Abstract—
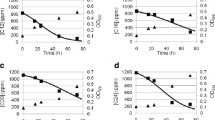
3-Chlorobenzoic acid (3-CBA) is widely used as a precursor/preservative in industry and agriculture and is therefore a known environmental contaminant. The key stages of 3-CBA decomposition by Rhodococcus opacus strains 1CP and 6a were studied. Comparative characterization of the substrate specificity of 3-chlorobenzoate 1,2-dioxygenase (3-CBA 1,2-DO), induced in the strains grown in the presence of 3‑CBA, was carried out. These enzymes were established to have a wider substrate specificity than the benzoate 1,2-dioxygenase (1,2-BDO) of R. opacus strain 1CP, which is induced during growth of R. opacus strain 1CP in the presence of benzoate. Benzoate, 3-CBA, and 3,4-dihydroxybenzoate served as substrates for 3‑CBA 1,2-DO. During the degradation of 3-CBA by R. opacus 1CP cells, both 3-chloro- and 4-chlorocatechol (3-CCat and 4-CCat) were detected. R. opacus 6a efficiently degraded 3-CBA without accumulation of intermediates. The difference in the pathways of 3-CBA degradation by these strains was shown: via the pathway of ortho-cleavage of 3-chlorocatechol in R. opacus 1CP and of 4-chlorocatechol in R. opacus 6a. In the genome of the strain R. opacus 6a, the genes encoding chlorocatechol 1,2-dioxygenase and chloromuconate cycloisomerase were found, which were 98-99% identical to the genes of R. opacus 1CP encoding 4‑chlorocatechol 1,2-dioxygenase (4-CCat 1,2-DO) and 3-chloromuconate cycloisomerase (3-CMCI) of the modified ortho-cleavage pathway for conversion of 4-chlorocatechol (the intermediate of 4-chlorophenol degradation). It was shown for the strains under study that implementation of different pathways for 3-CBA decomposition was predestined not by the metabolic capabilities of bacteria, but by the substrate specificity of 3-CBA 1,2-DO, the enzyme that initiates 3-CBA degradation.


Similar content being viewed by others
REFERENCES
Abbasian, F., Lockington, R., Megharaj, M., and Naidu, R., A review on the genetics of aliphatic and aromatic hydrocarbon degradation, Appl. Biochem. Biotechnol., part A: Enzyme Eng. Biotechnol., 2016, vol. 178, pp. 224–250.
Adebusoye, S.A., Biological degradation of 4-chlorobenzoic acid by a PCB-metabolizing bacterium through a pathway not involving (chloro)catechol, Biodegradation, 2017, vol. 28, pp. 37–51.
Ajithkumar, P.V. and Kunhi, A.A.M., Pathways for 3-chloro- and 4-chlorobenzoate degradation in Pseudomonas aeruginosa 3mT, Biodegradation, 2000, vol. 11, pp. 247–261.
Aken, B.V., Correa, P.A., and Schnoor, J.L., Phytoremediation of polychlorinated biphenyls: new trends and promises, Environ. Sci. Technol., 2010, vol. 44, pp. 2767–2776.
Baggi, G. and Zangrossi, M., Assessment of the biodegradative potential versus chlorobenzoates as single or mixed compounds in a stable microbial consortium, Annu. Rev. Microbiol., 2001, vol. 51, pp. 179–188.
Boyle, A.W., Silvin, C.J., Hassett, J.P., Nakas, J.P., and Tanenbaum, S.W., Bacterial PCB degradation, Biodegradation, 1992, vol. 3, pp. 285–298.
Engesser, K.H., Auling, G., Busse, J., and Knackmusss, H.-J., 3-Fluorobenzoate enriched bacterial strain FLB300 degrades benzoate and all three isomeric monofluorobenzoates, Arch. Microbiol., 1990, vol. 153, pp. 193–199.
Field, J.A. and Sierra-Alvarez, R., Microbial transformation and degradation of polychlorinated biphenyls, Environ. Pollut., 2008, vol. 155, pp. 1–12.
