Abstract
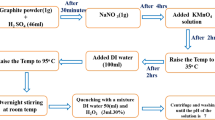
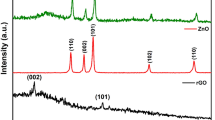
Nitrogen-sulfur quantum dot co-modified graphene nanosheets composites (NSG) were fabricated via a facile hydrothermal method as a high performance metal-free catalyst for methyl orange (MO) degradation. Physical-chemical properties of the obtained samples were characterized by XRD, FTIR, SEM, TEM, EDX, TGA, BET, and UV–Vis DRS technologies. Compared with RGO, NSG exhibited much better photocatalytic performance for MO degradation under visible light, especially, NSG(30) showed the best photocatalytic degradation rate of 100% within 180 min. XRD, TG, and UV–Vis DRS results demonstrated that the addition of N and S can enhance the stability of oxygen containing functional groups to supply more adsorption active centers for organic contaminant molecular, adjust the band gap to a certain extent and increase the number of photogenerated electrons, improve the visible light utilization rate of the material and thus improve the photocatalytic degradation performance of the NSG composites. The degradation activity of the catalyst remained above 90% after 3 recycles. Finally, a possible photodegradation mechanism of MO on the obtained NSG sample was proposed.







Similar content being viewed by others
REFERENCES
G. McMullan, C. Meehan, A. Conneely, N. Kirby, T. Robinson, P. Nigam, I. Banat, R. Marchant, and W. Smyth, Appl. Microbiol. Biot. 56, 81 (2001). https://doi.org/10.1007/s002530000587
F. Wang, Chem. Eng. Res. Des. 128, 155 (2017). https://doi.org/10.1016/j.cherd.2017.10.007
F. Wang, J. Porous. Mater. 24, 1309 (2017). https://doi.org/10.1007/s10934-017-0372-7
F. Wang and Y. Q. Ma, Mater. Chem. Phys. 208, 157 (2018). https://doi.org/10.1016/j.matchemphys.2018.01.049
A. Yang, G. Zhang, F. Meng, P. Y. Zhang, and Y. Chen, Bioresour. Technol. 253, 378 (2018). https://doi.org/10.1016/j.biortech.2018.01.034
Y. Q. Zhan, X. Y. Wan, S. J. He, Q. B. Yang, and Y. He, Chem. Eng. J. 333, 132 (2018). https://doi.org/10.1016/j.cej.2017.09.147
F. Wang, Chem. Eng. Res. Des. 142, 189 (2019). https://doi.org/10.1016/j.cherd.2018.12.013
J. X. Yao, K. Zhang, W. Wang, X. Q. Zuo, Q. Yang, H. B. Tang, M. Z. Wu, and G. Li, ACS Appl. Mater. Interface 10, 19564 (2018). https://doi.org/10.1021/acsami.8b01240
L. N. Yang, J. G. Hu, L. L. He, J. Tang, Y. Ch. Zhou, J. Li, and K. X. Ding, Chem. Eng. J. 327, 694 (2017). https://doi.org/10.1016/j.cej.2017.06.162
J. Di, Ch. Chen, Ch. Zhu, M. X. Ji, J. X. Xia, Yan Cheng, W. Hao, Sh. Zh. Li, H. M. Li, and Zh. Liu, Appl. Catal., B: Environ. 238, 119 (2018). https://doi.org/10.1016/j.apcatb.2018.06.066
S. Shanmuganathan, P. Loganathan, M. A. H. Johir, and S. Vigneswaran, Desalination 401, 134 (2016). https://doi.org/10.1111/j.1749-6632.1986.tb47973.x
Y. Q. Liao, Y. L. Huang, D. Shu, Y. Y. Zhong, J. N. Hao, Ch. He, J. Zhong, and X. N. Song, Electrochim. Acta 194, 136 (2016). https://doi.org/10.1016/j.electacta.2016.02.067
