Abstract
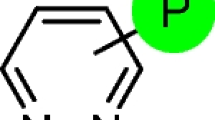
Reaction of diethyl chlorophosphate with aminopyridines furnished a series of new diethyl phosphoramidates. The effect on the reactivity of substituents in the pyridine ring was studied. Structure of the obtained compound was characterized by 1H, 13C, 31P, 2D HMBC 1H–15N NMR spectroscopy, mass-spectrometry, and single crystal X-ray diffraction analysis.


Similar content being viewed by others
REFERENCES
Demkowicz, S., Rachon, J., Daśko, M., and Kozak, W., RSC Adv., 2016, vol. 6, no. 9, p. 7101. https://doi.org/10.1039/C5RA25446A
Yu, H., Yang, H., Shi, E., and Tang, W., Med. Drug Discov., 2020, vol. 8, Art. no. 100063. https://doi.org/10.1016/j.medidd.2020.100063
McGuigan, С., Murziani, P., Slusarczyk, M., Gonczy, B., Voorde, J.V., Liekens, S., and Balzarini, J., J. Med. Chem., 2011, vol. 54, no. 20, p. 7247. https://doi.org/10.1021/jm200815w
Mehellou, Y., Balzarini, J., McGuigan, C., ChemMedChem, 2009, vol. 4, no. 11, p. 1779. https://doi.org/10.1002/cmdc.200900289
Maiti, M., Persoons, L., Andrei, G., Snoeck, R., Balzarini, J., and Herdewijn, P., ChemMedChem, 2013, vol. 8, no. 6, p. 985. https://doi.org/10.1002/cmdc.201300035
Reader, J.S., Ordoukhanian, P.T., Kim, J.-G., de Crecy-Lagard, V., Hwang, I., Farrand, S., and Schimmel, P., Science, 2005, vol. 309, no. 5740, p. 1533. https://doi.org/10.1126/science.1116841
Nguyen, T.-M., Chang, S., Condon, B., Slopek, R., Graves, E., and Yoshioka-Tarver, M., Ind. Eng. Chem. Res., 2013, vol. 52, no. 13, p. 4715. https://doi.org/10.1021/ie400180f
Neisius, M., Liang, S., Mispreuve, H., and Gaan, S., Ind. Eng. Chem. Res., 2013, vol. 52, no. 29, p. 9752. https://doi.org/10.1021/ie400914u
Van Overtveldt, M., Heugebaert, T.S.A., Verstraeten, I., Geelen, D., and Stevens, C.V., Org. Biomol. Chem., 2015, vol. 13, no. 18, p. 5260. https://doi.org/10.1039/c5ob00137d
Altaff, S.K.M., Rajeswari, T.R., and Subramanyam, C., Phosphorus, Sulfur, Silicon, Relat. Elem., 2021, vol. 196, no. 4, p. 389. https://doi.org/10.1080/10426507.2020.1845679
Kumar, M.P.P., Anuradha, V., Subramanyam, C., and Babu, V.V.H., Phosphorus, Sulfur, Silicon, Relat. Elem., 2021, vol. 196, no. 11, p. 1010. https://doi.org/10.1080/10426507.2021.1960833
Addanki, H.R., Vallabhaneni, M.R., Chennamsetty, S., Pullagura, P., Sagurthi, S.R., and Pasupuleti, V.R., Synth. Commun. 2022, vol. 52, no. 2, p. 300. https://doi.org/10.1080/00397911.2021.2024574
Gao, X., Tang, Z., Lu, M., Liu, H., Jiang, Y., Zhao, Y., and Cai, Z., Chem. Commun., 2012, vol. 48, no. 82, p. 10198. https://doi.org/10.1039/c2cc36091h
Yadav, L.D.S., Awasthi, C., Rai, V.K., and Rai, A., Tetrahedron Lett., 2007, vol. 48, no. 45, p. 8037. https://doi.org/10.1016/j.tetlet.2007.09.044