Field, J.A. and Sierra-Alvarez, R., Microbial transformation of chlorinated benzoates, Rev. Environ. Sci. Bio/Technol., 2008, vol. 7, pp. 191–210.
Gorlatov, S.N., Maltseva, O.V., Shevchenko, V.I., and Golovleva, L.A., Degradation of chlorophenols by a culture of Rhodococcus erythropolis, Microbiology (Moscow), 1989, vol. 58, pp. 647–651.
Gribble, G.W., The abundant natural sources and uses of chlorinated chemicals, Am. J. Public Health, 1994, vol. 84, p. 1183.
Gröning, J.A.D., Roth, C., Kaschabek, S.R., Sträter, N., and Schlömann, M., Recombinant expression of a unique chloromuconolactone dehalogenase ClcF from Rhodococcus opacus 1CP and identification of catalytically relevant residues by mutational analysis, Arch. Biochem. Biophys., 2012, vol. 526, pp. 69–77.
Harayama, S., Rekik, M., Bairoch, A., Neidle, E.L., and Ornston, L.N., Potential DNA slippage structures acquired during evolutionary divergence of Acinetobacter calcoaceticus chromosomal benABC and Pseudomonas putida TOL pWW0 plasmid xylXYZ, genes encoding benzoate dioxygenases, J. Bacteriol., 1991, vol. 173, pp. 7540–7548.
Hartmann, J., Reineke, W., and Knackmuss, H.-J., Metabolism of 3-chloro-, 4-chloro-, and 3,5-dichlorobenzoate by a pseudomonad, Appl. Environ. Microbiol., 1979, vol. 37, pp. 421–428.
Horvath, R.S. and Alexander, M., Cometabolism of meta-chlorobenzoate by an Arthrobacter, Appl. Microbiol., 1970, vol. 20, pp. 254–258.
Ichihara, A., Adachi, K., Hosokawa, K., and Takeda, Y., The enzymatic hydroxylation of aromatic carboxylic acids; substrate specificities of anthranilate and benzoate oxidases, J. Biol. Chem., 1962, vol. 237, pp. 2296–2302.
Ihn, W. Janke, D., and Tresselt, D., Critical steps in degradation of chloroaromatics by rhodococci. III. Isolation and identification of accumulating intermediates and dead-end products, J. Basic Microbiol., 1989, vol. 29, pp. 291–297.
Janke, D., Al-Mofarji, T., and Schukat, B., Critical steps in degradation of chloroaromatics by rhodococci. II. Whole-cell turnover of different monochloroaromatic non-growth substrates by Rhodococcus sp. An 117 and An 213 in the absence/presence of glucose, J. Basic Microbiol., 1988a, vol. 28, pp. 519–528.
Janke, D., Al-Mofarji, T., Straube, G., Schumann, P., and Prauser, H., Critical steps in degradation of chloroaromatics by rhodococci. I. Initial enzyme reactions involved in catabolism of aniline, phenol and benzoate by Rhodococcus sp. An 117 and An 213, J. Basic Microbiol., 1988b, vol. 28, pp. 509–518.
Janke, D. and Ihn, W., Cometabolic turnover of aniline, phenol and some of their monochlorinated derivatives by the Rhodococcus mutant strain AM 144, Arch. Microbiol., 1989, vol. 152, pp. 347–352.
Janke, D., Ihn, W., and Tresselt, D., Critical steps in degradation of chloroaromatics by rhodococci IV. Detailed kinetics of substrate removal and product formation by resting pre-adapted cells, J. Basic Microbiol., 1989, vol. 29, pp. 305–314.
Kitagawa, W., Miyauchi, K., Masai, E., and Fukuda, M., Cloning and characterization of benzoate catabolic genes in the gram-positive polychlorinated biphenyl degrader Rhodococcus sp. strain RHA1, J. Bacteriol., 2001, vol. 183, pp. 6598–6606.
Konovalova, E.I., Solyanikova, I.P., and Golovleva, L.A., Degradation of 4-chlorophenol by bacterium Rhodococcus opacus 6a, Microbiology (Moscow), 2009, vol. 78, pp. 805–807.
