Abstract
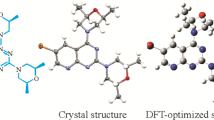
A new pyrido[2,3-d]pyrimidine derivative 1-(3-((6-bromopyrido[2,3-d]pyrimidin-4-yl)oxy)phenyl)-3-cyclopentylurea is designed and synthesized. The final structure is characterized by 1H, 13C, and 2D NMR, MS, FTIR. In addition, the crystal structure of the title compound is determined by X-ray diffraction. With the 6-311G(2d,p) basis set, the molecule is further explored using density functional theory (DFT) by the B3LYP method. The final results show that the DFT optimized structure of the title molecule is consistent with the crystal structure determined by X-ray diffraction. The Hirshfeld surface analysis and the 2D fingerprint plot are given to support the quantitative analysis of intermolecular interactions and contacts generated by supramolecular accumulation in crystals. The interactions of the title molecule are analyzed by the natural bond orbital analysis. Finally, the molecular electrostatic potential and frontier molecular orbitals are further investigated using DFT.











Similar content being viewed by others
REFERENCES
F. Buron, J. Y. Mérour, M. Akssira, G. Guillaumet, and S. Routier. Eur. J. Med. Chem., 2015, 95, 76-95. https://doi.org/10.1016/j.ejmech.2015.03.029
H. Elzahabi, E. S. Nossier, N. M. Khalifa, R. A. Alasfoury, and M. A. El-Manawaty. J. Enzyme Inhib. Med. Chem., 2018, 33, 546-557. https://doi.org/10.1080/14756366.2018.1437729
J. W. Ellingboe, M. Antane, T. T. Nguyen, M. D. Collini, S. Antane, R. Bender, D. Hartupee, V. White, J. McCallum, C. H. Park, and A. Et. J. Med. Chem., 1994, 37, 542-550. https://doi.org/10.1021/jm00030a013
O. A. Abdelaziz, W. M. El Husseiny, K. B. Selim, and H. M. Eisa. Bioorg. Chem., 2019, 90, 103076. https://doi.org/10.1016/j.bioorg.2019.103076
A. S. Kalgutkar, D. A. Griffith, T. Ryder, H. Sun, Z. Miao, J. N. Bauman, M. T. Didiuk, K. S. Frederick, S. X. Zhao, C. Prakash, J. R. Soglia, S. W. Bagley, B. M. Bechle, R. M. Kelley, K. Dirico, M. Zawistoski, J. Li, R. Oliver, A. Guzman-Perez, K. K. C. Liu, D. P. Walker, J. W. Benbow, and J. Morris. Chem. Res. Toxicol., 2010, 23, 1115-1126. https://doi.org/10.1021/tx100137n
N. Kahriman, K. Peker, V. Serdaroğlu, A. Aydin, A. Usta, S. Fandakli, and N. Yayli. Bioorg. Chem., 2020, 99, 103805. https://doi.org/10.1016/j.bioorg.2020.103805
M. Lavanya, I. V. Asharani, and D. Thirumalai. Chem. Biol. Drug Des., 2019, 93, 464-472. https://doi.org/10.1111/cbdd.13434
A. E. Rashad, H. H. Sayed, A. H. Shamroukh, and H. M. Awad. Phosphorus, Sulfur Silicon Relat. Elem., 2005, 180, 2767-2777. https://doi.org/10.1080/104265090968118
L. N. Bheemanapalli, R. R. Akkinepally, and S. R. Pamulaparthy. Chem. Pharm. Bull. (Tokyo), 2008, 56, 1342-1348. https://doi.org/10.1002/chir.20471
L. G. Marileo, M. A. Jorquera, M. Hernández, G. Briceño, M. de La Luz Mora, R. Demanet, and G. Palma. Appl. Soil Ecol., 2016, 101, 141-151. https://doi.org/10.1016/j.apsoil.2016.02.003
W. T. Lambert, A. M. Buysse, and F. J. Wessels. Pest Manag. Sci., 2019, 76, 497-508. https://doi.org/10.1002/ps.5537