Zhao, Sh. Sh, T. T. Yan, H. Wang, G. R. Chen, L. Huang, J. P. Zhang, L. Y. Shi, and D. S. Zhang, Appl. Surf. Sci. 36, 460 (2016). https://doi.org/10.1016/j.apsusc.2016.02.085
J. Zhou, H. L. Shen, Zh. H. Li, Sh. Zhang, Y. T. Zhao, X. Bi, Y. Sh. Wang, H. Y. Cui, and Sh. P. Zhuo, Electrochim. Acta 209, 557 (2016). https://doi.org/10.1016/j.electacta.2016.05.127
D. Hadis and N. E. Alireza, J. Colloid Interface Sci. 490, 628 (2017). https://doi.org/10.1016/j.jcis.2016.11.102
J. M. Yang, B. Ch. Yang, and Y. Zhang, Microporous Mesoporous Mater. 292, 109764 (2020). https://doi.org/10.1016/j.micromeso.2019.109764
J. Y. Zhang, D. Xu, W. J. Qian, J. Y. Zhu, and F. Yan, Carbon 105, 183 (2016). https://doi.org/10.1016/j.carbon.2016.04.034
W. Zh. Chen, J. J. Shi, T. Sh. Zhu, Q. Wang, J. L. Qiao, and J. J. Zhang, Electrochim. Acta 17, 327 (2015). https://doi.org/10.1016/j.electacta.2015.01.093
J. H. Li, G. P. Zhang, Ch. P. Fu, L. B. Deng, R. Sun, and Ch. P. Wong, J. Power Sources 345, 146 (2017). https://doi.org/10.1016/j.jpowsour.2017.02.011
M. Shah, A. R. Park, K. Zhang, J. H. Park, and P. J. Yoo, ACS Appl. Mater. Interface 4, 3893 (2012). https://doi.org/10.1021/am301287m
V. V. Walatka, Jr., M. M. Labes, and J. H. Perlstein, Phys. Rev. Lett. 31, 1139 (1973). https://doi.org/10.1103/PhysRevLett.31.1139
F. Wang, J. Porous Mater. 24, 1309 (2017). https://doi.org/10.1007/s10934-017-0372-7
L. Chen, L. Song, Y. Zhang, P. Wang, Z. Xiao, Y. Guo, and F. Cao, ACS Appl. Mater. Interface 8, 11255 (2016). https://doi.org/10.1021/acsami.6b01030
J. Wang, S. Zheng, Y. Shao, J. Liu, Z. Xu, and D. Zhu, J. Colloid Interface Sci. 349, 293 (2010). https://doi.org/10.1016/j.jcis.2010.05.010
X. Zh. Guo, Sh. Sh. Han, J. M. Yang, et al., Ind. Eng. Chem. Res. 59, 2113 (2020). https://doi.org/10.1021/acs.iecr.9b05715
W. Zhang, R. Zh. Zhang, Y. Yin, et al., J. Mol. Liq. 302, 112616 (2020).
S. Kimiagar and F. Abrinaei, Nanophotonics 7, 243 (2018). https://doi.org/10.1080/19443994.2016.1183233
S. Gao, H. Liu, K. Geng, and X. J. Wei, Nano Energy 12, 785 (2015). https://doi.org/10.1016/j.nanoen.2015.02.004
ACKNOWLEDGMENTS
The work was supported by the Shandong Natural Science Foundation of China (ZR2018QB007), Key R&D Program Projects in Shandong Province (2019GSF109117), a project of Shandong Province Higher Educational Science and Technology Program (J14LC54), a project of Binzhou City science and technology development project (2014ZC0212), and Binzhou University (BZXYHZ20161010, BZXYG1607, and BZXYL1801) research Funds.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Fang Wang, Zhang, H. & Xu, B. Nitrogen and Sulfur Quantum Dot Co-Modified Graphene Nanosheet with Enhanced Photocatalytic Activity for Methyl Orange Degradation. Russ. J. Phys. Chem. 94, 2299–2305 (2020). https://doi.org/10.1134/S0036024420110333
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0036024420110333