Kweon, O., Kim, S.J., Freeman, J.P., Song, J., Baek, S., and Cerniglia, C.E., Substrate specificity and structural characteristics of the novel Rieske nonheme iron aromatic ring-hydroxylating oxygenases NidAB and NidA3B3 from Mycobacterium vanbaalenii PYR-1, mBio, 2010, vol. 1. pii: e00135-10.
Liang, B., Jiang, J., Zhang, J., Zhao, Y., and Li, S., Horizontal transfer of dehalogenase genes involved in the catalysis of chlorinated compounds: evidence and ecological role, Crit. Rev. Microbiol., 2012, vol. 38, pp. 95–110.
Moiseeva, O.V., Linko, E.V., Baskunov, B.P., and Golovleva, L.A., Degradation of 2-chlorophenol and 3-chlorobenzoate by Rhodococcus opacus 1CP, Microbiology (Moscow), 1999, vol. 68, pp. 400–405.
Moiseeva, O.V., Solyanikova, I.P., Kaschabek, S., Thiel, M., Golovleva, L.A., and Schlömann, M., A new modified ortho-cleavage pathway of 2-chlorophenol degradation by Rhodococcus opacus 1CP: genetic and biochemical evidences, J. Bacteriol., 2002, vol. 184, pp. 5282–5292.
Moisseeva, O.V., Belova, O.V., Solyanikova, I.P., Schlömann, M., and Golovleva, L.A., Enzymes of novel modified ortho-pathway of Rhodococcus opacus 1CP, utilizing 2-chlorophenol, Biochemistry (Moscow), 2001, vol. 66, pp. 548–555.
Parales, R.E. and Resnick, S.M., Aromatic ring hydroxylating dioxygenases, Mol. Biol. Emerg. Iss., 2006, vol. 4, pp. 287–340.
Reineke, W. and Knackmuss, H.-J., Chemical structure and biodegradability of halogenated aromatic compounds. Substituent effects on 1,2-dioxygenation of benzoic acid, Biochim. Biophys. Acta,1978, vol. 542, pp. 412–423.
Schlömann, M., Evolution of chlorocatechol catabolic pathways, Biodegradation, 1994, vol. 5, pp. 301–321.
Schlömann, M., Schmidt, E., and Knackmuss, H.-J., Different types of dienelactone hydrolase in 4-fluorobenzoate-utilizing bacteria, J. Bacteriol., 1990, vol. 172, pp. 5112–5118.
Solyanikova, I.P., Borzova, O.V., Emelyanova, E.V., Shumkova, E.S., Prisyazhnaya, N.V., Plotnikova, E.G., and Golovleva, L.A., Dioxygenases of chlorobiphenyl-degrading species Rhodococcus wratislaviensis G10 and chlorophenol-degrading species Rhodococcus opacus 1CP induced in benzoate-grown cells and genes potentially involved in these processes, Biochemistry (Moscow), 2016, vol. 81, pp. 986–999.
Solyanikova, I.P., Emelyanova, E.V., Borzova, O.V., and Golovleva, L.A., Benzoate degradation by Rhodococcus opacus 1CP after a dormancy: characterization of dioxygenases involved in the process, J. Environ. Sci. Health. Part B, 2016, vol. 51, pp. 182–191.
Solyanikova, I.P., Maltseva, O.V., Vollmer, M.D., Golovleva, L.A., and Schlömann, M., Characterization of muconate and chloromuconate cycloisomerase from Rhodococcus erythropolis 1CP: indications for functionally convergent evolution among bacterial cycloisomerases, J. Bacteriol., 1995, vol. 177, pp. 2821–2826.
Solyanikova, I.P., Plotnikova, E.G., Shumkova, E.S., Robota, I.V., Prisyazhnaya, N.V., and Golovleva, L.A., Chloromuconolactone dehalogenase ClcF of actinobacteria, J. Environ. Sci. Health. Part B, 2014, vol. 49, pp. 422–431.