E. Leo, A. Krämer, A. Hochhaus, F. Krasniqi, R. Hehlmann, and A. Ho. Ann. Hematol., 2002, 81, 467-469. https://doi.org/10.1007/s00277-002-0505-0
Y. Wei, P. Yang, S. Cao, and L. Zhao. Arch. Pharm. Res., 2018, 41, 1-13. https://doi.org/10.1007/s12272-017-0979-x
L. Crotti, K. E. Odening, and M. C. Sanguinetti. Cardiovasc. Res., 2020, 116, 1542-1556. https://doi.org/10.1093/cvr/cvaa068
J. S. Fortin, J. Lacroix, M. Desjardins, A. Patenaude, É. Petitclerc, and R. C. Gaudreault. Bioorg. Med. Chem., 2007, 15, 4456-4469. https://doi.org/10.1016/j.bmc.2007.04.028
J. Liu, Y. Li, X. Zhang, D. Cheng, W. Wei, C. Wu, Y. Xie, L. Xiong, and Z. Li. Chin. J. Chem., 2017, 35, 368-374. https://doi.org/10.1002/cjoc.201600711
X. Zhang, X. G. Lu, and C. Cai. Green Chem., 2016, 18, 5580-5585. https://doi.org/10.1039/C6GC01742H
E. Margiotta, C. Stephanie, G. Paragi, L. A. Santos, G. Paragi, S. Moro, C. F. Guerra, and F. Bickelhaupt. J. Chem. Inf. Model., 2020, 60, 1317-1328. https://doi.org/10.1021/acs.jcim.9b00946
M. Z. Hernandes, S. M. T. Cavalcanti, D. R. M. Moreira, W. F. de Azevedo Jr., and A. C. L. Leite. Curr. Drug Targets, 2010, 11, 303-314. https://doi.org/10.2174/138945010790711996
R. Adepu, R. Sunke, C. L. T. Meda, D. Rambabu, G. R. Krishna, C. M. Reddy, G. S. Deora, K. V. L. Parsa, and M. Pal. Chem. Commun., 2013, 49, 190-192. https://doi.org/10.1039/C2CC37070K
B. Bernd, G. Bindu, and P. Solang. Patent WO 2010/097335, 2009.
T. C. Leboho, S. F. van Vuuren, J. P. Michael, and C. B. de Koning. Org. Biomol. Chem., 2014, 12, 307-315. https://doi.org/10.1039/C3OB41798K
C. M. Lacbay, M. Menni, J. A. Bernatchez, M. Götte, and Y. S. Tsantrizos. Bioorg. Med. Chem., 2018, 26, 1713-1726. https://doi.org/10.1016/j.bmc.2018.02.017
Z. Sun, H. Wang, K. Wen, Y. Li, and E. Fan. J. Org. Chem., 2011, 76, 4149-4153. https://doi.org/10.1021/jo2003715
P. Tanja, M. Marina, and V. Srinivasamurthy. Patent WO 2019/229464, 2019.
S. Gupta, P. Chaudhary, L. Seva, S. Sabiah, and J. Kandasamy. RSC Adv., 2015, 5, 89133-89138. https://doi.org/10.1039/C5RA18080E
X. Liu, D. Kang, P. Zhan, and Z. Wang. Patent CN 108409734, 2018.
Y. Liu, Y. Zhao, Q. Ren, Z. Zhou, H. Chai, and C. Zhao. J. Mol. Struct., 2020, 1208, 127869. https://doi.org/10.1016/j.molstruc.2020.127869
A. V. Ivachtchenko, P. M. Yamanushkin, O. D. Mitkin, and O. I. Kiselev. Mendeleev Commun., 2010, 20, 111/112. https://doi.org/10.1016/j.mencom.2010.03.016
G. Sheldrick. Acta Crystallogr., Sect. A: Found. Adv., 2014, 70, C1437. https://doi.org/10.1107/S2053273314085623
P. Geerlings, F. De Proft, and W. Langenaeker. Chem. Rev., 2003, 103, 1793-1874. https://doi.org/10.1021/cr990029p
M. Frisch, G. Trucks, H. Schlegel, G. Scuseria, M. Robb, J. Cheeseman, G. Scalmani, V. Barone, B. Mennucci, and G. Petersson. Gaussian09, Revision A. 1. Wallingford, CT: Gaussian Inc., 2009.
B. J. Deppmeier, A. J. Driessen, T. S. Hehre, W. J. Hehre, J. A. Johnson, P. E. Klunzinger, J. M. Leonard, I. N. Pham, W. J. Pietro, and J. Yu. Spartan08. Irvine, CA: Wavefunction Inc., 2009.
