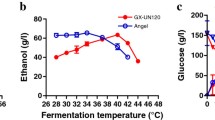
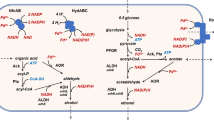
Abstract
Alcohol oxidase (AOX) has been purified 8-fold from a genetically constructed over-producing strain of the methylotrophic yeast Hansenula polymorpha C-105 (gcr1 catX) with impaired glucose-induced catabolite repression and completely devoid of catalase. The final enzyme preparation was homogeneous as judged by polyacrylamide gel electrophoresis and HPLC. Some physicochemical and biochemical properties of AOX were studied in detail: molecular weight (∼620 kD), isoelectric point (pI6.1), and UV-VIS, circular dichroism (CD), and fluorescence spectra. The content of different secondary structure motifs of the enzyme has been calculated from the CD spectra using a computer program. It was found that the native protein contains about 50% α-helix, 25% β-sheet, and about 20% random structures. The kinetic parameters for different substrates, such as methanol, ethanol, and formaldehyde, were measured using a Clark oxygen electrode. The rate of enzymatic oxidation of formaldehyde by alcohol oxidase from H. polymorpha is only twice lower compared to the best substrate of the enzyme, methanol.
Similar content being viewed by others
References
Vonck, J., and van Bruggen, E. F. J. (1990) Biochim. Biophys. Acta, 1038, 74–79.
Wierenga, R. K., Terpstra, P., and Hol, W. G. J. (1986) J. Mol. Biol., 187, 101–107.
Averbakh, A. Z., Pekel, N. D., Seredenko, V. I., Kulikov, A. V., Gvozdev, R. I., and Rudakova, I. P. (1995) Biochem. J., 310, 601–614.
Fujii, T., and Tonomura, K. (1975) Agric. Biol. Chem., 39, 2325–2330.
Kato, N., Omori, Y., Tani, Y., and Ogata, K. (1976) Eur. J. Biochem., 64, 341–350.
Couderc, R., and Baratti, J. (1980) Agric. Biol. Chem., 44, 2279–2289.
Verduyn, C., van Dijken, J. P., and Scheffers, W. A. (1983) Biotechnol. Bioeng., 25, 1049–1055.
Guilbault, G. G., Palleschi, G., and Lubrano, G. (1995) Biosens. Bioelectron., 10, 379–392.
Nanjo, M., and Guilbault, G. G. (1975) Anal. Chim. Acta, 75, 169–180.
Chen, H. F., Chen, T. J., and Fang, H. Y. (1988) Zhongguo Nongye Huaxue Huizhi, 26, 287–301.
Azevedo, A. M., Cabral, J. M. S., Prazeres, D. M. F., Gibson, T. D., and Fonseca, L. P. (2004) J. Mol. Cat. B: Enz., 27, 37–45.
Gonchar, M. V., Ksheminska, G. P., Hladarevska, N. M., and Sibirny, A. A. (1980) in Genetics of Respiratory Enzymes in Yeasts (Lachowicz, T. M., ed.) Proc. Int. Conf., Wroclaw University Press, Wroclaw, pp. 222–228.
Gonchar, M. V., Kostryk, L. B., and Sibirny, A. A. (1997) Appl. Microbiol. Biotechnol., 48, 454–458.
Sibirnyi, A. A., Titorenko, V. I., Efremov, B. D., and Tolstorukov, I. I. (1987) Yeast, 33, 233–241.
Ehresmann, B., Imbault, P., and Weil, J. H. (1973) Analyt. Biochem., 54, 454–463.
Davis, B. J. (1964) Ann. NY Acad. Sci., 121, 404–427.
Laemmli, U. K. (1970) Nature, 227, 680–685.
Osterman, L. A. (1983) Study of Biological Macromolecules by Electrofocusing, Immunoelectrophoresis, and Radioisotopic Methods [in Russian], Nauka, Moscow.
Shleev, S. V., Morozova, O. V., Nikitina, O. V., Gorshina, E. S., Rusinova, T. V., Serezhenkov, V. A., Burbaev, D. S., Gazaryan, I. G., and Yaropolov, A. (2004) Biochimie, 86, 693–703.
Suye, S.-I. (1997) Curr. Microbiol., 34, 374–377.
Hopkins, T. R., and Muller, F. (1987) in Microbial Growth on C1 Components (van Verseveld, H. W., and Duine, J. A., eds.) Martinus Vijholf Publishers, Dordrecht, pp. 150–157.
Sahm, H., and Wagner, F. (1973) Eur. J. Biochem., 36, 250–256.
Whitby, L. G. (1953) Biochem. J. 54, 437–442.
Bringer, S., Sprey, B., and Sahm, H. (1979) Eur. J. Biochem., 101, 563–570.
Tani, Y., Miya, T., and Ogata, K. (1972) Agr. Biol. Chem., 36, 76–83.
Maskevich, A. A., Artsukevich, I. M., and Stepuro, V. I. (1997) J. Mol. Struct., 408/409, 261–264.
Maskevich, A. A., Artsukevich, I. M., Maskevich, S. A., and Chernikevich, I. P. (1997) Bioorg. Khim., 23, 479–485.
Patel, R. N., Hou, C. T., Laskin, A. I., and Derelanko, P. (1981) Arch. Biochem. Biophys., 210, 481–488.
Dickinson, F. M., and Wadforth, C. (1992) Biochem. J., 282, 325–331.
Kabsch, W., and Sander, C. (1983) Biopolymers, 22, 2577–2637.
Woodward, J. R. (1990) Adv. Autotroph. Microbiol. OneCarbon Metab., 1, 193–225.
Author information
Authors and Affiliations
Additional information
Published in Russian in Biokhimiya, 2006, Vol. 71, No. 3, pp. 312–319.
Originally published in Biochemistry (Moscow) On-Line Papers in Press, as Manuscript BM05-087, December 18, 2005.
Rights and permissions
About this article
Cite this article
Shleev, S.V., Shumakovich, G.P., Nikitina, O.V. et al. Purification and characterization of alcohol oxidase from a genetically constructed over-producing strain of the methylotrophic yeast Hansenula polymorpha . Biochemistry (Moscow) 71, 245–250 (2006). https://doi.org/10.1134/S0006297906030035
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1134/S0006297906030035