Thiel, M., Entwicklung von PCR-Primern zum Nachweis von Genen des Chloraromaten-Abbaus in mikrobiellen Lebensgemeinschaften, PhD Thesis, Freiberg: Techn. Univ. Bergakademie, 2004.
Wang, H., Hu, J., Xu, K., Tang, X., Xu, X., and Shen, C., Biodegradation and chemotaxis of polychlorinated biphenyls, biphenyls, and their metabolites by Rhodococcus spp., Biodegradation, 2018, vol. 29, pp. 1–10. https://doi.org/10.1007/s10532-017-9809-6
Willes, R.F., Nestmann, E.R., Miller, P.A., Orr, J.C., and Munro, I.C., Scientific principles for evaluating the potential for adverse effects from chlorinated organic chemicals in the environment, Regul. Toxicol. Pharmacol., 1993, vol. 18, pp. 313–356.
Xu, C., Zang, X., Hang, X., Liu, X., Yang, H., Liu, X., and Jiang J., Degradation of three monochlorobenzoate isomers by different bacteria isolated from a contaminated soil, Int. Biodeter. Biodegr., 2017, vol. 120, pp. 192–202. https://doi.org/10.1016/j.ibiod.2017.02.020
Yamaguchi, M. and Fujisawa, H., Reconstitution of iron-sulfur cluster of NADH-cytochrome c reductase, a component of benzoate 1,2-dioxygenase system from Pseudomonas arvilla C-1, J. Biol. Chem., 1981, vol. 256, pp. 6783–6787.
Zhang, G., Yang, X., Xie, F., Chao, Y., and Qian, S., Cometabolic degradation of mono-chloro benzoic acids by Rhodococcus sp. R04 grown on organic carbon sources, World J. Microbiol. Biotechnol., 2009, vol. 25, pp. 1169–1174.
ACKNOWLEDGMENTS
The authors are very grateful to Prof. Dr. M. Schlömann (TU Bergakademie, Freiberg, Germany) for the possibility of working on gene cloning at his laboratory and to Tech. Acc. J. Gröning for assistance in the work and for providing the strain Escherichia coli DH5α.
Funding
The work was supported by the Russian Science Foundation (grant no. 14-14-00368) and by the DAAD Stipendium of I.P. Solyanikova.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflict of interest. This article does not contain any studies involving animals or human participants performed by any of the authors.
Additional information
Translated by E. Makeeva
Abbreviations: 1,2-BDO, benzoate 1,2-dioxygenase; DLH, dienelactone hydrolase; 3,5- and 4,5-DCCat, 3,5- and 4,5-dichlorocatechols; OD, optical density; 3-MCat and 4-MCat, 3- and 4‑methylcatechol; Cat, catechol; 1,2-CDO, catechol 1,2-dioxygenase; PCB – polychlorinated biphenyl; 3-CA, 3-chloroaniline; CBA, chlorobenzoic acid; 2-CBA, 2-chlorobenzoic acid; 3‑CBA, 3-chlorobenzoic acid; 4-CBA, 4-chlorobenzoic acid; 3‑CBA 1,2-DO, 3-chlorobenzoate-1,2-dioxygenase; 2-CM, 2‑chloromuconate; 5-CML, 5-chloromuconolactone; CMLD, chloromuconolactone dehalogenase; 3-CMCI, 3‑chloromuconate cycloisomerase; 3-CCat and 4-CCat, 3- and 4-chlorocatechol; 4-CCat 1,2-DO, 4-chlorocatechol 1,2-dioxygenase; 2-CP, 2-chlorophenol; 3-CP, 3-chlorophenol; 4-CP, 4‑chlorophenol.
Rights and permissions
About this article
Cite this article
Solyanikova, I.P., Emelyanova, E.V., Shumkova, E.S. et al. Pathways of 3-Chlorobenzoate Degradation by Rhodococcus opacus strains 1CP and 6a. Microbiology 88, 563–572 (2019). https://doi.org/10.1134/S002626171905014X
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S002626171905014X