N. Kanagathara, F. MaryAnjalin, V. Ragavendran, D. Dhanasekaran, R. Usha, and R. G. S. Rao. J. Mol. Struct., 2021, 1223, 128965. https://doi.org/10.1016/j.molstruc.2020.128965
Q. Wu, Y. Chen, D. Chen, and Z. Zhou. J. Mol. Struct., 2021, 1229, 129782. https://doi.org/10.1016/j.molstruc.2020.129782
A. S. M. Gad El-Hak, A. A. K. Mohammed, A. F. Abdel Hakiem, and R. M. Mahfouz. Spectrochim. Acta, Part A, 2019, 222, 117200. https://doi.org/10.1016/j.saa.2019.117200
R. I. Al-Wabli, K. S. Resmi, Y. S. Mary, C. Y. Panicker, M. I. Attia, A. A. El-Emam, and C. Van Alsenoy. J. Mol. Struct., 2016, 1123, 375-383. https://doi.org/10.1016/j.molstruc.2016.07.044
S. Gatfaoui, N. Issaoui, T. Roisnel, and H. Marouani. J. Mol. Struct., 2019, 1191, 183-196. https://doi.org/10.1016/j.molstruc.2019.04.093
Y. Lu, W. Xu, H. Sun, J. Jin, H. Liu, S. Jin, D. Wang, and M. Guo. J. Mol. Struct., 2019, 1178, 639-654. https://doi.org/10.1016/j.molstruc.2018.10.080
H. Gomes, P. Rosina, P. Holakooei, T. Solomon, and C. Vaccaro. J. Archaeol. Sci., 2013, 40, 4073-4082. https://doi.org/10.1016/j.jas.2013.04.017
V. Ferraresi-Curotto, G. A. Echeverría, O. E. Piro, and R. Pis-Diez. J. Mol. Struct., 2017, 1133, 436-447. https://doi.org/10.1016/j.molstruc.2016.12.018
K. Karrouchi, S. Fettach, M. M. Jotani, A. Sagaama, S. Radi, H. A. Ghabbour, Y. N. Mabkhot, B. Himmi, M. El Abbes Faouzi, and N. Issaoui. J. Mol. Struct., 2020, 1221, 128800. https://doi.org/10.1016/j.molstruc.2020.128541
L. V. de Freitas, C. C. P. Da Silva, J. Ellena, L. A. S. Costa, and N. A. Rey. Spectrochim. Acta, Part A, 2013, 116, 41-48. https://doi.org/10.1016/j.saa.2013.06.105
P. F. Bernath and E. L. Sibert III. J. Phys. Chem. A, 2020, 124, 9991-10000. https://doi.org/10.1021/acs.jpca.0c09185
D. Luo, L. Ma, Z. Zhou, and Z. Huang. J. Mol. Struct., 2019, 1198, 126857. https://doi.org/10.1016/j.molstruc.2019.07.104
M. Watanabe, H. Kajiwara, K. Awazu, and K. Aizawa. Surg. Today, 2001, 31, 626-633. https://doi.org/10.1007/s005950170097
K. Karrouchi, S. A. Brandán, Y. Sert, H. El-marzouqi, S. Radi, M. Ferbinteanu, M. E. A. Faouzi, Y. Garcia,and M. H. Ansar. J. Mol. Struct., 2020, 1219, 128541. https://doi.org/10.1016/j.molstruc.2020.128541
M. J. Berman, B. M. Schmid, D. J. Mendlein, and N. Kaplan. US Patent 20060142265, 2006.
L. Tiwari, V. Kumar, B. Kumar, and D. Mahajan. RSC Adv., 2018, 8, 21585-21595. https://doi.org/10.1039/c8ra03761b
Funding
This work was supported by the Guizhou Provincial Natural Science Foundation ([2020]1Y393) and the Science and Technology Program Platform for Talents of Guizhou Province ([2018]5781).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflict of interests.
Additional information
Russian Text © The Author(s), 2022, published in Zhurnal Strukturnoi Khimii, 2022, Vol. 63, No. 1, pp. 26-29.https://doi.org/10.26902/JSC_id85837
Supplementary material
Rights and permissions
About this article
Cite this article
Luo, R.S., Mao, S.N., Liu, C.J. et al. SYNTHESIS, X-RAY STRUCTURE ANALYSIS, AND VIBRATIONAL SPECTRAL STUDIES OF 1-(3-((6-BROMOPYRIDO[2,3-d]PYRIMIDIN-4-YL) OXY)PHENYL)-3-CYCLOPENTYLUREA. J Struct Chem 63, 37–51 (2022). https://doi.org/10.1134/S002247662201005X
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S002247662201005X