Abstract
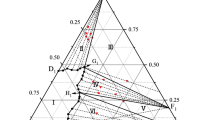
The solubility of components in the NaClO3 · 2CO(NH2)2-NH2C2H4OH · CH3COOH-H2O system was studied by the visual polythermal method over wide temperature and concentration ranges. In the phase diagram, crystallization fields were determined for ice, urea, diurea sodium chlorate, acetic acid, monoethanolamine acetate, and the compound CO(NH2)2 · NH2C2H4OH · CH3COOH. The compound was identified by chemical, thermogravimetric, and X-ray powder diffraction analyses.
Similar content being viewed by others
References
N. F. Zubkova, O. G. Nadotchaya, and L. D. Stonov, Khim. Sel’sk. Khoz. 13(8), 32 (1975).
F. G. Isaev, Proceedings of XII Mendeleev’s Congress on General and Applied Chemistry (Moscow, 1981), No. 6, p. 157 [in Russian].
L. D. Nikel, Plant Grwoth Regulators (Kolos, Moscow, 1984) [in Russian].
L. I. Mirkin, Handbook of X-Ray Analysis of Polycrystalline Materials (Fizmatgiz, Moscow, 1961; Plenum Press, New York, 1964).
M. T. Abdullaeva, Uzb. Khim. Zh., No. 3, 50 (2008).
Author information
Authors and Affiliations
Additional information
Original Russian Text © Zh.S. Shukurov, S.S. Ishankhodzhaev, M.K. Askarova, S. Tukhtaev, 2011, published in Zhurnal Neorganicheskoi Khimii, 2011, Vol. 56, No. 3, pp. 502–505.
Rights and permissions
About this article
Cite this article
Shukurov, Z.S., Ishankhodzhaev, S.S., Askarova, M.K. et al. Study of the solubility of components in the NaClO3 · 2CO(NH2)2-NH2C2H4OH · CH3COOH-H2O system. Russ. J. Inorg. Chem. 56, 463–466 (2011). https://doi.org/10.1134/S0036023611010207
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0036023611010207