Abstract
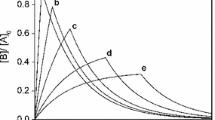
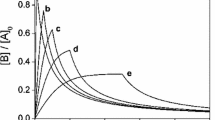
A new method is suggested for solving the inverse problem of chemical kinetics. This method provides means to derive, from unsteady-state experimental data, the rate constants of catalytic reactions in which each step involves at least one main reactant. Examples of applying this method to two- and three-step reactions are presented.
Similar content being viewed by others
References
G. S. Yablonskii and S. I. Spivak, Mathematical Models of Chemical Kinetics (Znanie, Moscow, 1977) [in Russian].
V. N. Lukashenok, Teor. Osn. Khim. Tekhnol. 14, 86 (1980).
S. I. Spivak and V. G. Gorskii, Dokl. Akad. Nauk SSSR 257, 412 (1981).
L. S. Polak, M. Ya. Goldenberg, and A. A. Levitskii, Computational Methods in Chemical Kinetics (Nauka, Moscow, 1984) [in Russian].
S. I. Spivak and A. S. Ismagilova, Inverse Problems of Chemical Kinetics (Lap Lambert Academic, Saarbrücken, 2013).
M. I. Temkin, Kinet. Katal. 17, 1095 (1976).
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © V.Kh. Fedotov, N.I. Kol’tsov, 2016, published in Khimicheskaya Fizika, 2016, Vol. 35, No. 10, pp. 9–15.
Rights and permissions
About this article
Cite this article
Fedotov, V.K., Kol’tsov, N.I. Method of solving the inverse problem of chemical kinetics for catalytic reactions in which each step involves main reactants. Russ. J. Phys. Chem. B 10, 753–759 (2016). https://doi.org/10.1134/S1990793116050195
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1990793116050195