. Laurie and J. N. Pratt, J. Chem. Soc., Faraday Trans. 60, 1391 (1964). https://doi.org/10.1039/TF9646001391
C. Luef, A. Paul, H. Flandorfer, et al., J. Alloys Compd. 391, 67 (2005). https://doi.org/10.1016/j.jallcom.2004.08.056
A. S. Pavlenko, E. G. Kabanova, and V. N. Kuznetsov, Russ. J. Phys. Chem. A 94, 2691 (2020). https://doi.org/10.1134/s0036024420130178
Thermo-Calc Software PURE5/SGTE Pure Element Database. https://thermocalc.com/about-us/methodology/the-calphad-methodology/assessment-of-thermodynamic-data/
G. Ghosh, C. Kantner, and G. B. Olson, J. Phase Equilib. 20, 295 (1999). https://doi.org/10.1361/105497199770335811
W. Gierlotka, Y. C. Huang, and S. W. Chen, Metall. Mater. Trans. A 39, 3199 (2008). https://doi.org/10.1007/s11661-008-9671-6
G. Vassilev, V. Gova, N. Milcheva, et al., Calphad 43, 133 (2013). https://doi.org/10.1016/j.calphad.2013.03.003
S. Cui, J. Wang, Z. You, et al., Intermetallics 126, 106945 (2020). https://doi.org/10.1016/j.intermet.2020.106945
O. Redlish and A. T. Kister, Ind. Eng. Chem. 40, 345 (1948). https://doi.org/10.1021/ie50458a036
G. W. Toop, Trans. Metall. Soc. AIME 233, 850 (1965).
J.-O. Andersson, T. Heler, L. Höglund, et al., Calphad 26, 273 (2002). https://doi.org/10.1016/s0364-5916(02)00037-8
A. S. Pavlenko, E. A. Ptashkina, G. P. Zhmurko, E. G. Kabanova, M. A. Kareva, A. V. Khoroshilov, and V. N. Kuznetsov, Russ. J. Phys. Chem. A 97, 42 (2023). https://doi.org/10.1134/S0036024423010235
A. S. Pavlenko, E. G. Kabanova, M. A. Kareva, et al., Materials 16, 1690 (2023). https://doi.org/10.3390/ma16041690
V. N. Kuznetsov and E. G. Kabanova, Calphad 51, 346 (2015). https://doi.org/10.1016/j.calphad.2015.01.011
S. Cui, J. Wang, and I. H. Jung, Metall. Mater. Trans. A 53, 4296 (2022). https://doi.org/10.1007/s11661-022-06825-9
Funding
The present research work was financially supported by Russian Science Foundation (RSF) grant no. 22-23-00565 (https://rscf.ru/en/project/22-23-00565/).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
The authors declare that they have no conflicts of interest.
Rights and permissions
About this article
Cite this article
Pavlenko, A.S., Zhmurko, G.P., Kabanova, E.G. et al. CALPHAD Modelling of Ag–Pd–Sn Ternary System. Russ. J. Phys. Chem. 97, 2006–2012 (2023). https://doi.org/10.1134/S0036024423090145
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0036024423090145