Yadav, L.D.S., Srivastava, V.P., and Patel, R., Tetrahedron Lett., 2008, vol. 49, no. 39, p. 5652. https://doi.org/10.1016/j.tetlet.2008.07.069
Dwek, R.A., Chem. Rev., 1996, vol. 96, no. 2, p. 683. https://doi.org/10.1021/cr940283b
Garcia, P., Lau, Y.Y., Perry, M.R., and Schafer, L.L., Angew. Chem. Int. Ed., 2013, vol. 52, no. 35, p. 9144. https://doi.org/10.1002/anie.201304153
Ding, M., Zhou, F., Liu, Y.-L., Wang, C.-H., Zhao, X.-L., and Zhou, J., Chem. Sci., 2011, vol. 2, no. 10, p. 2035. https://doi.org/10.1039/c1sc00390a
Yu, J.-S., Liao, F.-M., Gao, W.-M., Liao, K., Zuo, R.-L., and Zhou, J., Angew. Chem. Int. Ed., 2015, vol. 54, no. 25, p. 7381. https://doi.org/10.1002/anie.201501747
Nishikawa, Y., Nakano, S., Tahira, Y., Terazawa, K., Yamazaki, K., Kitamura, C., and Hara, O., Org. Lett., 2016, vol. 18, no. 9, p. 2004. https://doi.org/10.1021/acs.orglett.6b00608
Predvoditelev, D.A., Suvorkin, S.V., and Nifant’ev, E.E., Russ. J. Gen. Chem., 2001, vol. 71, no. 6, p. 873. https://doi.org/10.1023/A:1012319132072
Luff, B.D.W. and Kipping, F.S., J. Chem. Soc. Trans., 1909, vol. 95, p. 1993. https://doi.org/10.1039/ct9099501993
Le Corre, S.S., Berchel, M., Couthon-Gourvès, H., Haelters, J.-P., and Jaffrès, P.-A., Beilstein J. Org. Chem., 2014, vol. 10, p. 1166. https://doi.org/10.3762/bjoc.10.117
Dangroo, N.A., Dar, A.A., Shankar, R., Khuroo, M.A., and Sangwan, P.L., Tetrahedron Lett., 2016, vol. 57, no. 25, p. 2717. https://doi.org/10.1016/j.tetlet.2016.05.003
Pan, C., Jin, N., Zhang, H., Han, J., and Zhu, C., J. Org. Chem., 2014, vol. 79, p. 9427. https://doi.org/10.1021/jo5018052
Meazza, M., Kowalczuk, A., Shirley, L., Yang, J.W., Guo, H., and Rios, R., Adv. Synth. Catal., 2016, vol. 358, no. 5, p. 719. https://doi.org/10.1002/adsc.201501068
Dhineshkumar, J. and Prabhu, K.R., Org. Lett., 2013, vol. 15, no. 23, p. 6062. https://doi.org/10.1021/ol402956b
Dar, B.A., Dangroo, N.A., Gupta, A., Wali, A., Khuroo, M.A., Vishwakarma, R.A., and Singh, B., Tetrahedron Lett., 2014, vol. 55, no. 9, p. 1544. https://doi.org/10.1016/j.tetlet.2014.01.064
Chen, X., Xiao, Z., Chu, H., Wang, B., and Peng, A.-Y., Org. Biomol. Chem., 2018, vol. 16, no. 36, p. 6783. https://doi.org/10.1039/c8ob01840e
Metlushka, K.E., Sadkova, D.N., Nikitina, K.A., Shaimardanova, L.N., Kataeva, L.N., and Alfonsov, V.A., Russ. J. Gen. Chem., 2016, vol. 86, no. 3, p. 515. https://doi.org/10.1134/S107036321603004X
Kaboudin, B., Karimi, M., and Zahedi, H., Org. Prep. Proced. Int., 2008, vol. 40, no. 4, p. 399. https://doi.org/10.1080/00304940809458101
Sardarian, A.R. and Shahsavari-Fard, Z., Synlett, 2008, vol. 9, p. 1391. https://doi.org/10.1055/s-2008-1072767
Zarei, M., J. Chem. Res., 2012, vol. 36, no. 2, p. 118. https://doi.org/10.3184/174751912X13282660033937
