Abstract
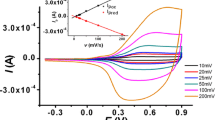
A new acetophenone-functionalized thiophene monomer, 2-hydroxy-6-[2-(thiophene-3-yl)ethoxy]acetophenone, is synthesized by the reaction of 2.6-dihydroxyacetophenone with 2-(thiophene-3-yl)ethane-1-ol under mild conditions. The synthesis is performed in order to study its electrochemical oxidation and prepare a functionalized conducting material for ion sensors with a receptor group for divalent heavy-metal cations. The redox behavior of the functionalized thiophene is studied by simultaneous in situ ESR and Vis-NIR spectroscopy. The formation of soluble thiophene oligomeric structures during the electrochemical oxidation of the monomer is found. Direct evidence for the formation of oligomers is given by ex situ MALDI-TOF and LDI-TOF mass spectrometry. The acetophenone-functionalized monomer and 3-methylthiophene are electrochemically copolymerized and the copolymeric nature of the product is proved by MALDI-TOF mass spectrometry as well. The copolymer film at the electrode surface is characterized in its redox behavior.
Similar content being viewed by others
References
Roncali, J., Chem. Rev., 1992, vol. 92, p. 711.
Chan, H.S.O. and Ng, S.C., Prog. Polym. Sci., 1998, vol. 23, p. 1167.
MacDiarmid, A.G., Angew. Chem., Int. Ed. Engl., 2001, vol. 40, p. 2581.
McQuade, D.T., Pullen, A.E., and Swager, T.M., Chem. Rev., 2000, vol. 100, p. 2537.
Fabre, B. and Simonet, J., Coord. Chem. Rev., 1998, vol. 178, p. 1211.
Cha, S.K. and Abruna, H.D., Anal. Chem., 1990, vol. 62, p. 274.
Townsend, A., Worsfold, P.J., Haswell, S.J., Macrae, R., Werner, H.W., and Wilson, I.D., in Encyclopedia of Analytical Science, London: Academic, 1995, vol. 4, p. 2298.
Gomis, J.A., Morallon, E., Montiel, V., and Aldaz, A., J. Electrochem. Soc., 1996, vol. 143, p. 3166.
Rapta, P., Neudeck, A., Petr, A., and Dunsch, L., J. Chem. Soc., Faraday Trans. 2, 1998, vol. 94, p. 3625.
Rapta, P., Faber, R., Dunsch, L., Neudeck, A., and Nuyken, O., Spectrochim. Acta, 2000, vol. 56A, p. 357.
Rapta, P., Lukkari, J., Tarábek, J., Salomaki, M., Jussila, M., Yohannes, G., Riekkola, M.L., Kankare, J., and Dunsch, L., Phys. Chem. Chem. Phys., 2004, vol. 6, p. 434.
Ruiz, V., Colina, A., Heras, A., Lopez-Palacios, J., and Seeber, R., Helv. Chim. Acta, 2001, vol. 84, p. 3628.
Olbrich-Stock, M., Posdorfer, J., and Schindler, R.N., J. Electroanal. Chem., 1994, vol. 368, p. 173.
Raimundo, J.-M., Levillain, E., Gallego-Planas, N., and Roncali, J., Electrochem. Commun., 2000, vol. 2, p. 211.
Alberti, A., Ballarin, B., Guerra, M., Macciantelli, D., Mucci, A., Parenti, F., Schenetti, L., Seeber, R., and Zanardi, C., Chem. Phys. Chem., 2003, vol. 4, p. 1216.
Vonk, B.C., Langeveld-Voss, B.M.W., van Dongen, J.L.J., Janssen, R.A.J., Claessens, H.A., and Cramers, C.A., J. Chromatogr., A, 2001, vol. 911, p. 13.
van Haare, J.A.E.H., Havinga, E.E., van Dongen, J.L.J., Janssen, R.A.J., Cornil, J., and Brédas, J.-L., Chem.-Eur. J., 1998, vol. 4, p. 1509.
Lemaire, M., Garreau, R., Roncali, J., Delabouglise, D., Youssoufi, H.K., and Gamier, F., New J. Chem., 1989, vol. 13, p. 863.
Roncali, J., Garreau, R., Delabouglise, D., Gamier, F., and Lemaire, M., Synth. Met., 1989, vol. 25, p. 0341.
Tarábek, J., Rapta, P., Jähne, E., Ferse, D., Adler, H.-J., Maumy, M., and Dunsch, L., Electrochim. Acta, 2005, vol. 50, p. 1643.
Author information
Authors and Affiliations
Corresponding author
Additional information
Published in Russian in Elektrokhimiya, 2006, Vol. 42, No. 11, pp. 1302–1311.
Based on the report delivered at the 8th International Frumkin Symposium “Kinetics of the Electrode Processes,” October 18–22, 2005, Moscow.
The text was submitted by the authors in English.
Rights and permissions
About this article
Cite this article
Tarábek, J., Jähne, E., Rapta, P. et al. New acetophenone-functionalized thiophene monomer for conducting films on electrodes in chemical ion-sensorics: The synthesis and spectroelectrochemical study. Russ J Electrochem 42, 1169–1176 (2006). https://doi.org/10.1134/S1023193506110036
Received:
Issue Date:
DOI: https://doi.org/10.1134/S1023193506110036