Abstract
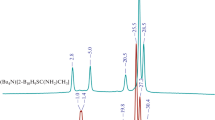
The reaction of sulfonium closo-decaborate derivatives containing carboxyl groups with sulfuryl chloride SO2Cl2 as a halogenating agent was studied. The reaction gave fully halogenated derivatives of the closo-decaborate anion, (n-Bu4N)[2-B10Cl9SR2] (R = –CH2COOEt, –CH2COOH), which was confirmed by 11B, 1H, and 13C NMR and IR spectral data. The structure of the [2-B10Cl9S(CH2COOEt)2]– anion was established using single crystal X-ray diffraction.




Similar content being viewed by others
REFERENCES
Sivaev, I.B., Prikaznov, A.V., and Naoufal, D., Collect. Czech. Chem. Commun., 2010, vol. 75, no. 11, pp. 1149–1199. https://doi.org/10.1135/cccc2010054
Avdeeva, V.V., Malinina, E.A., Sivaev, I.B., Bregadze, V.I., and Kuznetsov, N.T., Crystals, 2016, vol. 6, p. 60. https://doi.org/10.3390/cryst6050060
Avdeeva, V.V., Malinina, E.A., Zhizhin, K.Y., and Kuznetsov, N.T., Russ. J. Inorg. Chem., 2020, vol. 65, no. 4, pp. 514–534. https://doi.org/10.1134/S0036023620040026
Avdeeva, V.V., Polyakova, I.N., Churakov, A.V., Vologzhanina, A.V., Malinina, E.A., Zhizhin, K.Y., and Kuznetsov, N.T., Polyhedron, 2019, vol. 162, pp. 65–70. https://doi.org/10.1016/j.poly.2019.01.051
Kubasov, A.S., Golubev, A.V., Bykov, A.Yu., Matveev, E.Yu., Zhizhin, K.Yu., and Kuznetsov, N.T., J. Mol. Struct., 2021, vol. 1241, p. 130591. https://doi.org/10.1016/j.molstruc.2021.130591
Golubev, A.V., Kubasov, A.S., Bykov, A.Yu., Zhizhin, K.Yu., Kravchenko, E.A., Gippius, A.A., Zhurenko, S.V., Semenova, V.A., Korlyukov, A.A., and Kuznetsov, N.T., Inorg. Chem., 2021, vol. 60, no. 12, pp. 8592–8604. https://doi.org/10.1021/acs.inorgchem.1c00516
Kim, K.C., Reed, C.A., Long, G.S., and Sen, A., J. Am. Chem. Soc., 2002, vol. 124, no. 26, pp. 7662–7663. https://doi.org/10.1021/ja0259990
Reed, C.A., Acc. Chem. Res., 2010, vol. 43, no. 1, pp. 121–128. https://doi.org/10.1021/ar900159e
Ali, F., Hosmane, N.S., and Zhu, Y., Molecules, 2020, vol. 25, no. 4, p. 828. https://doi.org/10.3390/molecules25040828
Olejniczak, A.B., Nawrot, B., and Lesnikowski, Z.J., Int. J. Mol. Sci., 2018, vol. 19, p. 3501. https://doi.org/10.3390/ijms19113501
Kubasov, A.S., Matveev, E.Y., Turyshev, E.S., Polyakova, I.N., Nichugovskiy, A.I., Zhizhin, K.Y., and Kuznetsov, N.T., Inorg. Chim. Acta, 2018, vol. 477, pp. 277–283. https://doi.org/10.1016/j.ica.2018.03.013
Sheldrick, G.M., Acta Crystallogr., Sect. C: Struct. Chem., 2015, vol. 71, pp. 3–8. https://doi.org/10.1107/S2053229614024218
Dolomanov, O.V., Bourhis, L.J., Gildea, R.J., Howard, J.A.K., and Puschmann, H., J. Appl. Crystallogr., 2009, vol. 42, no. 2, pp. 339–341. https://doi.org/10.1107/S0021889808042726
Funding
The study was supported by the Russian Foundation for Basic Research (project no. 20-33-90118). The study was supported by the state assignment for the Kurnakov Institute of General and Inorganic Chemistry, RAS. X-ray diffraction, IR, and NMR measurements were carried out using the research equipment supported by the state assignment for IGIC RAS.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflicts of interest.
Additional information
Translated by Z. Svitanko
This study was submitted to the virtual issue “Young Scientists of the RAS.”
Rights and permissions
About this article
Cite this article
Golubev, A.V., Kubasov, A.S., Bykov, A.Y. et al. Chlorination of Sulfonium Derivatives of closo-Decaborate Anion with Carboxyl-Containing Substituents. Dokl Chem 500, 205–208 (2021). https://doi.org/10.1134/S0012500821100025
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0012500821100025