Yarovenko, V.N., Shirokov, A.V., Zavarzin, I.V., Krupinova, O.N., Ignatenko, A.V., and Krayushkin, M.M., Synthesis, 2004, no. 1, p. 17. https://doi.org/10.1055/s-2003-44377
McNulty, J., Krishnamoorthy, V., and Robertson, A., Tetrahedron Lett., 2008, vol. 49, no. 44, p. 6344. https://doi.org/10.1016/j.tetlet.2008.08.074
Lee, H.-W., Kang, T.W., Cha, K.H., Kim, E.-N., Choi, N.-H., Kim, J.-W., and Hong, C.I., Synth. Commun., 1998, vol. 28, no. 1, p. 35. https://doi.org/10.1080/00397919808005071
Zhang, W., Zhu, Y., Wei, D., Zhang, C., Sun, D., and Tang, M., Comp. Theor. Chem., 2011, vol. 963, no. 1, p. 13. https://doi.org/10.1016/j.comptc.2010.09.003
Rao, R.N. and Chanda, K., Chem. Commun., 2022, vol. 58, no. 3, p. 343. https://doi.org/10.1039/D1CC04602K
Sedehizadeh, S., Keogh, M., and Maddison, P., Clin. Neuropharmacol., 2012, vol. 35, no. 4, p. 191. https://doi.org/10.1097/wnf.0b013e31825a68c5
Krutikov, V.I., Erkin, A.V., and Krutikova, V.V., Russ. J. Gen. Chem., 2012, vol. 82, no. 5, p. 822. https://doi.org/10.1134/S1070363212050039
Kostadinova, I. and Danchev, N., Pharmacia, 2019, vol. 66, no. 2, p. 67. https://doi.org/10.3897/pharmacia.66.e35976
Deady, L.W. and Stillman, D.C., Aust. J. Chem., 1978, vol. 31, no. 8, p. 1725. https://doi.org/10.1071/ch9781725
Deady, L.W. and Stillman, D.C., Aust. J. Chem., 1979, vol. 32, no. 2, p. 381. https://doi.org/10.1071/ch9790381
Arbusov, A.E., J. Org. Chem. USSR, 1966, vol. 2, no. 12, p. 2148.
Chamberland, S., Lee, V.J., Leger, R., and Malouin, F., US Patent WO 2008/113161 A1, 2008.
CrysAlisPro, Rigaku Oxford Diffraction, Version 1.171.41.103a, 2021.
Sheldrick, G.M., Acta Crystallogr. (A), 2015, vol. 71, p. 3. https://doi.org/10.1107/S2053273314026370
Sheldrick, G.M., Acta Crystallogr. (C), 2015, vol. 71, p. 3. https://doi.org/10.1107/S2053229614024218
Dolomanov, O.V., Bourhis, L.J., Gildea, R.J., Howard, J.A.K., and Puschmann, H., J. Appl. Cryst., 2009, vol. 42, p. 339. https://doi.org/10.1107/S0021889808042726
Funding
This work was financially supported by the Ministry of Science and Higher Education of the Russian Federation (project no. 0785.00.X6019) using the equipment of the Engineering Center of the St. Petersburg State Technological Institute and the Resource Centers “X-ray diffraction methods of research” and “Methods of analysis of the composition of matter” of the Science Park of St. Petersburg State University.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
No conflict of interest was declared by the authors.
Supplementary information
Rights and permissions
About this article
Cite this article
Krylov, A.S., Piterskaya, Y.L., Gurzhiy, V.V. et al. Synthesis of Phosphoramidates Based on Aminopyridines. Russ J Gen Chem 92, 1912–1918 (2022). https://doi.org/10.1134/S1070363222100061
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070363222100061